Abstract
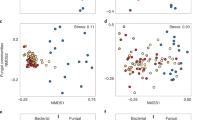
Wildfires subject soil microbes to extreme temperatures and modify their physical and chemical habitat. This might immediately alter their community structure and ecosystem functions. We burned a fire-prone shrubland under controlled conditions to investigate (1) the fire-induced changes in the community structure of soil archaea, bacteria and fungi by analysing 16S or 18S rRNA gene amplicons separated through denaturing gradient gel electrophoresis; (2) the physical and chemical variables determining the immediate shifts in the microbial community structure; and (3) the microbial drivers of the change in ecosystem functions related to biogeochemical cycling. Prokaryotes and eukaryotes were structured by the local environment in pre-fire soils. Fire caused a significant shift in the microbial community structure, biomass C, respiration and soil hydrolases. One-day changes in bacterial and fungal community structure correlated to the rise in total organic C and NO −3 –N caused by the combustion of plant residues. In the following week, bacterial communities shifted further forced by desiccation and increasing concentrations of macronutrients. Shifts in archaeal community structure were unrelated to any of the 18 environmental variables measured. Fire-induced changes in the community structure of bacteria, rather than archaea or fungi, were correlated to the enhanced microbial biomass, CO2 production and hydrolysis of C and P organics. This is the first report on the combined effects of fire on the three biological domains in soils. We concluded that immediately after fire the biogeochemical cycling in Mediterranean shrublands becomes less conservative through the increased microbial biomass, activity and changes in the bacterial community structure.





Similar content being viewed by others
References
Acea MJ, Carballas T (1996) Changes in physiological groups of microorganisms in soil following wildfire. FEMS Microbiol Ecol 20:33–39
Acea MJ, Carballas T (1999) Microbial fluctuations after soil heating and organic amendment. Bioresour Technol 67:65–71
Acinas SG, Sarma-Rupavtarm R, Klepac-Ceraj V, Polz MF (2005) PCR-induced sequence artifacts and bias: insights from comparison of two 16S rRNA clone libraries constructed from the same sample. Appl Environ Microbiol 71:8966–8969
Bahn V, Krohn WB, O’Connor RJ (2008) Dispersal leads to spatial autocorrelation in species distributions: a simulation model. Ecol Model 213:285–292
Bárcenas-Moreno G, Bååth E (2009) Bacterial and fungal growth in soil heated at different temperatures to simulate a range of fire intensities. Soil Biol Biochem 41:2517–2526
Bremner JM, Mulvaney CS (1982) Nitrogen-total. In: Page AL, Miller RH, Keeny DR (eds) Methods of soil analysis. Part 2. Chemical and microbiological properties, 2nd edn. American Society of Agronomy, Madison, pp 595–622
Brink RH, Dubar P, Linch DL (1960) Measurement of carbohydrates in soil hydrolysates with anthrone. Soil Sci 89:157–166
Busse MD, Hubbert KR, Fiddler GO, Shestak CJ, Powers RF (2005) Lethal soil temperatures during burning of masticated forest residues. Int J Wildland Fire 14:267–276
Campbell CD, Cameron CM, Bastias BA, Chen C, Cairney JWG (2008) Long term repeated burning in a wet sclerophyll forest reduces fungal and bacterial biomass and responses to carbon substrates. Soil Biol Biochem 40:2246–2252
Certini G (2005) Effects of fire on properties of forest soils: a review. Oecologia 143:1–10
Choromanska U, DeLuca TH (2002) Microbial activity and nitrogen mineralization in forest mineral soils following heating: evaluation of post-fire effects. Soil Biol Biochem 34:263–271
Ciardi C, Nannipieri P (1990) A comparison of methods for measuring ATP in soil. Soil Biol Biochem 22:725–727
Daniel RM, Cowan DA (2000) Biomolecular stability and life at high temperatures. Cell Mol Life Sci 57:250–264
De Marco A, Gentile AE, Arena C, De Santo AV (2005) Organic matter, nutrient content and biological activity in burned and unburned soils of a Mediterranean maquis area of southern Italy. Int J Wildland Fire 14:365–377
Dworkin M (2006) Prokaryotic life cycles. In: Dworkin M, Falkow S, Rosenberg E, Schleifer K-H, Stackebrandt E (eds) The prokaryotes. A handbook on the biology of bacteria. Ecophysiological and biochemical aspects, vol 2. Springer, New York, pp 140–166
Eivazi F, Tabatabai MA (1988) Glucosidases and galactosidases in soils. Soil Biol Biochem 20:601–606
Ekinci H (2006) Effect of forest fire on some physical, chemical and biological properties of soil in Çanakkale, Turkey. Int J Agric Biol 8:102–106
FAO–ISRIC–IUSS (2006) World Reference Base for Soil Resources. FAO, Rome
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci U S A 103:626–631
Finlay BJ (2002) Global dispersal of free-living microbial eukaryote species. Science 296:1061
Goberna M, Insam H, Klammer S, Pascual JA, Sánchez J (2005) Microbial community structure at different depths in disturbed and undisturbed semiarid Mediterranean forest soils. Microb Ecol 50:315–326
Goberna M, Sánchez J, Pascual JA, García C (2007) Pinus halepensis Mill. plantations did not restore the native microbial communities in a semiarid Mediterranean soil. Appl Soil Ecol 36:107–115
González MA, Roger A, Courtois EA, Jabot F, Norden N, Paine CET, Baraloto C, Thébaud C, Chave J (2010) Shifts in species and phylogenetic diversity between sapling and tree communities indicate negative density dependence in a lowland rain forest. J Ecol 98:137–146
Großkopf R, Janssen PH, Liesack W (1998) Diversity and structure of the methanogenic community in anoxic rice paddy soil microcosms as examined by cultivation and direct 16S rRNA gene sequence retrieval. Appl Environ Microbiol 64:960–969
Guerrero C, Mataix-Solera J, Gómez I, García-Orenes F, Jordán MM (2005) Microbial recolonization and chemical changes in soils heated at different temperatures. Int J Wildland Fire 14:385–400
Hamman ST, Burke IC, Stromberger ME (2007) Relationships between microbial community structure and soil environmental conditions in a recently burned system. Soil Biol Biochem 39:1703–1711
Hart SC, DeLuca TH, Newman GS, MacKenzie MD, Boyle S (2005) Post-fire vegetative dynamics as drivers of microbial community structure and function in forest soils. For Ecol Manag 220:166–184
Hernández T, García C, Reinhardt I (1997) Short-term effect of wildfire on the chemical, biochemical and microbiological properties of Mediterranean pine forest soils. Biol Fertil Soils 25:109–116
Heuer H, Krsek M, Baker P, Smalla K, Wellington EMH (1997) Analysis of actinomycete communities by specific amplification of genes encoding 16S rRNA and gel-electrophoretic separation in denaturing gradients. Appl Environ Microbiol 63:3233–3241
Hoare JP, Laidler KJ (1950) The molecular kinetics of the urea–urease system. II. The inhibition by products. J Am Chem Soc 72:2487–2489
Izzo A, Canright M, Bruns T (2006) The effects of heat treatments on ectomycorrhizal resistant propagules and their ability to colonize bioassay seedlings. Mycol Res 110:196–202
Jia Z, Conrad R (2009) Bacteria rather than Archaea dominate microbial ammonia oxidation in an agricultural soil. Environ Microbiol 11:1658–1671
Jiménez-Esquilín AE, Stromberger ME, Shepperd WD (2008) Soil scarification and wildfire interactions and effects on microbial communities and carbon. Soil Sci Soc Am J 72:111–118
Kandeler E, Gerber H (1988) Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol Fertil Soils 6:68–72
Kuske CR, Ticknor LO, Miller ME, Dunbar JM, Davis JA, Barns SM, Belnap J (2002) Comparison of soil bacterial communities in rhizospheres of three plant species and the interspaces in an arid grassland. Appl Environ Microbiol 68:1854–1863
Leininger S, Urich T, Schloter M, Schwark L, Qi J, Nicol GW, Prosser JI, Schuster SC, Schleper C (2006) Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 442:806–809
Madigan MT, Martinko JM, Dunlap PV, Clark DP (2009) Brock. Biology of microorganisms. Pearson, Upper Saddle River
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Martiny JBH, Bohannan BJM, Brown JH, Colwell RK, Fuhrman JA, Green JL, Horner-Devine MC, Kane M, Adams Krumins J, Kuske CR, Morin PJ, Naeem S, Øvreås L, Reysenbach AL, Smith VH, Staley JT (2006) Microbial biogeography: putting microorganisms on the map. Nat Rev Microbiol 4:102–112
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nannipieri P, Grego S, Ceccanti B (1990) Ecological significance of the biological activity in soil. In: Bollag JM, Stotzky G (eds) Soil biochemistry. Marcel Dekker, New York, pp 293–355
Neary DG, Klopatek CC, DeBano LF, Folliott PF (1999) Fire effects on belowground sustainability: a review and synthesis. For Ecol Manag 122:51–71
Nicol GW, Leininger S, Schleper C, Prosser JI (2008) The influence of soil pH on the diversity, abundance and transcriptional activity of ammonia oxidizing archaea and bacteria. Environ Microbiol 10:2966–2978
Noguez AM, Arita HT, Escalante AE, Forney LJ, García-Oliva F, Souza V (2005) Microbial macroecology: highly structured prokaryotic soil assemblages in a tropical deciduous forest. Global Ecol Biogeogr 14:241–248
Nübel U, Engelen B, Felske A, Snaidr J, Wieshuber A, Amann RI, Ludwig W, Backhaus H (1996) Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. J Bacteriol 178:5636–5643
Peay KG, Garbelotto M, Bruns TD (2009) Spore heat resistance plays an important role in disturbance-mediated assemblage shift of ectomycorrhizal fungi colonizing Pinus muricata seedlings. J Ecol 97:537–547
Prieto-Fernández A, Carballas M, Carballas T (2004) Inorganic and organic N pools in soils burned or heated: immediate alterations and evolution after forest wildfires. Geoderma 121:291–306
R Development Core Team (2007) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. URL: http://www.R-project.org
Rincón A, Pueyo JJ (2010) Effect of fire severity and site slope on diversity and structure of the ectomycorrhizal fungal community associated with post-fire regenerated Pinus pinaster Ait. seedlings. For Ecol Manag 260:361–369
Rothschild LJ, Mancinelli RL (2001) Life in extreme environments. Nature 402:1092–1101
Sekiguchi H, Tomioka N, Nakahara T, Uchiyama H (2001) A single band does not always represent single bacterial strains in denaturing gradient gel electrophoresis analysis. Biotechnol Lett 23:1205–1208
Sims JR, Haby VA (1971) Simplified colorimetric determination of soil organic matter. Soil Sci 112:137–141
Sinsabaugh RL (1994) Enzymic analysis of microbial pattern and process. Biol Fertil Soils 17:69–74
Skujiņš J, Klubek B (1982) Soil biological properties of a montane forest sere: corroboration of Odum's postulates. Soil Biol Biochem 14:505–513
Smith NR, Kishchuk BE, Mohn WW (2008) Effects of wildfire and harvest disturbances on forest soil bacterial communities. Appl Environ Microbiol 74:216–224
Stetter KO (1999) Extremophiles and their adaptation to hot environments. FEBS Lett 452:22–25
Tabatabai MA (1994) Soil enzymes. In: Weaver RW (ed) Methods of soil analysis, part 2. Microbiological and biochemical properties. Soil Science Society of America, Madison, pp 775–833
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307
Telford RJ, Vandvik V, Birks HJB (2006) Dispersal limitations matter for microbial morphospecies. Science 312:1015
ter Braak CJF, Šmilauer P (2002) CANOCO Reference Manual and CanoDraw for Windows User's Guide: Software for Canonical Community Ordination (version 4.5). Microcomputer Power, Ithaca NY, USA
Vainio EJ, Hantula J (2000) Direct analysis of wood-inhabiting fungi using denaturing gradient gel electrophoresis of amplified ribosomal DNA. Mycol Res 104:927–936
Van der Heijden MGA, Bardgett RD, Van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707
Vázquez FJ, Acea MJ, Carballas T (1993) Soil microbial populations after wildfire. FEMS Microbiol Ecol 13:93–104
Webster JJ, Hampton GJ, Leach FR (1984) ATP in soil: a new extractant and extraction procedure. Soil Biol Biochem 16:335–342
Yeager CM, Northup DE, Grow CC, Barns SM, Kuske CR (2005) Changes in nitrogen-fixing and ammonia-oxidizing bacterial communities in soil of a mixed conifer forest after wildfire. Appl Environ Microbiol 71:2713–2722
Yu Z, García-González R, Schanbacher FL, Morrison M (2008) Evaluations of different hypervariable regions of archaeal 16S rRNA genes in profiling of methanogens by Archaea-specific PCR and denaturing gradient gel electrophoresis. Appl Environ Microbiol 74:889–893
Acknowledgements
Financial support was provided by the EU Marie Curie Programme (FP7-PEOPLE-2009-RG-248155) and the Spanish Ministry of Science and Innovation (JAE-Doc Programme) to MG and CG, and the projects VAMPIRO (CGL2008-05289-C02-01) and Linktree (EUI2008-03721) to MV. Experimental burning was organised within the GRACCIE research net (CONSOLIDER-Ingenio program, Ministry of Science and Innovation, Spain). We thank the organisers, especially Jaime Baeza, for kindly inviting us to participate and providing us with data on soil temperature during burning. Thanks to Santiago Donat, Mª Dolores Martínez Soto and Eva Mª Andreu Gumbau for technical assistance. The authors appreciate comments on the manuscript by three anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goberna, M., García, C., Insam, H. et al. Burning Fire-Prone Mediterranean Shrublands: Immediate Changes in Soil Microbial Community Structure and Ecosystem Functions. Microb Ecol 64, 242–255 (2012). https://doi.org/10.1007/s00248-011-9995-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-011-9995-4