Abstract
Purpose
To evaluate the diagnostic performance of histogram analysis of intravoxel incoherent motion (IVIM) parameters for differentiating prostate cancer (PCa) from benign prostatic hyperplasia (BPH), and compare with the monoexponential model, with in-bore MR-guided biopsy as pathological reference.
Methods
Thirty patients were included in this study. DWI images were processed with Matlab R2015b software by IVIM and monoexponential model for quantitation of diffusion coefficient (D), pseudo diffusion coefficient (D*), perfusion fraction (f), and apparent diffusion coefficient (ADC). The multiparametric data were compared between PCa and BPH group. Correlations between parameters and Gleason scores of PCa were assessed with Spearman rank test. ROC analysis was used to evaluate and compare the diagnostic ability of each parameter for discriminating PCa from BPH. Logistic regression model was used to evaluate the diagnostic performance of combination of different histogram parameters.
Results
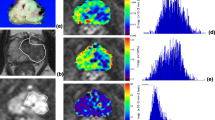
Sixteen PCa lesions and 20 BPH nodules were analyzed in this study. For IVIM-derived D, the histogram mean, 75th, 90th, and max of PCa were significantly lower than BPH. PCa had significantly lower min and 10th D* than BPH. For f, histogram mean, min, 10th, 25th, 50th, 75th, 90th, max and skew showed significant differences between PCa and BPH. For ADC, PCa were significantly lower than BPH in terms of histogram mean, min, 10th, 25th, 50th, 75th, 90th, max and kurtosis. Histogram mean D and min, 25th D* show significantly negative correlation with Gleason score (r = − 0.582, − 0.534, − 0.554, respectively). Histogram max D and mean f and min ADC showed higher diagnostic performance than other parameters (AUC = 0.925, 0.881, 0.969, respectively). The IVIM model (combined with max D, min D* and mean f) (AUC = 0.950 [0.821, 0.995]) didn’t show significant difference from the monoexponential model (AUC = 0.969 [0.849, 0.999], p = 0.23). Besides, combination of the IVIM and monoexponential model didn’t improve diagnostic performance compared with the single model (p = 0.362 and 0.763, respectively).
Conclusions
Histogram analyses of IVIM and monoexponential model were both useful methods for discriminating PCa from BPH. The diagnostic performance of IVIM model seemed to be not superior to that of monoexponential model. Combination of IVIM and monoexponential model did not add significant information to the single model alone.





Similar content being viewed by others
References
Torre LA, Siegel RL, Ward EM, Jemal A (2016) Global Cancer Incidence and Mortality Rates and Trends--An Update. Cancer Epidemiol Biomarkers Prev 25:16-27. https://doi.org/10.1158/1055-9965.epi-15-0578
Monn MF, Tatem AJ, Cheng L (2016) Prevalence and management of prostate cancer among East Asian men: Current trends and future perspectives. Urol Oncol 34:58.e1-9. https://doi.org/10.1016/j.urolonc.2015.09.003
Siddiqui MM, Rais-Bahrami S, Turkbey B, George AK, Rothwax J, Shakir N, Okoro C, Raskolnikov D, Parnes HL, Linehan WM, Merino MJ, Simon RM, Choyke PL, Wood BJ, Pinto PA (2015) Comparison of MR/ultrasound fusion-guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. JAMA 313:390-397. https://doi.org/10.1001/jama.2014.17942
Taira AV, Merrick GS, Galbreath RW, Andreini H, Taubenslag W, Curtis R, Butler WM, Adamovich E, Wallner KE (2010) Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis 13:71-77. https://doi.org/10.1038/pcan.2009.42
Hoeks CM, Hambrock T, Yakar D, de Kaa CA H, Feuth T, Witjes JA, Fütterer JJ, Barentsz JO (2013) Transition zone prostate cancer: detection and localization with 3-T multiparametric MR imaging. Radiology 266:207-217. https://doi.org/10.1148/radiol.12120281
Thai JN, Narayanan HA, George AK, Siddiqui MM, Shah P, Mertan FV, Merino MJ, Pinto PA, Choyke PL, Wood BJ, Turkbey B (2018) Validation of PI-RADS Version 2 in Transition Zone Lesions for the Detection of Prostate Cancer. Radiology 288:485-491. https://doi.org/10.1148/radiol.2018170425
Wu LM, Xu JR, Ye YQ, Lu Q, Hu JN (2012) The clinical value of diffusion-weighted imaging in combination with T2-weighted imaging in diagnosing prostate carcinoma: a systematic review and meta-analysis. AJR Am J Roentgenol 199:103-110. https://doi.org/10.2214/ajr.11.7634
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S (2016) PI-RADS Prostate Imaging - Reporting and Data System: 2015, Version 2. Eur Urol 69:16-40. https://doi.org/10.1016/j.eururo.2015.08.052
Oto A, Kayhan A, Jiang Y, Tretiakova M, Yang C, Antic T, Dahi F, Shalhav AL, Karczmar G, Stadler WM (2010) Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology 257:715-723. https://doi.org/10.1148/radiol.10100021
Lee H, Hwang SI, Lee HJ, Byun SS, Lee SE, Hong SK (2018) Diagnostic performance of diffusion-weighted imaging for prostate cancer: Peripheral zone versus transition zone. PLoS One 13:e0199636. https://doi.org/10.1371/journal.pone.0199636
Akin O, Sala E, Moskowitz CS, Kuroiwa K, Ishill NM, Pucar D, Scardino PT, Hricak H (2006) Transition zone prostate cancers: features, detection, localization, and staging at endorectal MR imaging. Radiology 239:784-792. https://doi.org/10.1148/radiol.2392050949
Le BD, Breton E, Lallemand D, Aubin ML, Vignaud J, Laval-Jeantet M (1988) Separation of diffusion and perfusion in intravoxel incoherent motion MR imaging. Radiology 168:497-505. https://doi.org/10.1148/radiology.168.2.3393671
Valerio M, Zini C, Fierro D, Giura F, Colarieti A, Giuliani A, Laghi A, Catalano C, Panebianco V (2016) 3T multiparametric MRI of the prostate: Does intravoxel incoherent motion diffusion imaging have a role in the detection and stratification of prostate cancer in the peripheral zone. Eur J Radiol 85:790-794. https://doi.org/10.1016/j.ejrad.2016.01.006
Shinmoto H, Tamura C, Soga S, Shiomi E, Yoshihara N, Kaji T, Mulkern RV (2012) An intravoxel incoherent motion diffusion-weighted imaging study of prostate cancer. AJR Am J Roentgenol 199:W496-500. https://doi.org/10.2214/ajr.11.8347
Bao J, Wang X, Hu C, Hou J, Dong F, Guo L (2017) Differentiation of prostate cancer lesions in the Transition Zone by diffusion-weighted MRI. Eur J Radiol Open 4:123-128. https://doi.org/10.1016/j.ejro.2017.08.00
Donati OF, Mazaheri Y, Afaq A, Vargas HA, Zheng J, Moskowitz CS, Hricak H, Akin O (2014) Prostate cancer aggressiveness: assessment with whole-lesion histogram analysis of the apparent diffusion coefficient. Radiology 271:143-152. https://doi.org/10.1148/radiol.13130973
Nagel KN, Schouten MG, Hambrock T, Litjens GJ, Hoeks CM, ten HB, Barentsz JO, Fütterer JJ (2013) Differentiation of prostatitis and prostate cancer by using diffusion-weighted MR imaging and MR-guided biopsy at 3 T. Radiology 267:164-172. https://doi.org/10.1148/radiol.12111683
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA (2016) The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am J Surg Pathol 40:244-252. https://doi.org/10.1097/pas.0000000000000530
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837-845.
Kobus T, Vos PC, Hambrock T, De Rooij M, de Kaa CA H, Barentsz JO, Heerschap A, Scheenen TW (2012) Prostate cancer aggressiveness: in vivo assessment of MR spectroscopy and diffusion-weighted imaging at 3 T. Radiology 265:457-467. https://doi.org/10.1148/radiol.12111744
Hambrock T, Somford DM, Huisman HJ, van Oort IM, Witjes JA, de Kaa CA H, Scheenen T, Barentsz JO (2011) Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259:453-461. https://doi.org/10.1148/radiol.11091409
Peng Y, Jiang Y, Yang C, Brown JB, Antic T, Sethi I, Schmid-Tannwald C, Giger ML, Eggener SE, Oto A (2013) Quantitative analysis of multiparametric prostate MR images: differentiation between prostate cancer and normal tissue and correlation with Gleason score--a computer-aided diagnosis development study. Radiology 267:787-796. https://doi.org/10.1148/radiol.13121454
Le BD, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M (1986) MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology 161:401-407. https://doi.org/10.1148/radiology.161.2.3763909
Le BD (2008) Intravoxel incoherent motion perfusion MR imaging: a wake-up call. Radiology 249:748-752. https://doi.org/10.1148/radiol.2493081301
Iima M, Le BD (2016) Clinical Intravoxel Incoherent Motion and Diffusion MR Imaging: Past, Present, and Future. Radiology 278:13-32. https://doi.org/10.1148/radiol.2015150244
Liu X, Zhou L, Peng W, Wang C, Wang H (2013) Differentiation of central gland prostate cancer from benign prostatic hyperplasia using monoexponential and biexponential diffusion-weighted imaging. Magn Reson Imaging 31:1318-1324. https://doi.org/10.1016/j.mri.2013.03.002
Enkhbaatar NE, Inoue S, Yamamuro H, Kawada S, Miyaoka M, Nakamura N, Sadahiro S, Imai Y (2018) MR Imaging with Apparent Diffusion Coefficient Histogram Analysis: Evaluation of Locally Advanced Rectal Cancer after Chemotherapy and Radiation Therapy. Radiology 288:129-137. https://doi.org/10.1148/radiol.2018171804
Peng Y, Jiang Y, Antic T, Giger ML, Eggener SE, Oto A (2014) Validation of quantitative analysis of multiparametric prostate MR images for prostate cancer detection and aggressiveness assessment: a cross-imager study. Radiology 271:461-471. https://doi.org/10.1148/radiol.14131320
Kyriazi S, Collins DJ, Messiou C, Pennert K, Davidson RL, Giles SL, Kaye SB, Desouza NM (2011) Metastatic ovarian and primary peritoneal cancer: assessing chemotherapy response with diffusion-weighted MR imaging--value of histogram analysis of apparent diffusion coefficients. Radiology 261:182-192. https://doi.org/10.1148/radiol.11110577
Zhang YD, Wang Q, Wu CJ, Wang XN, Zhang J, Liu H, Liu XS, Shi HB (2015) The histogram analysis of diffusion-weighted intravoxel incoherent motion (IVIM) imaging for differentiating the gleason grade of prostate cancer. Eur Radiol 25:994-1004. https://doi.org/10.1007/s00330-014-3511-4
Pang Y, Turkbey B, Bernardo M, Kruecker J, Kadoury S, Merino MJ, Wood BJ, Pinto PA, Choyke PL (2013) Intravoxel incoherent motion MR imaging for prostate cancer: an evaluation of perfusion fraction and diffusion coefficient derived from different b-value combinations. Magn Reson Med 69:553-562. https://doi.org/10.1002/mrm.24277
Lemke A, Stieltjes B, Schad LR, Laun FB (2011) Toward an optimal distribution of b values for intravoxel incoherent motion imaging. Magn Reson Imaging 29:766-776. https://doi.org/10.1016/j.mri.2011.03.004
Shinmoto H, Oshio K, Tanimoto A, Higuchi N, Okuda S, Kuribayashi S, Mulkern RV (2009) Biexponential apparent diffusion coefficients in prostate cancer. Magn Reson Imaging 27:355-359. https://doi.org/10.1016/j.mri.2008.07.008
Barbieri S, Brönnimann M, Boxler S, Vermathen P, Thoeny HC (2017) Differentiation of prostate cancer lesions with high and with low Gleason score by diffusion-weighted MRI. Eur Radiol 27:1547-1555. https://doi.org/10.1007/s00330-016-4449-5
Acknowledgements
This work was supported by grants from Dongcheng District Outstanding Talent Project and Beijing Hospital Nova Project (BJ-2016-037).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cui, Y., Li, C., Liu, Y. et al. Differentiation of prostate cancer and benign prostatic hyperplasia: comparisons of the histogram analysis of intravoxel incoherent motion and monoexponential model with in-bore MR-guided biopsy as pathological reference. Abdom Radiol 45, 3265–3277 (2020). https://doi.org/10.1007/s00261-019-02227-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-019-02227-5