Abstract
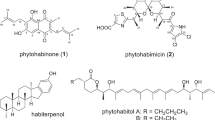
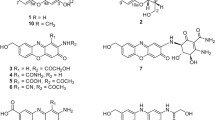
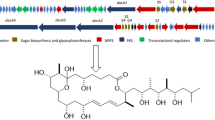
Ten new pentangular polyphenols, namely amexanthomycins A–J (1–10) were isolated from the strain Amycolatopsis mediterranei S699∆rifA constructed by deleting the polyketide synthase genes responsible for the biosynthesis of rifamycins. Their structures were elucidated on the basis of 1D and 2D NMR spectroscopic data and high-resolution ESIMS. Amexanthomycins A–C (1–3) showed inhibitory activity against human DNA topoisomerases.





Similar content being viewed by others
References
August PR, Tang L, Yoon YJ, Ning S, Muller R, Yu TW, Taylor M, Hoffmann D, Kim CG, Zhang X, Hutchinson CR, Floss HG (1998) Biosynthesis of the ansamycin antibiotic rifamycin: deductions from the molecular analysis of the rif biosynthetic gene cluster of Amycolatopsis mediterranei S699. Chem Biol 5(2):69–79. https://doi.org/10.1016/S1074-5521(98)90141-7
Berdy J (2005) Bioactive microbial metabolites. J Antibiot (Tokyo) 58(1):1–26. https://doi.org/10.1038/ja.2005.1
Cragg GM, Newman DJ (2013) Natural products: a continuing source of novel drug leads. Biochim Biophys Acta 1830(6):3670–3695. https://doi.org/10.1016/j.bbagen.2013.02.008
Demain AL (2014) Importance of microbial natural products and the need to revitalize their discovery. J Ind Microbiol Biotechnol 41(2):185–201. https://doi.org/10.1007/s10295-013-1325-z
Hertweck C (2009) The biosynthetic logic of polyketide diversity. Angew Chem Int Ed Engl 48(26):4688–4716. https://doi.org/10.1002/anie.200806121
Hertweck C, Luzhetskyy A, Rebets Y, Bechthold A (2007) Type II polyketide synthases: gaining a deeper insight into enzymatic teamwork. Nat Prod Rep 24(1):162–190. https://doi.org/10.1039/b507395m
Kang HS, Brady SF (2014a) Arixanthomycins A-C: phylogeny-guided discovery of biologically active eDNA-derived pentangular polyphenols. ACS Chem Biol 9(6):1267–1272. https://doi.org/10.1021/cb500141b
Kang HS, Brady SF (2014b) Mining soil metagenomes to better understand the evolution of natural product structural diversity: pentangular polyphenols as a case study. J Am Chem Soc 136(52):18111–18119. https://doi.org/10.1021/ja510606j
Kim CG, Kirschning A, Bergon P, Ahn Y, Wang JJ, Shibuya M, Floss HG (1992) Formation of 3-amino-5-hydroxybenzoic acid, the precursor of Mc(7)N units in ansamycin antibiotics, by a new variant of the shikimate pathway. J Am Chem Soc 114:4941-4943 doi: Doi https://doi.org/10.1021/Ja00038a090
Ma FX, Kim JH, Kim SB, Seo YG, Chang YK, Hong SK, Kim CJ (2008) Medium optimization for enhanced production of rifamycin B by Amycolatopsis mediterranei S699: combining a full factorial design and a statistical approach. Process Biochem 43(9):954–960. https://doi.org/10.1016/j.procbio.2008.04.021
Nagarajan R (1991) Antibacterial activities and modes of action of vancomycin and related glycopeptides. Antimicrob Agents Chemother 35(4):605–609. https://doi.org/10.1128/AAC.35.4.605
Sensi P, Margalith P, Timbal MT (1959) Rifomycin, a new antibiotic; preliminary report. Farmaco Sci 14(2):146–147
Stratmann A, Toupet C, Schilling W, Traber R, Oberer L, Schupp T (1999) Intermediates of rifamycin polyketide synthase produced by an Amycolatopsis mediterranei mutant with inactivated rifF gene. Microbiology 145(12):3365–3375. https://doi.org/10.1099/00221287-145-12-3365
Tang B, Zhao W, Zheng H, Zhuo Y, Zhang L, Zhao GP (2012) Complete genome sequence of Amycolatopsis mediterranei S699 based on de novo assembly via a combinatorial sequencing strategy. J Bacteriol 194(20):5699–5700. https://doi.org/10.1128/JB.01295-12
Tiwari K, Gupta RK (2012) Rare actinomycetes: a potential storehouse for novel antibiotics. Crit Rev Biotechnol 32(2):108–132. https://doi.org/10.3109/07388551.2011.562482
Verma M, Kaur J, Kumar M, Kumari K, Saxena A, Anand S, Nigam A, Ravi V, Raghuvanshi S, Khurana P, Tyagi AK, Khurana JP, Lal R (2011) Whole genome sequence of the rifamycin B-producing strain Amycolatopsis mediterranei S699. J Bacteriol 193(19):5562–5563. https://doi.org/10.1128/JB.05819-11
Woo S, Jung J, Lee C, Kwon Y, Na Y (2007) Synthesis of new xanthone analogues and their biological activity test—cytotoxicity, topoisomerase II inhibition, and DNA cross-linking study. Bioorg Med Chem Lett 17(5):1163–1166. https://doi.org/10.1016/j.bmcl.2006.12.030
Woo S, Kang DH, Nam JM, Lee CS, Ha EM, Lee ES, Kwon Y, Na Y (2010) Synthesis and pharmacological evaluation of new methyloxiranylmethoxyxanthone analogues. Eur J Med Chem 45(9):4221–4228. https://doi.org/10.1016/j.ejmech.2010.06.017
Woodley CL, Kilburn JO (1982) In vitro susceptibility of Mycobacterium avium complex and Mycobacterium tuberculosis strains to a spiro-piperidyl rifamycin. Am Rev Respir Dis 126(3):586–587. https://doi.org/10.1164/arrd.1982.126.3.586
Xu J, Mahmud T, Floss HG (2003) Isolation and characterization of 27-O-demethylrifamycin SV methyltransferase provides new insights into the post-PKS modification steps during the biosynthesis of the antitubercular drug rifamycin B by Amycolatopsis mediterranei S699. Arch Biochem Biophys 411(2):277–288. https://doi.org/10.1016/S0003-9861(03)00004-3
Xu J, Wan E, Kim CJ, Floss HG, Mahmud T (2005) Identification of tailoring genes involved in the modification of the polyketide backbone of rifamycin B by Amycolatopsis mediterranei S699. Microbiology 151(8):2515–2528. https://doi.org/10.1099/mic.0.28138-0
Yu TW, Muller R, Muller M, Zhang X, Draeger G, Kim CG, Leistner E, Floss HG (2001) Mutational analysis and reconstituted expression of the biosynthetic genes involved in the formation of 3-amino-5-hydroxybenzoic acid, the starter unit of rifamycin biosynthesis in Amycolatopsis Mediterranei S699. J Biol Chem 276(16):12546–12555. https://doi.org/10.1074/jbc.M009667200
Zhang ZQ, Wu XK, Song RT, Zhang JL, Wang HX, Zhu J, Lu CH, Shen YM (2017) Ansavaricins F-I, new DNA topoisomerase inhibitors produced by Streptomyces sp S012. RSC Adv 7(24):14857–14867. https://doi.org/10.1039/c7ra00961e
Funding
This work was supported by the National Natural Science Foundation of China (81373304, 81673317), Science Foundation of Two Sides of Strait (U1405223), and Program for Changjiang Scholars and Innovative Research Team in University (IRT_17R68).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Electronic supplementary material (ESI) available: The NMR spectra and HRESIMS of all the compounds and disruption of PKS genes.
Electronic supplementary material
ESM 1
(PDF 14512 kb)
Rights and permissions
About this article
Cite this article
Li, X., Wu, X., Zhu, J. et al. Amexanthomycins A–J, pentangular polyphenols produced by Amycolatopsis mediterranei S699∆rifA . Appl Microbiol Biotechnol 102, 689–702 (2018). https://doi.org/10.1007/s00253-017-8648-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8648-z