Abstract
Introduction
The current study seeks to provide preliminary data regarding this central, adaptive response during tongue motor tasks utilizing functional magnetic resonance imaging (fMRI) before and after glossectomy.
Methods
Six patients, with confirmed histological diagnoses of oral tongue cancer, underwent fMRI before and 6 months after partial glossectomy. These data were compared to nine healthy controls. All subjects performed three tongue motor tasks during fMRI: tongue tapping (TT), dry swallow (Dry), and wet swallow (Wet).
Results
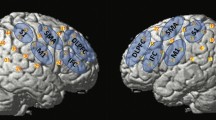
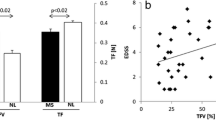
Following surgery, increased activation was subjectively observed in the superior parietal lobule, supplementary motor area, and anterior cingulate. Region of interest (ROI) analysis of the precentral gyrus confirmed increased cortical activity following surgery. In addition, comparisons between pre-surgical scans and controls suggested the dry swallow task was sensitive to elicit tongue-related activation in the precentral gyrus (p ≤ 0.05).
Conclusions
The adaptive changes in the cortex following partial glossectomy reflect recruitment of the parietal, frontal, and cingulate cortex during tongue motor tasks. In addition, post-operative activation patterns more closely approximated control levels than the pre-operative scans. Furthermore, the dry swallow task appears most specific to elicit tongue-related cortical activity.




Similar content being viewed by others
References
American Cancer Society (2008) Cancer Facts & Figures 2008. Atlanta
Sawczuk A, Mosier KM (2001) Neural control of tongue movement with respect to respiration and swallowing. Crit Rev Oral Bio Med 12(1):18–37
Pauloski B, Rademaker A, Logemann J, McConnel F, Heiser M, Cardinale S, Lazarus C, Peizer H, Stein D, Beery Q (2004) Surgical variables affecting swallowing in patients treated for oral/pharyngeal cancer. Head Neck 26:625–636
Pauloski B, Logemann J, Colangelo L, Rademaker A, McConnel F, Heiser M, Cardinale S, Shedd D, Stein D, Beery Q, Myers E, Lewin J, Haxer M, Esclamado R (2008) Surgical variables affecting speech in treated patients with oral and oropharyngeal cancer. Laryngoscope 108(6):908–916
LaBlance G, Kraus K, Steckol K (1991) Rehabilitation of swallowing and communication following glossectomy. Rehabil Nurs 16(5):266–270
Naeser M, Martin P, Nicholas M, Errol B, Seekins H, Kobayashi M, Theoret H, Fregni F, Maria-Tormos J, Kurland J, Doron K, Pascual-Leone A (2005) Improved picture naming in chronic aphasia after TMS to part of Broca’s area: An open-protocol study. Brain Lang 93:95–105
Mosier K, Patel R, Liu WC, Kalnin A, Maldjian J, Baredes S (1999) Cortical representation of swallowing in normal adults: functional implications. Laryngoscope 109(9):1417–1423
Rossini PM, Dal Forno G (2004) Integrated technology for evaluation of brain function and neural plasticity. Phys Med Rehabil Clin N Am 15(1):263–306
Weiller C, Chollet F, Friston KJ, Wise RJ, Frackowiak RS (1992) Functional reorganization of the brain in recovery form striatocapsular infarction in man. Ann Neurol 31(5):463–472
Mosier K, Liu WC, Behin B, Lee C, Baredes S (2005) Cortical adaptation following partial glossectomy with primary closure: implications for reconstruction of the oral tongue. Ann Otol Rhinol Laryngol 114(9):681–687
Cox RW (1996) AFNI: Software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res 29(3):162–173
Bandettni P, Jesmanowicz A, Wong E, Hyde J (1993) Processing strategies for time-course data sets in functional MRI of the human brain. Magn Reson Med 30:161–173
Mosier K, Bereznaya I (2001) Parallel cortical networks for volitional control of swallowing in humans. Exp Brain Res 140:280–289
Ertekin C, Aydogdu I (2003) Neurophysiology of swallowing. Clin Neurophysiol 114:2226–2244
Malandraki G, Sutton B, Perlman A, Karampinos D, Conway C (2009) Neural activation of swallowing and swallowing-related tasks in healthy young adults: An attempt to separate the components of deglutition. Human Brain Mapping 30(10):3209–3226
Watanabe Y, Abe S, Ishikawa T, Yamada Y, Yamane GY (2004) Cortical regulation during the early stage of initiation of voluntary swallowing in humans. Dysphagia 19(2):100–108
Martin R, Goodyear B, Gati J, Menon R (2001) Cerebral cortical representation of automatic and volitional swallowing in humans. J Neurophysiol 85:938–950
Malandraki GA, Sutton BP, Perlman AL, Karampinos DC (2009) Age-related differences in laterality of cortical activations in swallowing. Dysphagia. doi:10.1007/s00455-009-9250-z
Hajnal J, Myers R, Oatridge A et al (1994) Artifacts due to stimulus correlated motion in funcational imaging of the brain. Magn Reson Med 31:283–291
Friston KJ, Zarahan E, Josephs O, Henson RNA, Dale AM (1999) Stochastic designs in event-related fMRI. Neuroimage 10:607–619
Dale AM, Buckner RL (1997) Selective averaging of individual trials using fMRI. Hum Brain Mapp 5:329–340
Acknowledgements
The study was funded by Hackers for Hope, the Langeloth Foundation, and the Garban Fund.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was presented at the 2010 Combined Otolaryngology Spring Meetings/American Head and Neck Society.
Rights and permissions
About this article
Cite this article
Haupage, S., Peck, K.K., Branski, R.C. et al. Functional MRI of tongue motor tasks in patients with tongue cancer: observations before and after partial glossectomy. Neuroradiology 52, 1185–1191 (2010). https://doi.org/10.1007/s00234-010-0748-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-010-0748-8