Abstract
Radiomics and radiogenomics are attractive research topics in prostate cancer. Radiomics mainly focuses on extraction of quantitative information from medical imaging, whereas radiogenomics aims to correlate these imaging features to genomic data. The purpose of this review is to provide a brief overview summarizing recent progress in the application of radiomics-based approaches in prostate cancer and to discuss the potential role of radiogenomics in prostate cancer.


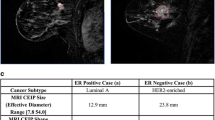
Reprinted from Stoyanova R et al. PMID: 27438142 with permission of Oncotarget
Similar content being viewed by others
References
Lambin P, Rios-Velazquez E, Leijenaar R, et al. (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer. 48(4):441–446
Kumar V, Gu Y, Basu S, et al. (2012) Radiomics: the process and the challenges. Magn Reson Imaging. 30(9):1234–1248
Thompson I, Thrasher JB, Aus G, et al. (2007) Guideline for the management of clinically localized prostate cancer: 2007 update. J Urol. 177(6):2106–2131
Fernandes ET, Sundaram CP, Long R, Soltani M, Ercole CJ (1997) Biopsy Gleason score: how does it correlate with the final pathological diagnosis in prostate cancer? Br J Urol. 79(4):615–617
Kvåle R, Møller B, Wahlqvist R, et al. (2009) Concordance between Gleason scores of needle biopsies and radical prostatectomy specimens: a population-based study. BJU Int. 103(12):1647–1654
Siddiqui MM, Rais-Bahrami S, Turkbey B, et al. (2015) Comparison of MR/ultrasound fusion-guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. JAMA. 313(4):390–397
Aihara M, Wheeler TM, Ohori M, Scardino PT (1994) Heterogeneity of prostate cancer in radical prostatectomy specimens. Urology. 43(1):60–66 ((discussion 6-7))
El-Shater Bosaily A, Valerio M, Hu Y, et al. (2016) The concordance between the volume hotspot and the grade hotspot: a 3-D reconstructive model using the pathology outputs from the PROMIS trial. Prostate Cancer Prostatic Dis. 19(3):258–263
Boutros PC, Fraser M, Harding NJ, et al. (2015) Spatial genomic heterogeneity within localized, multifocal prostate cancer. Nat Genet. 47(7):736–745
Weinreb JC, Barentsz JO, Choyke PL, et al. (2016) PI-RADS prostate imaging–reporting and data system: 2015, version 2. Eur Urol. 69(1):16–40
Greer MD, Brown AM, Shih JH, et al. (2017) Accuracy and agreement of PI-RADS v2 for prostate cancer mpMRI: a multireader study. J Magn Reson Imaging. 45(2):579–585
Langer DL, van der Kwast TH, Evans AJ, et al. (2010) Prostate tissue composition and MR measurements: investigating the relationships between ADC, T2, K(trans), v(e), and corresponding histologic features. Radiology. 255(2):485–494
Zelhof B, Pickles M, Liney G, et al. (2009) Correlation of diffusion-weighted magnetic resonance data with cellularity in prostate cancer. BJU Int. 103(7):883–888
Gibbs P, Liney GP, Pickles MD, et al. (2009) Correlation of ADC and T2 measurements with cell density in prostate cancer at 3.0 Tesla. Invest Radiol. 44(9):572–576
Haralick RM, Shanmugam K, Dinstein I (1973) Textural features for image classification. IEEE Trans Syst Man Cybernet. 6:610–621
Srinivasan G, Shobha G, editors. Statistical texture analysis. Proceedings of world academy of science, engineering and technology; 2008.
Bovik AC, Clark M, Geisler WS (1990) Multichannel texture analysis using localized spatial filters. IEEE Trans Pattern Anal Mach Intell. 12(1):55–73
Busch C (1997) Wavelet based texture segmentation of multi-modal tomographic images. Comput Gr. 21(3):347–358
Tang X (1998) Texture information in run-length matrices. IEEE Trans Image Process. 7(11):1602–1609
Sun C, Wee W (1983) Neighboring gray level dependence matrix for texture classification. Comput Vis Gr Image Process 23(3):341–352
Amadasun M, King R (1989) Texural features corresponding to textural properties. IEEE Trans Syst Man Cybern 23(3):1264–1274
Orlhac F, Soussan M, Maisonobe JA, et al. (2014) Tumor texture analysis in 18F-FDG PET: relationships between texture parameters, histogram indices, standardized uptake values, metabolic volumes, and total lesion glycolysis. J Nucl Med. 55(3):414–422
Chalkidou A, O’Doherty MJ, Marsden PK (2015) False discovery rates in PET and CT studies with texture features: a systematic review. PLoS One. 10(5):e0124165
Trabulsi EJ, Liu XS, Smith WR, Das AK (2013) Transrectal ultrasound of the prostate. In: Gilbert BR, Fulgham PF (eds) Practical urological ultrasound. New York: Springer, pp 155–170
Shen D, Lao Z, Zeng J, et al. (2004) Optimized prostate biopsy via a statistical atlas of cancer spatial distribution. Med Image Anal. 8(2):139–150
Zhan Y, Shen D, Zeng J, et al. (2007) Targeted prostate biopsy using statistical image analysis. IEEE Trans Med Imaging. 26(6):779–788
Xu S, Kruecker J, Turkbey B, et al. (2008) Real-time MRI-TRUS fusion for guidance of targeted prostate biopsies. Comput Aided Surg. 13(5):255–264
Wu P, Liu Y, Li Y, Liu B (2015) Robust prostate segmentation using intrinsic properties of TRUS images. IEEE Trans Med Imaging. 34(6):1321–1335
Gong L, Pathak SD, Haynor DR, Cho PS, Kim Y (2004) Parametric shape modeling using deformable superellipses for prostate segmentation. IEEE Trans Med Imaging. 23(3):340–349
K. Diaz, B. Castaneda (eds) Semi-automated segmentation of the prostate gland boundary in ultrasound images using a machine learning approach. Medical Imaging (SPIE, 2008).
H. Ning, D. B. Downey, A. Fenster, H. M. Ladak (eds) Prostate surface segmentation from 3D ultrasound images. Proceedings IEEE International Symposium on Biomedical Imaging (2002).
Cosío FA (2008) Automatic initialization of an active shape model of the prostate. Med Image Anal. 12(4):469–483
Ghose S, Oliver A, Mitra J, et al. (2013) A supervised learning framework of statistical shape and probability priors for automatic prostate segmentation in ultrasound images. Med Image Anal. 17(6):587–600
A. Zaim, T. Yi, R. Keck (eds) Feature-Based Classification of Prostate Ultrasound Images using Multiwavelet and Kernel Support Vector Machines. 2007 International Joint Conference on Neural Networks (12-17 Aug 2007).
Yan P, Xu S, Turkbey B, Kruecker J (2010) Discrete deformable model guided by partial active shape model for TRUS image segmentation. IEEE Trans Biomed Eng. 57(5):1158–1166
Murphy G, Haider M, Ghai S, Sreeharsha B (2013) The expanding role of MRI in prostate cancer. AJR Am J Roentgenol. 201(6):1229–1238
Toth R, Bloch BN, Genega EM, et al. (2011) Accurate prostate volume estimation using multifeature active shape models on T2-weighted MRI. Acad Radiol. 18(6):745–754
Chowdhury N, Toth R, Chappelow J, et al. (2012) Concurrent segmentation of the prostate on MRI and CT via linked statistical shape models for radiotherapy planning. Med Phys. 39(4):2214–2228
Shiradkar R, Podder TK, Algohary A, et al. (2016) Radiomics based targeted radiotherapy planning (Rad-TRaP): a computational framework for prostate cancer treatment planning with MRI. Radiat Oncol. 11(1):148
Pasquier D, Lacornerie T, Vermandel M, et al. (2007) Automatic segmentation of pelvic structures from magnetic resonance images for prostate cancer radiotherapy. Int J Radiat Oncol Biol Phys. 68(2):592–600
Costa MJ, Delingette H, Novellas S, Ayache N (2007) Automatic segmentation of bladder and prostate using coupled 3D deformable models. Med Image Comput Comput Assist Interv. 10(Pt 1):252–260
Klein S, van der Heide UA, Lips IM, et al. (2008) Automatic segmentation of the prostate in 3D MR images by atlas matching using localized mutual information. Med Phys. 35(4):1407–1417
Makni N, Puech P, Lopes R, et al. (2009) Combining a deformable model and a probabilistic framework for an automatic 3D segmentation of prostate on MRI. Int J Comput Assist Radiol Surg. 4(2):181–188
Chandra SS, Dowling JA, Shen KK, et al. (2012) Patient specific prostate segmentation in 3-d magnetic resonance images. IEEE Trans Med Imaging. 31(10):1955–1964
Litjens G, Toth R, van de Ven W, et al. (2014) Evaluation of prostate segmentation algorithms for MRI: the PROMISE12 challenge. Med Image Anal. 18(2):359–373
Makni N, Iancu A, Colot O, et al. (2011) Zonal segmentation of prostate using multispectral magnetic resonance images. Med Phys. 38(11):6093–6105
Litjens G, Debats O, van de Ven W, Karssemeijer N, Huisman H (2012) A pattern recognition approach to zonal segmentation of the prostate on MRI. Lect Notes Comput Sc. 7511:413–420
Maan B, van der Heijden F, Futterer JJ (2012) A new prostate segmentation approach using multispectral Magnetic Resonance Imaging and a statistical pattern classifier. Proc Spie. 8314:83142Q
Chilali O, Puech P, Lakroum S, et al. (2016) Gland and zonal segmentation of prostate on T2W MR images. J Digit Imaging. 29(6):730–736
Padgett K, Swallen A, Nelson A, Pollack A, Stoyanova R (2016) Robust atlas based segmentation of the prostate and peripheral zone regions on MRI utilizing multiple MRI system vendors. Med Phys. 43(6):3447
Cameron A, Khalvati F, Haider MA, Wong A (2016) MAPS: a quantitative radiomics approach for prostate cancer detection. IEEE Trans Biomed Eng. 63(6):1145–1156
Ginsburg SB, Algohary A, Pahwa S, et al. (2017) Radiomic features for prostate cancer detection on MRI differ between the transition and peripheral zones: preliminary findings from a multi-institutional study. J Magn Reson Imaging. 46(1):184–193
Han SM, Lee HJ, Choi JY (2008) Computer-aided prostate cancer detection using texture features and clinical features in ultrasound image. J Digit Imaging. 21(Suppl 1):S121–S133
Hussain L, Ahmed A, Saeed S, et al. (2018) Prostate cancer detection using machine learning techniques by employing combination of features extracting strategies. Cancer Biomark. 21(2):393–413
Khalvati F, Wong A, Haider MA (2015) Automated prostate cancer detection via comprehensive multi-parametric magnetic resonance imaging texture feature models. BMC Med Imaging. 15:27
Kwak JT, Xu S, Wood BJ, et al. (2015) Automated prostate cancer detection using T2-weighted and high-b-value diffusion-weighted magnetic resonance imaging. Med Phys. 42(5):2368–2378
Lay N, Tsehay Y, Greer MD, et al. (2017) Detection of prostate cancer in multiparametric MRI using random forest with instance weighting. J Med Imaging (Bellingham). 4(2):024506
Lopes R, Ayache A, Makni N, et al. (2011) Prostate cancer characterization on MR images using fractal features. Med Phys. 38(1):83–95
Madabhushi A, Feldman MD, Metaxas DN, Tomaszeweski J, Chute D (2005) Automated detection of prostatic adenocarcinoma from high-resolution ex vivo MRI. IEEE Trans Med Imaging. 24(12):1611–1625
Metzger GJ, Kalavagunta C, Spilseth B, et al. (2016) Detection of prostate cancer: quantitative multiparametric MR imaging models developed using registered correlative histopathology. Radiology. 279(3):805–816
Mohamed SS, Li J, Salama MM, Freeman G (2009) Prostate tissue texture feature extraction for suspicious regions identification on TRUS images. J Digit Imaging. 22(5):503–518
Stember JN, Deng FM, Taneja SS, Rosenkrantz AB (2014) Pilot study of a novel tool for input-free automated identification of transition zone prostate tumors using T2- and diffusion-weighted signal and textural features. J Magn Reson Imaging. 40(2):301–305
Castellano G, Bonilha L, Li LM, Cendes F (2004) Texture analysis of medical images. Clin Radiol. 59(12):1061–1069
Lv D, Guo X, Wang X, Zhang J, Fang J (2009) Computerized characterization of prostate cancer by fractal analysis in MR images. J Magn Reson Imaging. 30(1):161–168
Wang J, Wu CJ, Bao ML, et al. (2017) Machine learning-based analysis of MR radiomics can help to improve the diagnostic performance of PI-RADS v2 in clinically relevant prostate cancer. Eur Radiol. 27(10):4082–4090
Smith AD, Shah SN, Rini BI, Lieber ML, Remer EM (2010) Morphology, Attenuation, Size, and Structure (MASS) criteria: assessing response and predicting clinical outcome in metastatic renal cell carcinoma on antiangiogenic targeted therapy. AJR Am J Roentgenol. 194(6):1470–1478
Fehr D, Veeraraghavan H, Wibmer A, et al. (2015) Automatic classification of prostate cancer Gleason scores from multiparametric magnetic resonance images. Proc Natl Acad Sci USA 112(46):E6265–E6273
Nketiah G, Elschot M, Kim E, et al. (2017) T2-weighted MRI-derived textural features reflect prostate cancer aggressiveness: preliminary results. Eur Radiol. 27(7):3050–3059
Muller BG, Shih JH, Sankineni S, et al. (2015) Prostate Cancer: Interobserver Agreement and Accuracy with the Revised Prostate Imaging Reporting and Data System at Multiparametric MR Imaging. Radiology. 277(3):741–750
Mazurowski MA (2015) Radiogenomics: what it is and why it is important. J Am Coll Radiol. 12(8):862–866
Incoronato M, Aiello M, Infante T, et al. (2017) Radiogenomic analysis of oncological data: a technical survey. Int J Mol Sci. 18(4):805
Rutman AM, Kuo MD (2009) Radiogenomics: creating a link between molecular diagnostics and diagnostic imaging. Eur J Radiol. 70(2):232–241
Stoyanova R, Takhar M, Tschudi Y, et al. (2016) Prostate cancer radiomics and the promise of radiogenomics. Transl Cancer Res. 5(4):432–447
Thawani R, McLane M, Beig N, et al. (2018) Radiomics and radiogenomics in lung cancer: A review for the clinician. Lung Cancer. 115:34–41
Wu J, Tha KK, Xing L, Li R (2018) Radiomics and radiogenomics for precision radiotherapy. J Radiat Res. 59(Suppl 1):i25–i31
Pinker K, Shitano F, Sala E, et al. (2017) Background, current role, and potential applications of radiogenomics. J Magn Reson Imaging. 47(3):604–620
Lambin P, Leijenaar RTH, Deist TM, et al. (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol. 14(12):749–762
Valdora F, Houssami N, Rossi F, Calabrese M, Tagliafico AS (2018) Rapid review: radiomics and breast cancer. Breast Cancer Res Treat. 169(2):217–229
Zhou M, Scott J, Chaudhury B, et al. (2018) Radiomics in brain tumor: image assessment, quantitative feature descriptors, and machine-learning approaches. AJNR Am J Neuroradiol. 39(2):208–216
Litjens GJ, Elliott R, Shih NN, et al. (2016) Computer-extracted features can distinguish noncancerous confounding disease from prostatic adenocarcinoma at multiparametric MR imaging. Radiology. 278(1):135–145
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology. 278(2):563–577
Vignati A, Mazzetti S, Giannini V, et al. (2015) Texture features on T2-weighted magnetic resonance imaging: new potential biomarkers for prostate cancer aggressiveness. Phys Med Biol. 60(7):2685–2701
Wibmer A, Hricak H, Gondo T, et al. (2015) Haralick texture analysis of prostate MRI: utility for differentiating non-cancerous prostate from prostate cancer and differentiating prostate cancers with different Gleason scores. Eur Radiol. 25(10):2840–2850
Ahmed HU, El-Shater Bosaily A, Brown LC, et al. (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 389(10071):767–768
Arsov C, Rabenalt R, Blondin D, et al. (2015) Prospective randomized trial comparing magnetic resonance imaging (MRI)-guided In-bore Biopsy to MRI-ultrasound fusion and transrectal ultrasound-guided prostate biopsy in patients with prior negative biopsies. Eur Urol. 68(4):713–720
Bergdahl AG, Wilderang U, Aus G, et al. (2016) Role of magnetic resonance imaging in prostate cancer screening: a pilot study within the goteborg randomised screening trial. Eur Urol. 70(4):566–573
Porpiglia F, Manfredi M, Mele F, et al. (2016) Diagnostic pathway with multiparametric magnetic resonance imaging versus standard pathway: results from a randomized prospective study in biopsy-naive patients with suspected prostate cancer. Eur Urol. 72(2):282–288
Tonttila PP, Lantto J, Paakko E, et al. (2016) Prebiopsy multiparametric magnetic resonance imaging for prostate cancer diagnosis in biopsy-naive men with suspected prostate cancer based on elevated prostate-specific antigen values: results from a randomized prospective blinded controlled trial. Eur Urol. 69(3):419–425
Schena M, Shalon D, Davis RW, Brown PO (1995) Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science. 270(5235):467–470
Marzancola MG, Sedighi A, Li PC (2016) DNA microarray-based diagnostics. Methods Mol Biol. 1368:161–178
Geybels MS, Wright JL, Bibikova M, et al. (2016) Epigenetic signature of Gleason score and prostate cancer recurrence after radical prostatectomy. Clin Epigenet. 8:97
Poustka A, Pohl T, Barlow DP, et al. (1986) Molecular approaches to mammalian genetics. Cold Spring Harb Symp Quant Biol. 51(Pt 1):131–139
Wei L, Wang J, Lampert E, et al. (2016) Intratumoral and intertumoral genomic heterogeneity of multifocal localized prostate cancer impacts molecular classifications and genomic prognosticators. Eur Urol. 71(2):183–192
Wyatt AW, Azad AA, Volik SV, et al. (2016) Genomic alterations in cell-free dna and enzalutamide resistance in castration-resistant prostate cancer. JAMA Oncol. 2(12):1598–1606
el Bahassi M, Stambrook PJ (2014) Next-generation sequencing technologies: breaking the sound barrier of human genetics. Mutagenesis. 29(5):303–310
McCann SM, Jiang Y, Fan X, et al. (2016) Quantitative multiparametric MRI features and PTEN expression of peripheral zone prostate cancer: a pilot study. AJR Am J Roentgenol. 206(3):559–565
Stoyanova R, Pollack A, Takhar M, et al. (2016) Association of multiparametric MRI quantitative imaging features with prostate cancer gene expression in MRI-targeted prostate biopsies. Oncotarget. 7(33):53362–53376
Aerts HJ (2016) The potential of radiomic-based phenotyping in precision medicine: a review. JAMA Oncol. 2(12):1636–1642
The Cancer Genome Atlas (TCGA). Available from: https://cancergenome.nih.gov/. Accessed 12 March 2018
The Cancer Imaging Archive (TCIA). Available from: http://www.cancerimagingarchive.net/. Accessed 12 March 2018
Acknowledgements
This project has been funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. HHSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government. This research was also made possible through the NIH Medical Research Scholars Program, a public-private partnership supported jointly by the NIH and generous contributions to the Foundation for the NIH from the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This project has been funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health, under Contract No. HHSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government. This research was also made possible through the NIH Medical Research Scholars Program, a public-private partnership supported jointly by the NIH and generous contributions to the Foundation for the NIH from the Doris Duke Charitable Foundation, the American Association for Dental Research, the Colgate-Palmolive Company, Genentech, Elsevier, and other private donors.
Conflict of interest
None of the authors (CPS, MC, SM, SH, RS, PLC, BT) of this manuscript have any conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Not applicable.
Additional information
Peter L. Choyke, Stephanie Harmon and Baris Turkbey share the senior authorship.
Rights and permissions
About this article
Cite this article
Smith, C.P., Czarniecki, M., Mehralivand, S. et al. Radiomics and radiogenomics of prostate cancer. Abdom Radiol 44, 2021–2029 (2019). https://doi.org/10.1007/s00261-018-1660-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1660-7