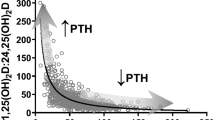
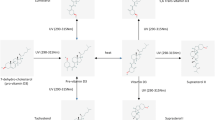
Abstract
Clinical interest in Vitamin D and its purported roles not only in calcium and bone metabolism but in several other medical conditions (diabetes, cardiovascular disease, multiple sclerosis, cancer, psychiatric disorders, neuro-muscular disease) has led to a surge in laboratory requests for 25 hydroxy vitamin D and 1,25 dihydroxy vitamin D measurement. Circulating 25 hydroxy vitamin D concentration is routinely used as the best indicator of vitamin D status, but measurement of other metabolites, especially the physiologically active 1,25 dihyroxy vitamin D, are of clinical value. Over the last 40 years the development of assays for vitamin D and its metabolites from early competitive binding assays through to immunoassay and liquid chromatography aligned to mass spectrometry have demonstrated various analytical challenges, the advantages and disadvantages of each method are constantly changing with new technological developments. Immunoassay remains the predominant mode of measurement for 25-hydroxy vitamin D although problems with equimolar recovery of the D2 and D3 metabolites remain an issue. Standardisation of all assays has been improved but not resolved with the currently available reference materials as evidenced by the international vitamin D external quality assurance scheme, DEQAS. The choice of method for each laboratory remains a balance mainly between turn around time, convenience, cost and the specificity and accuracy of the information obtained. With increasing discussion and clinical interest surrounding other vitamin D metabolites the vitamin D assay debate is set to continue.

Similar content being viewed by others
References
Bouillon R, Okamura WH, Norman AW (1995) Structure–function relationships in the vitamin D endocrine system. Endocr Rev 16:200–257
Clemens TL, Adams JS, Nolan JM, Holick MF (1982) Measurement of circulating vitamin D in man. Clin Chim Acta 121:301–308
Barragry JM, France MW, Corless D, Gupta SP, Switala S, Boucher BJ, Cohen RD (1978) Intestinal cholecalciferol absorption in the elderly and in younger adults. Clin Sci Mol Med 55:213–220
Clemens TL, Zhou XY, Myles M, Endres D, Lindsay R (1986) Serum vitamin D2 and vitamin D3 metabolite concentrations and absorption of vitamin D2 in elderly subjects. J Clin Endocrinol Metab 63:656–660
Gallacher SJ, Cowan RA, Fraser WD, Logue FC, Jenkins A, Boyle IT (1994) Acute effects of intravenous 1 alpha-hydroxycholecalciferol on parathyroid hormone, osteocalcin and calcitriol in man. Eur J Endocrinol 130:141–145
Gray RW, Caldas AE, Wilz DR, Lemann J Jr, Smith GA, DeLuca HF (1978) Metabolism and excretion of 3H–1,25-(OH)2-vitamin D3 in healthy adults. J Clin Endocrinol Metab 46:756–765
Jones G (2008) Pharmacokinetics of vitamin D toxicity. Am J Clin Nutr 88:582S–586S
Papapoulos SE, Clemens TL, Sandler LM, Fraher LJ, Winer J, O’Riordan JL (1982) The effect of renal function on changes in circulating concentrations of 1,25-dihydroxycholecalciferol after an oral dose. Clin Sci (Lond) 62:427–429
Haddad JG, Stamp TC (1974) Circulating 25-hydroxyvitamin D in man. Am J Med 57:57–62
Shephard RM, DeLuca HF (1980) Plasma concentrations of vitamin D3 and its metabolites in the rat as influenced by vitamin D3 or 25-hydroxyvitamin D3 intakes. Arch Biochem Biophys 202:43–53
Zehnder D, Bland R, Williams MC, McNinch RW, Howie AJ, Stewart PM, Hewison M (2001) Extrarenal expression of 25-hydroxyvitamin D3–1 alpha-hydroxylase. J Clin Endocrinol Metab 86:888–894
Need AG, O’Loughlin PD, Morris HA, Coates PS, Horowitz M, Nordin BE (2008) Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J Bone Miner Res 23:1859–1863
Haddad JG, Chyu KJ (1971) Competitive protein-binding radioassay for 25-hydroxycholecalciferol. J Clin Endocrinol Metab 33:992–995
Bayard F, Bec P, Louvet JP (1972) Measurement of plasma 25-hydroxycholecalciferol in man. Eur J Clin Invest 2:195–198
Belsey R, DeLuca HF, Potts JT Jr (1971) Competitive binding assay for vitamin D and 25-OH vitamin D. J Clin Endocrinol Metab 33:554–557
Bouillon R, Kerkhove PV, De MP (1976) Measurement of 25-hydroxyvitamin D3 in serum. Clin Chem 22:364–368
Edelstein S, Charman M, Lawson DE, Kodicek E (1974) Competitive protein-binding assay for 25-hydroxycholecalciferol. Clin Sci Mol Med 46:231–240
Preece MA, O’Riordan JL, Lawson DE, Kodicek E (1974) A competitive protein-binding assay for 25-hydroxycholecalciferol and 25-hydroxyergocalciferol in serum. Clin Chim Acta 54:235–242
Dorantes LM, Arnaud SB, Arnaud CD (1978) Importance of the isolation of 25-hydroxyvitamin D before assay. J Lab Clin Med 91:791–796
Glendenning P, Noble JM, Taranto M, Musk AA, McGuiness M, Goldswain PR, Fraser WD, Vasikaran SD (2003) Issues of methodology, standardization and metabolite recognition for 25-hydroxyvitamin D when comparing the DiaSorin radioimmunoassay and the Nichols Advantage automated chemiluminescence protein-binding assay in hip fracture cases. Ann Clin Biochem 40:546–551
Glendenning P, Taranto M, Noble JM, Musk AA, Hammond C, Goldswain PR, Fraser WD, Vasikaran SD (2006) Current assays overestimate 25-hydroxyvitamin D3 and underestimate 25-hydroxyvitamin D2 compared with HPLC: need for assay-specific decision limits and metabolite-specific assays. Ann Clin Biochem 43:23–30
Leventis P, Garrison L, Sibley M, Peterson P, Egerton M, Levin G, Kiely P (2005) Underestimation of serum 25-hydroxyvitamin D by the Nichols Advantage Assay in patients receiving vitamin D replacement therapy. Clin Chem 51:1072–1074
Hollis BW, Napoli JL (1985) Improved radioimmunoassay for vitamin D and its use in assessing vitamin D status. Clin Chem 31:1815–1819
Hollis BW, Kamerud JQ, Selvaag SR, Lorenz JD, Napoli JL (1993) Determination of vitamin D status by radioimmunoassay with an 125I-labeled tracer. Clin Chem 39:529–533
Ersfeld DL, Rao DS, Body JJ, Sackrison JL Jr, Miller AB, Parikh N, Eskridge TL, Polinske A, Olson GT, MacFarlane GD (2004) Analytical and clinical validation of the 25 OH vitamin D assay for the Liaison automated analyzer. Clin Biochem 37:867–874
Hollis BW (2000) Comparison of commercially available 125I-based RIA methods for the determination of circulating 25-hydroxyvitamin D. Clin Chem 46:1657–1661
Hypponen E, Turner S, Cumberland P, Power C, Gibb I (2007) Serum 25-hydroxyvitamin D measurement in a large population survey with statistical harmonization of assay variation to an international standard. J Clin Endocrinol Metab 92:4615–4622
Heijboer AC, Blankenstein MA, Kema IP, Buijs MM (2012) Accuracy of 6 routine 25-hydroxyvitamin D assays: influence of vitamin D binding protein concentration. Clin Chem 58:543–548
Binkley N, Krueger D, Lensmeyer G (2009) 25-hydroxyvitamin D measurement, 2009: a review for clinicians. J Clin Densitom 12:417–427
Binkley N, Krueger DC, Morgan S, Wiebe D (2010) Current status of clinical 25-hydroxyvitamin D measurement: an assessment of between-laboratory agreement. Clin Chim Acta 411:1976–1982
Carter GD, Carter R, Jones J, Berry J (2004) How accurate are assays for 25-hydroxyvitamin D? Data from the International Vitamin D External Quality Assessment Scheme. Clin Chem 50:2195–2197
Jones G (1978) Assay of vitamins D2 and D3, and 25-hydroxyvitamins D2 and D3 in human plasma by high-performance liquid chromatography. Clin Chem 24:287–298
Kleerekoper M, Schleicher RL, Eisman J, Bouillon R, Singh RJ, Holick MF (2011) Clinical applications for vitamin D assays: what is known and what is wished for. Clin Chem 57:1227–1232
Coldwell RD, Trafford DJ, Makin HL, Varley MJ, Kirk DN (1984) Specific estimation of 24,25-dihydroxyvitamin D in plasma by gas chromatography-mass spectrometry. Clin Chem 30:1193–1198
Eisman JA, Shepard RM, DeLuca HF (1977) Determination of 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3 in human plasma using high-pressure liquid chromatography. Anal Biochem 80:298–305
Gilbertson TJ, Stryd RP (1977) High-performance liquid chromatographic assay for 25-hydroxyvitamin D3 in serum. Clin Chem 23:1700–1704
Horst RL, Reinhardt TA, Hollis BW (1990) Improved methodology for the analysis of plasma vitamin D metabolites. Kidney Int Suppl 29:S28–S35
Liel Y, Ulmer E, Shary J, Hollis BW, Bell NH (1988) Low circulating vitamin D in obesity. Calcif Tissue Int 43:199–201
Lensmeyer GL, Wiebe DA, Binkley N, Drezner MK (2006) HPLC method for 25-hydroxyvitamin D measurement: comparison with contemporary assays. Clin Chem 52:1120–1126
Yeung B, Vouros P, Reddy GS (1993) Characterization of vitamin D3 metabolites using continuous-flow fast atom bombardment tandem mass spectrometry and high-performance liquid chromatography. J Chromatogr 645:115–123
Maunsell Z, Wright DJ, Rainbow SJ (2005) Routine isotope-dilution liquid chromatography-tandem mass spectrometry assay for simultaneous measurement of the 25-hydroxy metabolites of vitamins D2 and D3. Clin Chem 51:1683–1690
Tsugawa N, Suhara Y, Kamao M, Okano T (2005) Determination of 25-hydroxyvitamin D in human plasma using high-performance liquid chromatography–tandem mass spectrometry. Anal Chem 77:3001–3007
Vogeser M, Kyriatsoulis A, Huber E, Kobold U (2004) Candidate reference method for the quantification of circulating 25-hydroxyvitamin D3 by liquid chromatography-tandem mass spectrometry. Clin Chem 50:1415–1417
Annesley TM (2003) Ion suppression in mass spectrometry. Clin Chem 49:1041–1044
Knox S, Harris J, Calton L, Wallace AM (2009) A simple automated solid-phase extraction procedure for measurement of 25-hydroxyvitamin D3 and D2 by liquid chromatography-tandem mass spectrometry. Ann Clin Biochem 46:226–230
Chen H, McCoy LF, Schleicher RL, Pfeiffer CM (2008) Measurement of 25-hydroxyvitamin D3 (25OHD3) and 25-hydroxyvitamin D2 (25OHD2) in human serum using liquid chromatography-tandem mass spectrometry and its comparison to a radioimmunoassay method. Clin Chim Acta 391:6–12
Thibeault D, Caron N, Djiana R, Kremer R, Blank D (2012) Development and optimization of simplified LC-MS/MS quantification of 25-hydroxyvitamin D using protein precipitation combined with on-line solid phase extraction (SPE). J Chromatogr B Anal Technol Biomed Life Sci 883–884:120–127
Bunch DR, Miller AY, Wang S (2009) Development and validation of a liquid chromatography-tandem mass spectrometry assay for serum 25-hydroxyvitamin D2/D3 using a turbulent flow online extraction technology. Clin Chem Lab Med 47:1565–1572
Netzel BC, Cradic KW, Bro ET, Girtman AB, Cyr RC, Singh RJ, Grebe SK (2011) Increasing liquid chromatography-tandem mass spectrometry throughput by mass tagging: a sample-multiplexed high-throughput assay for 25-hydroxyvitamin D2 and D3. Clin Chem 57:431–440
Singh RJ, Taylor RL, Reddy GS, Grebe SK (2006) C-3 epimers can account for a significant proportion of total circulating 25-hydroxyvitamin D in infants, complicating accurate measurement and interpretation of vitamin D status. J Clin Endocrinol Metab 91:3055–3061
Lensmeyer G, Poquette M, Wiebe D, Binkley N (2012) The C-3 epimer of 25-hydroxyvitamin D3 is present in adult serum. J Clin Endocrinol Metab 97:163–168
Carter GD (2012) 25-Hydroxyvitamin D: a difficult analyte. Clin Chem 58:486–488
Schleicher RL, Encisco SE, Chaudhary-Webb M, Paliakov E, McCoy LF, Pfeiffer CM (2011) Isotope dilution ultra performance liquid chromatography-tandem mass spectrometry method for simultaneous measurement of 25-hydroxyvitamin D2, 25-hydroxyvitamin D3 and 3-epi-25-hydroxyvitamin D3 in human serum. Clin Chim Acta 412:1594–1599
Shah I, James R, Barker J, Petroczi A, Naughton DP (2011) Misleading measures in vitamin D analysis: a novel LC-MS/MS assay to account for epimers and isobars. Nutr J 10:46
Stepman HC, Vanderroost A, Van UK, Thienpont LM (2011) Candidate reference measurement procedures for serum 25-hydroxyvitamin D3 and 25-hydroxyvitamin D2 by using isotope-dilution liquid chromatography-tandem mass spectrometry. Clin Chem 57:441–448
Tai SS, Bedner M, Phinney KW (2010) Development of a candidate reference measurement procedure for the determination of 25-hydroxyvitamin D3 and 25-hydroxyvitamin D2 in human serum using isotope-dilution liquid chromatography-tandem mass spectrometry. Anal Chem 82:1942–1948
Cluse ZN, Fudge AN, Whiting MJ, McWhinney B, Parkinson I, O’Loughlin PD (2012) Evaluation of 25-hydroxy vitamin D assay on the immunodiagnostic systems iSYS analyser. Ann Clin Biochem 49:159–165
Cavalier E, Carlisi A, Bekaert AC, Rousselle O, Chapelle JP, Souberbielle JC (2012) Analytical evaluation of the new Abbott Architect 25-OH vitamin D assay. Clin Biochem 45:505–508
Cavalier E, Carlisi A, Bekaert AC, Rousselle O, Chapelle JP (2012) Human anti-animal interference in DiaSorin Liaison total 25(OH)-vitamin D assay: towards the end of a strange story? Clin Chim Acta 413:527–528
Farrell CJ, Martin S, McWhinney B, Straub I, Williams P, Herrmann M (2012) State-of-the-art vitamin D assays: a comparison of automated immunoassays with liquid chromatography-tandem mass spectrometry methods. Clin Chem 58:531–542
Carter GD, Jones JC (2009) Use of a common standard improves the performance of liquid chromatography-tandem mass spectrometry methods for serum 25-hydroxyvitamin-D. Ann Clin Biochem 46:79–81
Fraser WD (2009) Standardization of vitamin D assays: art or science? Ann Clin Biochem 46:3–4
Phinney KW, Bedner M, Tai SS, Vamathevan VV, Sander LC, Sharpless KE, Wise SA, Yen JH, Schleicher RL, Chaudhary-Webb M, Pfeiffer CM, Betz JM, Coates PM, Picciano MF (2012) Development and certification of a standard reference material for vitamin D metabolites in human serum. Anal Chem 84:956–962
Carter GD, Berry JL, Gunter E, Jones G, Jones JC, Makin HL, Sufi S, Wheeler MJ (2010) Proficiency testing of 25-hydroxyvitamin D (25-OHD) assays. J Steroid Biochem Mol Biol 121:176–179
Brumbaugh PF, Haussler DH, Bressler R, Haussler MR (1974) Radioreceptor assay for 1 alpha,25-dihydroxyvitamin D3. Science 183:1089–1091
Reinhardt TA, Horst RL, Orf JW, Hollis BW (1984) A microassay for 1,25-dihydroxyvitamin D not requiring high performance liquid chromatography: application to clinical studies. J Clin Endocrinol Metab 58:91–98
Clemens TL, Hendy GN, Graham RF, Baggiolini EG, Uskokovic MR, O’Riordan JL (1978) A radioimmunoassay for 1,25-dihydroxycholecalciferol. Clin Sci Mol Med 54:329–332
Bouillon R, De Moor P, Baggiolini EG, Uskokovic MR (1980) A radioimmunoassay for 1,25-dihydroxycholecalciferol. Clin Chem 26:562–567
Fraher LJ, Adami S, Clemens TL, Jones G, O’Riordan JL (1983) Radioimmunoassay of 1,25-dihydroxy vitamin D2: studies on the metabolism of vitamin D2 in man. Clin Endocrinol (Oxf) 19:151–165
Gray TK, McAdoo T, Pool D, Lester GE, Williams ME, Jones G (1981) A modified radioimmunoassay for 1,25-dihydroxycholecalciferol. Clin Chem 27:458–463
Hollis BW, Kamerud JQ, Kurkowski A, Beaulieu J, Napoli JL (1996) Quantification of circulating 1,25-dihydroxyvitamin D by radioimmunoassay with 125I-labeled tracer. Clin Chem 42:586–592
Clive DR, Sudhaker D, Giacherio D, Gupta M, Schreiber MJ, Sackrison JL, MacFarlane GD (2002) Analytical and clinical validation of a radioimmunoassay for the measurement of 1,25 dihydroxy vitamin D. Clin Biochem 35:517–521
Fraser WD, Durham BH, Berry JL, Mawer EB (1997) Measurement of plasma 1,25 dihydroxyvitamin D using a novel immunoextraction technique and immunoassay with iodine labelled vitamin D tracer. Ann Clin Biochem 34(Pt 6):632–637
Seiden-Long I, Vieth R (2007) Evaluation of a 1,25-dihydroxyvitamin D enzyme immunoassay. Clin Chem 53:1104–1108
Hollis BW (1995) 1,25-Dihydroxyvitamin D3–26,23-lactone interferes in determination of 1,25-dihydroxyvitamin D by RIA after immunoextraction. Clin Chem 41:1313–1314
Bjorkhem I, Holmberg I, Kristiansen T, Pedersen JI (1979) Assay of 1,25-dihydroxy vitamin D3 by isotope dilution–mass fragmentography. Clin Chem 25:584–588
Vreeken RJ, Honing M, van Baar BL, Ghijsen RT, de Jong GJ, Brinkman UA (1993) On-line post-column Diels-Alder derivatization for the determination of vitamin D3 and its metabolites by liquid chromatography/thermospray mass spectrometry. Biol Mass Spectrom 22:621–632
Kissmeyer AM, Sonne K (2001) Sensitive analysis of 1alpha,25-dihydroxyvitamin D3 in biological fluids by liquid chromatography-tandem mass spectrometry. J Chromatogr A 935:93–103
Aronov PA, Hall LM, Dettmer K, Stephensen CB, Hammock BD (2008) Metabolic profiling of major vitamin D metabolites using Diels-Alder derivatization and ultra-performance liquid chromatography-tandem mass spectrometry. Anal Bioanal Chem 391:1917–1930
Duan X, Weinstock-Guttman B, Wang H, Bang E, Li J, Ramanathan M, Qu J (2010) Ultrasensitive quantification of serum vitamin D metabolites using selective solid-phase extraction coupled to microflow liquid chromatography and isotope-dilution mass spectrometry. Anal Chem 82:2488–2497
Casetta B, Jans I, Billen J, Vanderschueren D, Bouillon R (2010) Development of a method for the quantification of 1alpha,25(OH)2-vitamin D3 in serum by liquid chromatography tandem mass spectrometry without derivatization. Eur J Mass Spectrom (Chichester, Eng) 16(1):81–89
Yuan C, Kosewick J, He X, Kozak M, Wang S (2011) Sensitive measurement of serum 1alpha,25-dihydroxyvitamin D by liquid chromatography/tandem mass spectrometry after removing interference with immunoaffinity extraction. Rapid Commun Mass Spectrom 25:1241–1249
Strathmann FG, Laha TJ, Hoofnagle AN (2011) Quantification of 1α,25-dihydroxy vitamin D by immunoextraction and liquid chromatography-tandem mass spectrometry. Clin Chem 57:1279–1285
Turner C, Dalton N, Inaoui R, Fogelman I, Fraser WD, Hampson G (2013) Effect of a 300 000-IU loading dose of ergocalciferol (vitamin D2) on circulating 1,25(OH) 2-vitamin D and Fibroblast Growth Factor-23 (FGF-23) in vitamin D insufficiency. J Clin Endocrinol Metab [Epub ahead of print]
Yetley EA, Pfeiffer CM, Schleicher RL, Phinney KW, Lacher DA, Christakos S, Eckfeldt JH, Fleet JC, Howard G, Hoofnagle AN, Hui SL, Lensmeyer GL, Massaro J, Peacock M, Rosner B, Wiebe D, Bailey RL, Coates PM, Looker AC, Sempos C, Johnson CL, Picciano MF (2010) NHANES monitoring of serum 25-hydroxyvitamin D: a roundtable summary. J Nutr 140:2030S–2045S
Acknowledgment
The authors thank Dr. Graham Carter for permission to use the synopsis of data from the January 2012 DEQAS report.
Disclosures
WD Fraser has given lectures on vitamin D measurements for all the companies listed in this manuscript currently involved in measuring vitamin D and provides advice on vitamin D measurement to IDS, Siemens, Roche and Nichols Diagnostics.
Author information
Authors and Affiliations
Corresponding author
Additional information
W. F. has a consultant/advisory role and has received funding from IDS, Siemens, Nichols, Roche, and Abbott and has patents/intellectual property with IDS. A. M. has stated no conflict of interest.
Rights and permissions
About this article
Cite this article
Fraser, W.D., Milan, A.M. Vitamin D Assays: Past and Present Debates, Difficulties, and Developments. Calcif Tissue Int 92, 118–127 (2013). https://doi.org/10.1007/s00223-012-9693-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-012-9693-3