Abstract
Purpose
The purpose of the study was to evaluate the feasibility of using contrast-enhanced computed tomography (CECT)-based texture analysis (CTTA) metrics to differentiate between juxtatumoral perinephric fat (JPF) surrounding low-grade (ISUP 1–2) versus high-grade (ISUP 3–4) clear cell renal cell carcinoma (ccRCC).
Methods
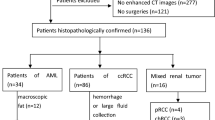
In this IRB-approved study, we retrospectively queried the surgical database between June 2009 and April 2016 and identified 83 patients with pathologically confirmed ccRCC (low grade: n = 54, mean age = 61.5 years, 18F/35M; high grade n = 30, mean age = 61.7 years, 8F/22M) who also had pre-operative multiphase CT acquisitions. CT images were transferred to a 3D workstation, and nephrographic phase JPF regions were manually segmented. Using an in-house developed Matlab program, a CTTA panel comprising of texture metrics extracted using six different methods, histogram, 2D- and 3D-Gray-level co-occurrence matrix (GLCM) and Gray-level difference matrix (GLDM), and 2D-Fast Fourier Transform (FFT) analyses, was applied to the segmented images to assess JPF textural heterogeneity in low- versus high-grade ccRCC. Univariate analysis and receiver-operator characteristics (ROC) analysis were used to assess interclass differences in texture metrics and their prediction accuracy, respectively.
Results
All methods except GLCM consistently revealed increased heterogeneity in the JPF surrounding high- versus low-grade ccRCC. FFT showed increased complexity index (p < 0.01). Histogram analysis showed increased kurtosis and positive skewness in (p < 0.03), and GLDM analysis showed decreased measure of correlation coefficient (MCC) (p < 0.04). Several of the GLCM metrics showed statistically significant (p < 0.04) textural differences between the two groups, but with no consistent trend. ROC analysis showed that MCC in GLCM analysis had an area under the curve of 0.75.
Conclusions
Our study suggests that CTTA of ccRCC shows statistically significant textural differences in JPF surrounding high- versus low-grade ccRCC.





Similar content being viewed by others
References
Siegel R, DeSantis C, Virgo K, Stein K, Mariotto A, Smith T, et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin. 2012;62(4):220–41.
Xing T, He H. Epigenomics of clear cell renal cell carcinoma: mechanisms and potential use in molecular pathology. Chin J Cancer Res. 2016;28(1):80–91.
Burrell RA, McGranahan N, Bartek J, Swanton C. The causes and consequences of genetic heterogeneity in cancer evolution. Nature. 2013;501(7467):338–45.
Shuch B, Amin A, Armstrong AJ, Eble JN, Ficarra V, Lopez-Beltran A, et al. Understanding pathologic variants of renal cell carcinoma: distilling therapeutic opportunities from biologic complexity. Eur Urol. 2015;67(1):85–97.
Casbas-Hernandez P, Sun X, Roman-Perez E, D'Arcy M, Sandhu R, Hishida A, et al. Tumor intrinsic subtype is reflected in cancer-adjacent tissue. Cancer Epidemiol Biomarkers Prev. 2015;24(2):406–14.
Panis C, Victorino VJ, Herrera AC, Cecchini AL, Simao AN, Tomita LY, et al. Can Breast Tumors Affect the Oxidative Status of the Surrounding Environment? A Comparative Analysis among Cancerous Breast, Mammary Adjacent Tissue, and Plasma. Oxid Med Cell Longev. 2015;2015:6429812.
Macleod LC, Hsi RS, Gore JL, Wright JL, Harper JD. Perinephric fat thickness is an independent predictor of operative complexity during robot-assisted partial nephrectomy. J Endourol. 2014;28(5):587–91.
Anderson KM, Lindler TU, Lamberton GR, Baron PW, Ojogho OK, Baldwin DD. Laparoscopic donor nephrectomy: effect of perirenal fat upon donor operative time. J Endourol. 2008;22(10):2269–74.
Kocher NJ, Kunchala S, Reynolds C, Lehman E, Nie S, Raman JD. Adherent perinephric fat at minimally invasive partial nephrectomy is associated with adverse peri-operative outcomes and malignant renal histology. BJU Int. 2016;117(4):636–41.
Varghese BA HD, Cen SY, Desai BB, Yap, FY, Gill I, Desai M, et al. . Fast fourier transform based analysis of renal masses on contrast-enhanced computed tomography images for grading of tumor. International Society for Optics and Photonics2017. p. 101600J-J-8.
Yu H, Scalera J, Khalid M, Touret AS, Bloch N, Li B, et al. Texture analysis as a radiomic marker for differentiating renal tumors. Abdom Radiol (NY). 2017.
Just N. Improving tumour heterogeneity MRI assessment with histograms. Br J Cancer. 2014;111(12):2205–13.
Yoo C, Song C, Hong JH, Kim CS, Ahn H. Prognostic significance of perinephric fat infiltration and tumor size in renal cell carcinoma. J Urol. 2008;180(2):486–91; discussion 91.
Skogen K, Ganeshan B, Good C, Critchley G, Miles K. Measurements of heterogeneity in gliomas on computed tomography relationship to tumour grade. J Neurooncol. 2013;111(2):213–9.
Yang L, Dong D, Fang M, Zhu Y, Zang Y, Liu Z, et al. Can CT-based radiomics signature predict KRAS/NRAS/BRAF mutations in colorectal cancer? Eur Radiol. 2018;28(5):2058–67.
Chen C, Kang Q, Xu B, Guo H, Wei Q, Wang T, et al. Differentiation of low- and high-grade clear cell renal cell carcinoma: Tumor size versus CT perfusion parameters. Clin Imaging. 2017;46:14–9.
Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141(1):39–51.
Mackin D, Fave X, Zhang L, Fried D, Yang J, Taylor B, et al. Measuring Computed Tomography Scanner Variability of Radiomics Features. Invest Radiol. 2015;50(11):757–65.
Duda RO, Hart PE, Stork DG. Pattern Classification (2nd Edition): Wiley-Interscience; 2000.
Haury AC, Gestraud P, Vert JP. The influence of feature selection methods on accuracy, stability and interpretability of molecular signatures. PLoS One. 2011;6(12):e28210.
Acknowledgements
The authors acknowledge FUJIFILM Medical Systems USA, Inc. for the use of their Synapse® 3D software for completing this work.
Funding
The project did not receive any funding.
Author information
Authors and Affiliations
Contributions
TG is the first author and drafted the manuscript. She manually segmented the JPF in Synapse 3D. BV performed data processing and reviewed manuscript. DH aided in CT analysis. SC performed the statistical analysis. MA performed pathological correlation. MA provided urological clinical significance. VD supervised the study, as well as reviewed and edited manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Vinay Duddalwar serves as a consultant to Intuitive Surgical and as an advisor to DeepTek. Dr. Monish Aron serves as a consultant to Intuitive Surgical. The other authors have no conflict of interest with respect to the work presented in this manuscript.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Gill, T.S., Varghese, B.A., Hwang, D.H. et al. Juxtatumoral perinephric fat analysis in clear cell renal cell carcinoma. Abdom Radiol 44, 1470–1480 (2019). https://doi.org/10.1007/s00261-018-1848-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1848-x