Abstract
Purpose
Glioblastoma multiforme (GBM) is one of the most aggressive human cancers. The c-MET receptor tyrosine kinase (RTK) which is frequently deregulated in GBM is considered as a promising target for GBM treatment. The c-MET plays a key role in cell proliferation, cell cycle progression, invasion, angiogenesis, and metastasis. Here, we investigated the anti-tumour activity of foretinib, a c-MET inhibitor, on three human GBM cells (T98G, U87MG and U251).
Methods
Anti-proliferative effect of foretinib was determined using MTT, crystal violet staining, and clonogenic assays. PI and Annexin V/PI staining flow cytometry were used to evaluate the effects of foretinib on cell cycle and apoptosis, respectively. Scratch assay, qRT-PCR, western blot, and zymography analyses were applied to elucidate the molecular mechanisms underlying the anti-tumour activity of foretinib.
Results
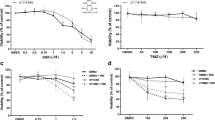
Foretinib treatment reduced phosphorylation of c-MET on T98G and U251 cells, but not in U87MG cells. The highest inhibitory effect was observed in T98G cells (IC50 = 4.66 ± 0.29 µM) and the lowest one in U87MG cells (IC50 = 29.99 ± 1.31 µM). The results showed that foretinib inhibited the proliferation of GBM cells through a G2/M cell cycle arrest and mitochondrial-mediated apoptosis in association with alternation in expression of the related genes and protein-regulated G2/M phase and apoptosis. Foretinib diminished GBM cell invasion through downregulation of the proteolytic cascade of MMP2, uPA and uPAR and epithelial–mesenchymal transition (EMT)-related genes. A different GBM cell sensitivity pattern was noticeable in all experiments which demonstrated T98G as a sensitive and U87MG as a resistant phenotype to foretinib treatment.
Conclusion
The results indicated that foretinib might have the therapeutic potential against human GBM which deserve further investigation.








Similar content being viewed by others
References
Haas-Kogan D, Shalev N, Wong M, Mills G, Yount G, Stokoe D (1998) Protein kinase B (PKB/Akt) activity is elevated in glioblastoma cells due to mutation of the tumor suppressor PTEN/MMAC. Curr Biol 8(21):1195-S1191
Thakkar JP, Dolecek TA, Horbinski C, Ostrom QT, Lightner DD, Barnholtz-Sloan JS, Villano JL (2014) Epidemiologic and molecular prognostic review of glioblastoma. Cancer Epidemiol Prev Biomarkers 23(8):2199
Tamimi AF, Juweid M (2017) Epidemiology and outcome of glioblastoma. In: Glioblastoma. Codon Publications
Omuro A, DeAngelis LM (2013) Glioblastoma and other malignant gliomas: a clinical review. JAMA 310(17):1842–1850
Pearson JR, Regad T (2017) Targeting cellular pathways in glioblastoma multiforme. Signal Transduct Target Ther 2:17040
Cruickshanks N, Zhang Y, Yuan F, Pahuski M, Gibert M, Abounader R (2017) Role and therapeutic targeting of the HGF/MET pathway in glioblastoma. Cancers 9(7):87
Hanif F, Muzaffar K, Perveen K, Malhi SM, Simjee SU (2017) Glioblastoma multiforme: a review of its epidemiology and pathogenesis through clinical presentation and treatment. Asian Pac J Cancer Prev 18(1):3
Puccini A, Marín-Ramos NI, Bergamo F, Schirripa M, Lonardi S, Lenz H-J, Loupakis F, Battaglin F (2019) Safety and tolerability of c-MET inhibitors in cancer. Drug Saf 42(2):1–23
Maulik G, Kijima T, Ma PC, Ghosh SK, Lin J, Shapiro GI, Schaefer E, Tibaldi E, Johnson BE, Salgia R (2002) Modulation of the c-Met/hepatocyte growth factor pathway in small cell lung cancer. Clin Cancer Res 8(2):620–627
Sawada K, Radjabi AR, Shinomiya N, Kistner E, Kenny H, Becker AR, Turkyilmaz MA, Salgia R, Yamada SD, Vande Woude GF, Tretiakova MS, Lengyel E (2007) c-Met overexpression is a prognostic factor in ovarian cancer and an effective target for inhibition of peritoneal dissemination and invasion. Can Res 67(4):1670–1679. https://doi.org/10.1158/0008-5472.can-06-1147
Chen G-Z, Dai W-S, Zhu H-C, Song H-M, Yang X, Wang Y-D, Min H, Lu Q, Liu S, Sun X-C (2017) Foretinib enhances the radiosensitivity in esophageal squamous cell carcinoma by inhibiting phosphorylation of c-Met. J Cancer 8(6):983
Petterson SA, Dahlrot RH, Hermansen SK, Munthe SK, Gundesen MT, Wohlleben H, Rasmussen T, Beier CP, Hansen S, Kristensen BW (2015) High levels of c-Met is associated with poor prognosis in glioblastoma. J Neurooncol 122(3):517–527
Nabeshima K, Shimao Y, Sato S, Kataoka H, Moriyama T, Kawano H, Wakisaka S, Koono M (1997) Expression of c-Met correlates with grade of malignancy in human astrocytic tumours: an immunohistochemical study. Histopathology 31(5):436–443
Shah MA, Wainberg ZA, Catenacci DV, Hochster HS, Ford J, Kunz P, Lee F-C, Kallender H, Cecchi F, Rabe DC (2013) Phase II study evaluating 2 dosing schedules of oral foretinib (GSK1363089), cMET/VEGFR2 inhibitor, in patients with metastatic gastric cancer. PLoS ONE 8(3):e54014
Qian F, Engst S, Yamaguchi K, Yu P, Won K-A, Mock L, Lou T, Tan J, Li C, Tam D (2009) Inhibition of tumor cell growth, invasion, and metastasis by EXEL-2880 (XL880, GSK1363089), a novel inhibitor of HGF and VEGF receptor tyrosine kinases. Cancer Res 69(20):8009–8016
Joo KM, Jin J, Kim E, Kim KH, Kim Y, Kang BG, Kang Y-J, Lathia JD, Cheong KH, Song PH (2012) MET signaling regulates glioblastoma stem cells. Cancer Res 72(15):3828–3838
Zillhardt M, Park S-M, Romero IL, Sawada K, Montag AG, Krausz T, Yamada SD, Peter ME, Lengyel E (2011) Foretinib (GSK1363089), an orally available multi-kinase inhibitor of c-Met and VEGFR, blocks proliferation, induces anoikis, and impairs ovarian cancer metastasis. Clin Cancer Res 3387:2010
Chen H-M, Tsai C-H, Hung W-C (2015) Foretinib inhibits angiogenesis, lymphangiogenesis and tumor growth of pancreatic cancer in vivo by decreasing VEGFR-2/3 and TIE-2 signaling. Oncotarget 6(17):14940
Naing A, Kurzrock R, Adams LM, Kleha JF, Laubscher KH, Bonate PL, Weller S, Fitzgerald C, Xu Y, LoRusso PM (2012) A comparison of the pharmacokinetics of the anticancer MET inhibitor foretinib free base tablet formulation to bisphosphate salt capsule formulation in patients with solid tumors. Invest New Drugs 30(1):327–334
Zhang Y, Xia M, Jin K, Wang S, Wei H, Fan C, Wu Y, Li X, Li X, Li G (2018) Function of the c-Met receptor tyrosine kinase in carcinogenesis and associated therapeutic opportunities. Mol Cancer 17(1):45
Logan TF (2013) Foretinib (XL880): c-MET inhibitor with activity in papillary renal cell cancer. Curr Oncol Rep 15(2):83–90
Seiwert T, Sarantopoulos J, Kallender H, McCallum S, Keer HN, Blumenschein G (2013) Phase II trial of single-agent foretinib (GSK1363089) in patients with recurrent or metastatic squamous cell carcinoma of the head and neck. Invest New Drugs 31(2):417–424
Choueiri TK, Vaishampayan U, Rosenberg JE, Logan TF, Harzstark AL, Bukowski RM, Rini BI, Srinivas S, Stein MN, Adams LM (2013) Phase II and biomarker study of the dual MET/VEGFR2 inhibitor foretinib in patients with papillary renal cell carcinoma. J Clin Oncol 31(2):181
Leighl NB, Tsao M-S, Liu G, Tu D, Ho C, Shepherd FA, Murray N, Goffin JR, Nicholas G, Sakashita S (2017) A phase I study of foretinib plus erlotinib in patients with previously treated advanced non-small cell lung cancer: Canadian cancer trials group IND 196. Oncotarget 8(41):69651
Eder JP, Shapiro GI, Appleman LJ, Zhu AX, Miles D, Keer H, Cancilla B, Chu F, Hitchcock-Bryan S, Sherman L (2010) A phase I study of foretinib, a multi-targeted inhibitor of c-Met and vascular endothelial growth factor receptor 2. Clin Cancer Res 16(13):3507–3516
Shapiro GI, McCallum S, Adams LM, Sherman L, Weller S, Swann S, Keer H, Miles D, Müller T, LoRusso P (2013) A Phase 1 dose-escalation study of the safety and pharmacokinetics of once-daily oral foretinib, a multi-kinase inhibitor, in patients with solid tumors. Invest New Drugs 31(3):742–750
Bender S, Gronych J, Warnatz H-J, Hutter B, Gröbner S, Ryzhova M, Pfaff E, Hovestadt V, Weinberg F, Halbach S (2016) Recurrent MET fusion genes represent a drug target in pediatric glioblastoma. Nat Med 22(11):1314
Knubel KH, Pernu BM, Sufit A, Nelson S, Pierce AM, Keating AK (2014) MerTK inhibition is a novel therapeutic approach for glioblastoma multiforme. Oncotarget 5(5):1338
Davare MA, Saborowski A, Eide CA, Tognon C, Smith RL, Elferich J, Agarwal A, Tyner JW, Shinde UP, Lowe SW (2013) Foretinib is a potent inhibitor of oncogenic ROS1 fusion proteins. Proc Natl Acad Sci USA 110(48):19519–19524
Feoktistova M, Geserick P (2016) Leverkus M (2016) Crystal violet assay for determining viability of cultured cells. Cold Spring Harb Protoc 4:prot087379
Franken NA, Rodermond HM, Stap J, Haveman J, Van Bree C (2006) Clonogenic assay of cells in vitro. Nat Protoc 1(5):2315
Toth M, Sohail A, Fridman R (2012) Assessment of gelatinases (MMP-2 and MMP-9) by gelatin zymography. In: Metastasis research protocols. Springer. pp 121–135
Stark GR, Taylor WR (2006) Control of the G 2/M transition. Mol Biotechnol 32(3):227–248
Archambault V, Carmena M (2012) Polo-like kinase-activating kinases: aurora A, aurora B and what else? Cell Cycle 11(8):1490–1495
Laoukili J, Kooistra MR, Brás A, Kauw J, Kerkhoven RM, Morrison A, Clevers H, Medema RH (2005) FoxM1 is required for execution of the mitotic programme and chromosome stability. Nat Cell Biol 7(2):126–136
Dibb M, Han N, Choudhury J, Hayes S, Valentine H, West C, Ang Y, Sharrocks A (2012) The FOXM1-PLK1 axis is commonly upregulated in oesophageal adenocarcinoma. Br J Cancer 107(10):1766–1775
Gross A, McDonnell JM, Korsmeyer SJ (1999) BCL-2 family members and the mitochondria in apoptosis. Genes Dev 13(15):1899–1911
Garg H, Suri P, Gupta JC, Talwar G, Dubey S (2016) Survivin: a unique target for tumor therapy. Cancer Cell Int 16(1):49
Hatoum A, Mohammed R, Zakieh O (2019) The unique invasiveness of glioblastoma and possible drug targets on extracellular matrix. Cancer Manag Res 11:1843
Stemmler MP, Eccles RL, Brabletz S, Brabletz T (2019) Non-redundant functions of EMT transcription factors. Nat Cell Biol 21(1):102–112
Awad AJ, Burns TC, Zhang Y, Abounader R (2014) Targeting MET for glioma therapy. Neurosurg Focus 37(6):E10
Organ SL, Tsao M-S (2011) An overview of the c-MET signaling pathway. Ther Adv Med Oncol 3(1 suppl):S7–S19
Kwak Y, Kim S-I, Park C-K, Paek SH, Lee S-T, Park S-H (2015) c-MET overexpression and amplification in gliomas. Int J Clin Exp Pathol 8(11):14932
Liu W, Fu Y, Xu S, Ding F, Zhao G, Zhang K, Du C, Pang B, Pang Q (2011) c-Met expression is associated with time to recurrence in patients with glioblastoma multiforme. J Clin Neurosci 18(1):119–121
Simiczyjew A, Dratkiewicz E, Van Troys M, Ampe C, Styczeń I, Nowak D (2018) Combination of EGFR inhibitor lapatinib and MET inhibitor foretinib inhibits migration of triple negative breast cancer cell lines. Cancers 10(9):335
Dratkiewicz E, Pietraszek-Gremplewicz K, Simiczyjew A, Mazur AJ, Nowak D (2018) Gefitinib or lapatinib with foretinib synergistically induce a cytotoxic effect in melanoma cell lines. Oncotarget 9(26):18254
Moghimi M, Sadeghi Tafti H, Namazi F, Salehi M (2019) Investigation of the impact of foretinib on AURKA and AURKB expression in T98 glioblastoma cell line. Res Mol Med 7(1):1–7
Maachani UB, Shankavaram U, Kramp T, Tofilon PJ, Camphausen K, Tandle AT (2016) FOXM1 and STAT3 interaction confers radioresistance in glioblastoma cells. Oncotarget 7(47):77365
Zhang X, Qiao-Li L, Huang Y-T, Zhang L-H, Zhou H-H (2017) Akt/FoxM1 signaling pathway-mediated upregulation of MYBL2 promotes progression of human glioma. J Exp Clin Cancer Res 36(1):105
Mancini M, De Santis S, Monaldi C, Bavaro L, Martelli M, Castagnetti F, Gugliotta G, Rosti G, Santucci M, Martinelli G (2019) Hyper-activation of aurora kinase a-polo-like kinase 1-FOXM1 axis promotes chronic myeloid leukemia resistance to tyrosine kinase inhibitors. J Exp Clin Cancer Res 38(1):1–11
Chakravarti A, Zhai GG, Zhang M, Malhotra R, Latham DE, Delaney MA, Robe P, Nestler U, Song Q, Loeffler J (2004) Survivin enhances radiation resistance in primary human glioblastoma cells via caspase-independent mechanisms. Oncogene 23(45):7494
Li F, Ambrosini G, Chu EY, Plescia J, Tognin S, Marchisio PC, Altieri DC (1998) Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 396(6711):580–584. https://doi.org/10.1038/25141
Zhang S, Zhang C, Song Y, Zhang J, Xu J (2018) Prognostic role of survivin in patients with glioma. Medicine 97(17):e0571
Kesanakurti D, Chetty C, Maddirela DR, Gujrati M, Rao J (2012) Functional cooperativity by direct interaction between PAK4 and MMP-2 in the regulation of anoikis resistance, migration and invasion in glioma. Cell Death Dis 3(12):e445
Badiga AV, Chetty C, Kesanakurti D, Are D, Gujrati M, Klopfenstein JD, Dinh DH, Rao JS (2011) MMP-2 siRNA inhibits radiation-enhanced invasiveness in glioma cells. PLoS ONE 6(6):e20614
Gondi CS, Kandhukuri N, Dinh DH, Gujrati M, Rao JS (2007) Down-regulation of uPAR and uPA activates caspase-mediated apoptosis and inhibits the PI3K/AKT pathway. Int J Oncol 31(1):19–27
Fotoohi M, Hadi N, Namazi F (2019) Effect of foretinib on matrix metalloproteinase-2 (MMP2) expression in glioblastoma. Res Mol Med 6(4):20–27
Mikheeva SA, Mikheev AM, Petit A, Beyer R, Oxford RG, Khorasani L, Maxwell J-P, Glackin CA, Wakimoto H, González-Herrero I (2010) TWIST1 promotes invasion through mesenchymal change in human glioblastoma. Mol Cancer 9(1):194
Han S-P, Kim J-H, Han M-E, Sim H-E, Kim K-S, Yoon S, Baek S-Y, Kim B-S, Oh S-O (2011) SNAI1 is involved in the proliferation and migration of glioblastoma cells. Cell Mol Neurobiol 31(3):489–496
Oh S-J, Ahn E-J, Kim O, Kim D, Jung T-Y, Jung S, Lee J-H, Kim K-K, Kim H, Kim EH (2019) The role played by SLUG, an epithelial-mesenchymal transition factor, in invasion and therapeutic resistance of malignant glioma. Cell Mol Neurobiol 56:1–14
Sottoriva A, Spiteri I, Piccirillo SG, Touloumis A, Collins VP, Marioni JC, Curtis C, Watts C, Tavaré S (2013) Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proc Natl Acad Sci 110(10):4009–4014
Patel AP, Tirosh I, Trombetta JJ, Shalek AK, Gillespie SM, Wakimoto H, Cahill DP, Nahed BV, Curry WT, Martuza RL (2014) Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 344(6190):1396–1401
Parker NR, Khong P, Parkinson JF, Howell VM, Wheeler HR (2015) Molecular heterogeneity in glioblastoma: potential clinical implications. Front Oncol 5:55
Noch EK, Ramakrishna R, Magge R (2018) Challenges in the treatment of glioblastoma: multisystem mechanisms of therapeutic resistance. World Neurosurg 116:505–517
Akbarnejad Z, Eskandary H, Dini L, Vergallo C, Nematollahi-Mahani SN, Farsinejad A, Abadi MFS, Ahmadi M (2017) Cytotoxicity of temozolomide on human glioblastoma cells is enhanced by the concomitant exposure to an extremely low-frequency electromagnetic field (100 Hz, 100 G). Biomed Pharmacother 92:254–264
Lee C-Y, Lai H-Y, Chiu A, Chan S-H, Hsiao L-P, Lee S-T (2016) The effects of antiepileptic drugs on the growth of glioblastoma cell lines. J Neurooncol 127(3):445–453
Ryu CH, Yoon WS, Park KY, Kim SM, Lim JY, Woo JS, Jeong CH, Hou Y, Jeun S-S (2012) Valproic acid downregulates the expression of MGMT and sensitizes temozolomide-resistant glioma cells. BioMed Res Int
Messaoudi K, Clavreul A, Lagarce F (2015) Toward an effective strategy in glioblastoma treatment. Part I: resistance mechanisms and strategies to overcome resistance of glioblastoma to temozolomide. Drug Discov Today 20(7):899–905
Towner RA, Ihnat M, Saunders D, Bastian A, Smith N, Pavana RK, Gangjee A (2015) A new anti-glioma therapy, AG119: pre-clinical assessment in a mouse GL261 glioma model. BMC Cancer 15(1):522
Acknowledgements
This work was supported by Cancer Biology Research Center in Cancer Institute of I.R. Iran at Tehran University of Medical Sciences and also partially supported by National Institute for Medical Research Development (NIMAD) Project No. 963331. Special thanks goes to Dr. Majid Momeny and Farima Moghaddaskho for the preliminary work on this project, Dr. Mohammad Hossein Ghahremani and Dr. Amir Shadboorestan for the technical support in western blot experiments, and MS Raziyeh khoshsohbat for the administrative support.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors disclose no potential conficts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gortany, N.K., Panahi, G., Ghafari, H. et al. Foretinib induces G2/M cell cycle arrest, apoptosis, and invasion in human glioblastoma cells through c-MET inhibition. Cancer Chemother Pharmacol 87, 827–842 (2021). https://doi.org/10.1007/s00280-021-04242-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-021-04242-0