Abstract
Purpose
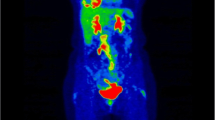
Quantitative analyses on FDG PET for response assessment are increasingly used in clinical studies, particularly with respect to tumours in which radiological assessment is challenging and complete metabolic response is rarely achieved after treatment. A typical example is malignant pleural mesothelioma (MPM), an aggressive tumour originating from mesothelial cells of the pleura. We present our results concerning the use of semiquantitative and quantitative parameters, evaluated at the baseline and interim PET examinations, for the prediction of treatment response and disease outcome in patients with MPM.
Methods
We retrospectively analysed data derived from 131 patients (88 men, 43 women; mean age 66 years) with MPM who were referred to our institution for treatment between May 2004 and July 2013. Patients were investigated using FDG PET at baseline and after two cycles of pemetrexed-based chemotherapy. Responses were determined using modified RECIST criteria based on the best CT response after treatment. Disease control rate, progression-free survival (PFS) and overall survival (OS) were calculated for the whole population and were correlated with semiquantitative and quantitative parameters evaluated at the baseline and interim PET examinations; these included SUVmax, total lesion glycolysis (TLG), percentage change in SUVmax (ΔSUVmax) and percentage change in TLG (ΔTLG).
Results
Disease control was achieved in 84.7 % of the patients, and median PFS and OS for the entire cohort were 7.2 and 14.3 months, respectively. The log-rank test showed a statistically significant difference in PFS between patients with radiological progression and those with partial response (PR) or stable disease (SD) (1.8 vs. 8.6 months, p < 0.001). Baseline SUVmax and TLG showed a statistically significant correlation with PFS and OS (p < 0.001). In the entire population, both ΔSUVmax and ΔTLG were correlated with disease control based on best CT response (p < 0.001). ΔSUVmax was significantly correlated with PFS in the entire population (p = 0.02) and with both PFS and OS in patients not undergoing talc pleurodesis (n = 65; p < 0.01 for PFS, p = 0.03 for OS), and in patients without pleurodesis presenting a SD and/or PR at CT after two cycles.
Conclusion
These results confirm the role of FDG PET in the assessment of disease prognosis and treatment efficacy in MPM patients receiving first-line pemetrexed-based chemotherapy. In particular, metabolic response evaluated using ΔSUVmax can be used to predict outcome in MPM patients not undergoing talc pleurodesis who achieve SD and/or PR at the interim CT evaluation.




Similar content being viewed by others
References
Robinson BWS, Musk AW, Lake AR. Malignant mesothelioma. Lancet. 2005;366:397–408.
Sugarbaker DJ, Flores RM, Jaklitsch MT, Richards WG, Strauss GM, Corson JM, et al. Resection margins, extrapleural nodal status, and cell type determine postoperative long-term survival in trimodality therapy of malignant pleural mesothelioma: results in 183 patients. J Thorac Cardiovasc Surg. 1999;117:54–65.
Byrne MJ, Nowak AK. Modified RECIST criteria for assessment of response in malignant pleural mesothelioma. Ann Oncol. 2004;15:257–60.
Armato SG, Labby ZE, Coolen J, Klabatsa A, Feigen M, Persigehl T, et al. Imaging in pleural mesothelioma: a review of the 11th International Conference of the International Mesothelioma Interest Group. Lung Cancer. 2013;82:190–6.
Armato III SG, Ogarek JL, Starkey A, Vogelzang NJ, Kindler HL, Kocherginsky M, et al. Variability in mesothelioma tumor response classification. AJR Am J Roentgenol. 2006;186:1000–6.
Oxnard GR, Armato III SG, Kindler HL. Modeling of mesothelioma growth demonstrates weakness of current response criteria. Lung Cancer. 2006;52:141–8.
Ceresoli GL, Chiti A, Zucali PA, Cappuzzo F, De Vincenzo F, Cavina R, et al. Assessment of tumor response in malignant pleural mesothelioma. Cancer Treat Rev. 2007;33:533–41.
Larson SM, Erdi Y, Akhurst T, Mazumdar M, Macapinlac HA, Finn RD, et al. Tumor treatment response based on visual and quantitative changes in global tumor glycolysis using PET-FDG imaging. The visual response score and the change in total lesion glycolysis. Clin Positron Imaging. 1999;2:159–71.
Ceresoli GL, Chiti A, Zucali PA, Rodari M, Lutman RF, Salamina S, et al. Early evaluation in malignant pleural mesothelioma by positron emission tomography with [18F]fluorodeoxyglucose. J Clin Oncol. 2006;24:4587–93.
Francis RJ, Byrne MJ, van der Schaaf AA, Boucek JA, Nowak AK, Phillips M, et al. Early prediction of response to chemotherapy and survival in malignant pleural mesothelioma using a novel semiautomated 3-dimensional volume-based analysis of serial 18F-FDG PET scans. J Nucl Med. 2007;48:1449–58.
Veit-Haibach P, Schaefer NG, Steinert HC, Soyka JD, Seifert B, Stahel RA. Combined FDG-PET/CT in response evaluation of malignant pleural mesothelioma. Lung Cancer. 2010;67:311–7.
Lee HY, Hyun SH, Lee KS, Kim BT, Kim J, Shim YM, et al. Volume-based parameter of 18F-FDG PET/CT in malignant pleural mesothelioma: prediction of therapeutic response and prognostic implications. Ann Surg Oncol. 2010;17:2787–94.
Genestreti G, Moretti A, Piciucchi S, Giovannini N, Galassi R, Scarpi E, et al. FDG PET/CT response evaluation in malignant pleural mesothelioma patients treated with talc pleurodesis and chemotherapy. J Cancer. 2012;3:241–5.
Schaefer NG, Veit-Haibach P, Soyka JD, Steinert HC, Stahel RA. Continued pemetrexed and platin-based chemotherapy in patients with malignant pleural mesothelioma (MPM): value of 18F-FDGPET/CT. Eur J Radiol. 2012;81:e19–25.
Klabatsa A, Chicklore S, Barrington S, Goh V, Lang-Lazdunski L, Cook GJ. The association of 18F-FDG PET/CT parameters with survival in malignant pleural mesothelioma. Eur J Nucl Med Mol Imaging. 2014;41:276–82.
Boucek J, Francis RJ, Green AJ. Automated approach to identification and quantitation of tumour volumes in chemotherapy monitoring using FDG PET [abstract]. J Nucl Med. 2005;46 Suppl:464P.
Lopci E, Zucali P, Giordano L, Perrino M, Ceresoli G, Gemelli M, et al. Validation of liver-based quantitative analysis on PET for response assessment in patients with malignant pleural mesothelioma. J Nucl Med. 2014;55 Suppl 1:458.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81.
Lang-Lazdunski L, Bille A, Lal R, Cane P, McLean E, Landau D, et al. Pleurectomy/decortication is superior to extrapleural pneumonectomy in the multimodality management of patients with malignant pleural mesothelioma. J Thorac Oncol. 2012;7:737–43.
Ellis P, Davies AM, Evans WK, Haynes AE, Lloyd NS. The use of chemotherapy in patients with advanced malignant pleural mesothelioma: a systematic review and practice guideline. J Thorac Oncol. 2006;1:591–601.
Ceresoli GL, Castagneto B, Zucali PA, Favaretto A, Mencoboni M, Grossi F, et al. Pemetrexed plus carboplatin in elderly patients with malignant pleural mesothelioma: combined analysis of two phase II trials. Br J Cancer. 2008;99(1):51–6.
Ahamad A, Stevens C, Smythe W, Liao Z, Vaporciyan AA, Rice D, et al. Promising early local control of malignant pleural mesothelioma following postoperative intensity modulated radiotherapy (IMRT) to the chest. Cancer J. 2003;9:476–84.
Sterman DH, Haas A, Moon E, Recio A, Schwed D, Vachani A, et al. A trial of intrapleural adenoviral-mediated interferon-alpha2b gene transfer for malignant pleural mesothelioma. Am J Respir Crit Care Med. 2011;184:1395–9.
Lucchi M, Chella A, Melfi F, Dini P, Tibaldi C, Fontanini G, et al. Four-modality therapy in malignant pleural mesothelioma: a phase II study. J Thorac Oncol. 2007;2:237–42.
Kruse M, Sherry SJ, Paidpally V, Mercier G, Subramaniam RM. FDG PET/CT in management of primary pleural tumors and pleural metastases. AJR Am J Roentgenol. 2013;201:W215–26.
Vogelzang NJ, Rusthoven JJ, Symanowski J, Denham C, Kaukel E, Ruffie P, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol. 2003;21:2636–44.
Nowak AK, Lake RA, Kindler HL, Robinson BW. New approaches for mesothelioma: biologics, vaccines, gene therapy, and other novel agents. Semin Oncol. 2002;29:82–96.
Terada T, Tabata C, Tabata R, Okuwa H, Kanemura S, Shibata E, et al. Clinical utility of 18-fluorodeoxyglucose positron emission tomography/computed tomography in malignant pleural mesothelioma. Exp Ther Med. 2012;4:197–200.
Cao C, Tian DH, Pataky KA, Yan TD. Systematic review of pleurectomy in the treatment of malignant pleural mesothelioma. Lung Cancer. 2013;81:319–27.
Herndon JE, Green MR, Chahinian AP, Corson JM, Suzuki Y, Vogelzang NJ. Factors predictive of survival among 337 patients with mesothelioma treated between 1984 and 1994 by the Cancer and Leukemia Group B. Chest. 1998;113:723–31.
Edwards JG, Abrams KR, Leverment JN, Spyt TJ, Waller DA, O’Byrne KJ. Prognostic factors for malignant mesothelioma in 142 patients: validation of CALGB and EORTC prognostic scoring systems. Thorax. 2000;55:731–5.
Nowak AK, Francis RJ, Phillips MJ, Millward MJ, van der Schaaf AA, Boucek J, et al. A novel prognostic model for malignant mesothelioma incorporating quantitative FDG-PET imaging with clinical parameters. Clin Cancer Res. 2010;16:2409–17.
Abakay A, Komek H, Abakay O, Palanci Y, Ekici F, Tekbas G, et al. Relationship between 18FDG PET-CT findings and survival of 177 patients with malignant pleural mesothelioma. Eur Rev Med Pharmacol Sci. 2013;17:1233–41.
Steinert HC, Santos Dellea MM, Burger C, Stahel R. Therapy response evaluation in malignant pleural mesothelioma with integrated PET-CT imaging. Lung Cancer. 2005;49 Suppl 1:S33–5.
Kwek BH, Auino SL, Fischman AJ. Fluorodeoxyglucose positron emission tomography after talc pleurodesis. Chest. 2004;125:2356–60.
Ahmadzadehfar H, Palmedo H, Strunk H, Biersack HJ, Habibi E, Ezziddin S. False positive 18F-FDG-PET/CT in a patient after talc pleurodesis. Lung Cancer. 2007;58:418–21.
Novak AK, Armato SG, Ceresoli GL, Yildirim H, Francis RJ. Imaging in pleural mesothelioma: a review of imaging research presented at the 9th International Meeting of the International Mesothelioma Interest Group. Lung Cancer. 2010;70:1–6.
Weber WA, Wieder H. Monitoring chemotherapy and radiotherapy of solid tumors. Eur J Nucl Med Mol Imaging. 2006;33 Suppl 13:S27–37.
Strorto G, Nicolai E, Salvatore M. [18F]FDG-PET/CT for early monitoring of tumor response: when and why. Q J Nucl Med Mol Imaging. 2009;53:167–80.
Young H, Baum R, Cremerius U, Herholz K, Hoekstra O, Lammertsma AA, et al. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. Eur J Cancer. 1999;35:1773–82.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50 Suppl 1:122S–50.
Curran D, Sahmoud T, Therasse P, van Meerbeeck J, Postmus PE, Giaccone G. Prognostic factors in patients with pleural mesothelioma: the European Organization for Research and Treatment of Cancer experience. J Clin Oncol. 1998;16:145–52.
Conflicts of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopci, E., Zucali, P.A., Ceresoli, G.L. et al. Quantitative analyses at baseline and interim PET evaluation for response assessment and outcome definition in patients with malignant pleural mesothelioma. Eur J Nucl Med Mol Imaging 42, 667–675 (2015). https://doi.org/10.1007/s00259-014-2960-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-014-2960-y