Abstract
Rationale
Recent evidence suggests the involvement of the endocannabinoid (EC) system in the regulation of anxiety.
Objectives
The aim of present work was to study the role of the EC system in cat odour-induced anxiety in rats. Materials and methods Male Wistar rats were exposed to cat odour in home and motility cages. Exposure of rats to elevated zero-maze was used to determine changes in anxiety. Effect of rimonabant (0.3–3 mg/kg), antagonist of CB1 receptors, was studied on cat odour-induced alterations in exploratory behaviour. Real-time PCR was used to determine gene expression levels of EC-related genes in the brain.
Results
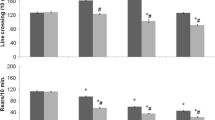
Anxiogenic-like action of cat odour was evident in the elevated zero-maze. Cat odour increased the expression of FAAH, the enzyme responsible for the degradation of anandamide, in the mesolimbic area. By contrast, in the amygdala and periaqueductal grey (PAG) levels of NAPE-PLD, the enzyme related to the synthesis of anandamide, and FAAH were remarkably decreased. Cat odour also decreased the expression of enzymes related to metabolism of 2-archidonoyl-glycerol in the amygdala and PAG. Pre-treatment of rats with rimonabant (0.3–3 mg/kg) reduced the exploratory behaviour of rats, but did not affect cat odour-induced changes.
Conclusion
Exposure to cat odour induces anxiogenic-like effect on the behaviour in rats. Cat odour also causes moderate increase in expression of EC-related genes in the mesolimbic area, whereas significant down-regulation is established in the amygdala and PAG. Relation of predator odour-induced anxiety to the inhibition of the EC system in the amygdala and PAG is supported by behavioural studies where blockade of CB1 receptors by rimonabant induces anxiogenic-like action.


Similar content being viewed by others
References
Adamec RE, Shallow T (1993) Lasting effects on rodent anxiety of a single exposure to a cat. Physiol Behav 54:101–109
Adamec RE, Blundell J, Burton P (2003) Phosphorylated cyclic AMP response element binding protein expression induced in the periaqueductal gray by predator stress: its relationship to the stress experience, behavior and limbic neural plasticity. Prog Neuropsychopharmacol Biol Psychiatry 27:1243–1267
Akinshola BE, Chakrabarti A, Onavi ES (1999) In-vitro and in-vivo action of cannabinoids. Neurochem Res 24:1233–1240
Areda T, Kõks S, Philips MA, Vasar E, Karis A, Asser T (2005) Alterations in opioid system of the rat brain after cat odor exposure. Neurosci Lett 377:136–139
Arévalo C, de Miquel R, Hernández-Tristán R (2001) Cannabinoid effects on anxiety-related behaviours and hypothalamic neurotransmitters. Pharmacol Biochem Behav 70:123–131
Berton F, Vogel E, Belzung C (1998) Modulation of mice anxiety in response to cat odor as a consequence of predators diet. Physiol Behav 65:247–254
Blanchard DC, Griebel G, Blanchard RJ (2003) Conditioning and residual emotionality effects of predator stimuli: some reflections on stress and emotion. Prog Neuropsychopharmacol Biol Psychiatry 27:1177–1185
Blundell J, Adamec R (2006) Elevated pCREB in the PAG after exposure to the elevated plus maze in rats previously exposed to a cat. Behav Brain Res 175:285–295
Cahill L, McGaugh JL (1998) Mechanisms of emotional arousal and lasting declarative memory. Trends Neurosci 21:294–299
Calvo-Torrent A, Brain PF, Martinez M (1999) Effect of predatory stress on sucrose intake and behavior on the plus-maze in male mice. Physiol Behav 67:189–196
Coplan JD, Lydiard RB (1998) Brain circuits in panic disorder. Biol Psychiatry 44:1264–1276
Davis M (1997) Neurobiology of fear responses: the role of the amygdala. J Neuropsychiatry Clin Neurosci 9:382–402
Day TA, Rakhshan F, Deutsch DG, Barker E-L (2001) Role of fatty acid amide hydrolase in the transport of the endogenous cannabinoid anandamide. Mol Pharmacol 59:1369–1375
Dielenberg RA, McGregor IS (2001) Defensive behavior in rats towards predatory odors: a review. Neurosci Biobehav Rev 25:597–609
Di Marzo V, Fontana A, Cadas H, Schinelli S, Cimino G et al (1994) Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature 372:686–691
Di Marzo V, Melck D, Bisogno T, De Petrocellis L (1998) Endocannabinoids: endogenous cannabinoid receptor ligands with neuromodulatory action. Trends Neurosci 21:521–528
Di Marzo V, Breivogel CS, Tao Q, Bridgen DT, Razdan RK, Zimmer AM, Zimmer A, Martin BR (2000) Levels, metabolism, and pharmacological activity of anandamide in CB (1) cannabinoid receptor knockout mice: evidence for non-CB (1), non-CB (2) receptor-mediated actions of anandamide in mouse brain. J Neurochem 75:2434–2444
Finn DA, Rutledge-Gorman MT, Crabbe JC (2003) Genetic animal models of anxiety. Neurogenetics 4:109–135
Griebel G, Stemmelin J, Scatton B (2005) Effects of the cannabinoid CB1 receptor antagonist rimonabant in models of emotional reactivity in rodents. Biol Psychiatry 57:261–267
Haller J, Bakos N, Szirmay M, Ledent C, Freund TF (2002) The effects of genetic and pharmacological blockade of the CB1 cannabinoid receptor on anxiety. Eur J Neurosci 16:1395–1398
Haller J, Varga B, Ledent C, Freund TF (2004) CB1 cannabinoid receptors mediate anxiolytic effects: convergent genetic and pharmacological evidence with CB1-specific agents. Behav Pharmacol 15:299–304
Hebb AL, Zacharko RM, Dominguez H, Trudel F, Laforest S, Drolet G (2002) Odor-induced variation in anxiety-like behavior in mice is associated with discrete and differential effects on mesocorticolimbic cholecystokinin mRNA expression. Neuropsychopharmacology 27:744–755
Hebb AL, Zacharko RM, Gauthier M, Drolet G (2003) Exposure of mice to a predator odor increases acoustic startle but does not disrupt the rewarding properties of VTA intracranial self-stimulation. Brain Res 982:195–210
Hendrie CA, Weiss SM, Eilam D (1996) Exploration and predation models of anxiety: evidence from laboratory and wild species. Pharmacol Biochem Behav 54:13–20
Herkenham M, Lynn AB, Johnson MR, Melvin LS, de Costa BR, Rice KC (1991) Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J Neurosci 11:563–583
Hohmann AG, Suplita RL, Bolton NM, Neely MH, Fegley D, Mangieri R, Krey JF, Walker JM, Holmes PV, Crystal JD, Duranti A, Tontini A, Mor M, Tarzia G, Piomelli D (2005) An endocannabinoid mechanism for stress-induced analgesia. Nature 435:1108–1112
Jackson ME, Moghaddam B (2001) Amygdala regulation of nucleus accumbens dopamine output is governed by the prefrontal cortex. J Neurosci 21:676–681
Kathuria S, Gaetani S, Fegley D, Valino F, Duranti A, Tontini A, Mor M, Tarzia G, La Rana G, Calignano A, Giustino A, Tattoli M, Palmery M, Cuomo V, Piomelli D (2003) Modulation of anxiety through blockade of anandamide hydrolysis. Nat Med 9:76–81
Katona I, Sperlagh B, Sik A, Kafalvi A, Vizi ES, Mackie K et al (1999) Presynaptically located CB1 cannabinoid receptors regulate GABA release from axon terminals of specific hippocampal interneurons. J Neurosci 19:4544–4558
Katona I, Rancz EA, Acsady L, Ledent C, Mackie K, Hajos N, Freund TF (2001) Distribution of CB1 cannabinoid receptors in the amygdala and their role in the control of GABAergic transmission. J Neurosci 21:9506–9518
Kelley AE (2004) Ventral striatal control of appetitive motivation: role in ingestive behavior and reward-related learning. Neurosci Biobehav Rev 27:765–776
Kirkham TC, Williams CM, Fezza F, Di Marzo V (2002) Endocannabinoid levels in rat limbic forebrain and hypothalamus in relation to fasting, feeding and satiation: stimulation of eating by 2-arachidonoyl glycerol. Br J Pharmacol 136:550–557
Klein TW, Newton C, Friedman H (1998) Cannabinoid receptors and the cytokine network. Adv Exp Med Biol 437:215–222
Kõks S, Luuk H, Nelovkov A, Areda T, Vasar E (2004) A screen for genes induced in the amygdaloid area during cat odor exposure. Genes Brain Behav 3:80–89
Lang PJ, Bradley MM, Cuthbert BN (1998) Emotion, motivation, and anxiety: brain mechanisms and psychophysiology. Biol Psychiatry 44:1248–1263
LeDoux J (1998) Fear and the brain: where have we been, and where are we going? Biol Psychiatry 44:1229–1238
LeDoux JE (2000) Emotion circuits in the brain. Annu Rev Neurosci 23:155–184
Leung D, Saghatelian A, Simon GM, Cravatt BF (2006) Inactivation of N-acyl phosphatidylethanolamine phospholipase D reveals multiple mechanisms for the biosynthesis of endocannabinoids. Biochemistry 45:4720–4726
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔC T method. Methods 25:402–408
Maccarrone M, Valverde O, Barbaccia ML, Castane A, Maldonado R, Ledent C, Parmentier M, Finazzi-Agro A (2002) Age-related changes of anandamide metabolism in CB1 cannabinoid receptor knockout mice: correlation with behavior. Eur J Neurosci 15:1178–1186
Marsicano G, Wotjak CT, Azad SC, Bisogno T, Rammes G, Cascio MG, Hermann H, Tang J, Hofmann C, Zieglgansberger W, Di Marzo V, Lutz B (2002) The endogenous cannabinoid system controls extinction of aversive memories. Nature 418:530–534
Martin M, Ledent C, Parmentier M, Maldonado R, Valverde O (2002) Involvement of CB1 cannabinoid receptors in emotional behavior. Psychopharmacology (Berl) 159:379–387
Matsuda La, Lolait SJ, Brownstein MJ, Young AC, Bonner TI (1990) Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 346:561–564
McGregor IS, Schrama L, Ambermoon P, Dielenberg RA (2002) Not all ‘predator odours’ are equal: cat odor but not 2,4,5 trimethylthiazoline (TMT; fox odor) elicits specific defensive behaviors in rats. Behav Brain Res 129:1–16
Mechoulam R, Fride E, Di Marzo V (1998) Endocannabinoids. Eur J Pharmacol 359:1–18
Millan MJ (2003) The neurobiology and control of anxious states. Prog Neurobiol 70:83–244
Mogenson GJ, Jones DL, Yim CY (1980) From motivation to action: functional interface between the limbic system and the motor system. Prog Neurobiol 14:69–97
Montgomery KC (1955) The relation between fear induced by novelty stimulation and exploratory behaviour. J Comp Physiol Psychol 48:254–260
Moreira CM, Masson S, Carvalho MC, Brandão ML (2007) Exploratory behaviour of rats in the elevated plus-maze is differentially sensitive to inactivation of the basolateral and central amygdaloid nuclei. Brain Res Bull 71:466–474
Navarro M, Hernandez E, Munoz RM, del Arco I, Villanua MA, Carrera MR, Rodriguez de Fonseca F (1997) Acute administration of the CB1 cannabinoid receptor antagonist SR 141716A induces anxiety-like responses in the rat. Neuroreport 20:491–496
Nelovkov A, Areda T, Innos J, Kõks S, Vasar E (2006) Rats displaying distinct exploratory activity also have different expression patterns of gamma-aminobutyric acid-and cholecystokinin-related genes in brain regions. Brain Res 1100:21–31
Phillips AG, Ahn S, Howland JG (2003) Amygdalar control of the mesocorticolimbic dopamine system: parallel pathways to motivated behavior. Neurosci Biobehav Rev 27:543–554
Pistis M, Perra S, Pillolla G, Melis M, Gessa GL, Muntoni AL (2004) Cannabinoids modulate neuronal firing in the rat basolateral amygdala: evidence for CB1- and non-CB1-mediated actions. Neuropharmacology 46:115–125
Porter AC, Felder CC (2001) The endocannabinoid nervous system: unique opportunities for therapeutic intervention. Pharmacol Ther 90:45–60
Pratt JA, Brett RR (1995) The benzodiazepine receptor inverse agonist FG 7142 induces cholecystokinin gene expression in rat brain. Neurosci Lett 184:197–200
Robbins TW, Everitt BJ (1996) Neurobehavioural mechanisms of reward and motivation. Curr Opin Neurobiol 6:228–236
Rodgers RJ (1997) Animal models of ‘anxiety’: where next? Behav Pharmacol 8:477–496
Rodgers RJ, Dalvi A (1997) Anxiety, defence and the elevated plus-maze. Neurosci Biobehav Rev 21:801–810
Rodgers RJ, Haller J, Halasz J, Mikics E (2003) ‘One-trial sensitization’ to the anxiolytic-like effects of cannabinoid receptor antagonist SR141716A in the mouse elevated plus-maze. Eur J Neurosci 17:1279–1286
Rutkowska M, Jamontt J, Gliniak H (2006) Effects of cannabinoids on the anxiety-like response in mice. Pharmacol Rep 58:200–206
Shepherd K, Grewal SS, Fletcher A, Bill DJ, Dourish CT (1994) Behavioral and pharmacological characterization of the elevated “zero-maze” as an animal model of anxiety. Psychopharmacology 116:56–64
Simon GM, Cravatt BF (2006) Endocannabinoid biosynthesis proceeding through glycerophospho-N-acyl ethanolamine and a role for alpha/beta-hydrolase 4 in this pathway. Biol Chem 281:26465–26472
Sugiura T, Kondo S, Sukagawa A, Nakane S, Shinoda A, Itoh K, Yamashita A, Waku K (1995) 2-Arachidonoylglycerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem Biophys Res Commun 215:89–97
Sugiura T, Kobayashi Y, Oka S, Waku K (2002) Biosynthesis and degradation of anandamide and 2-arachidonoylglycerol and their possible physiological significance. Prostaglandins Leukot Essent Fat Acids 66:173–192
Swanson LW (1998) Brain maps: structure of the rat brain, 2nd edn. Elsevier, Amsterdam
Tzavara ET, Wade M, Nomikos GG (2003) Biphasic effects of cannabinoids on acetylcholine release in the hippocampus: site and mechanism of action. J Neurosci 23:9374–9384
Van der Stelt M, Di Marzo V (2003) The endocannabinoid system in the basal ganglia and in the mesolimbic reward system: implications for neurological and psychiatric disorders. Eur J Pharmacol 480:133–150
Van Sickle MD, Duncan M, Kingsley PJ, Mouihate A, Urbani P, Mackie K, Stella N, Makriyannis A, Piomelli D, Davison JS, Marnett LJ, Di Marzo V, Pittman QJ, Patel KD, Sharkey KA (2005) Identification and functional characterization of brainstem cannabinoid CB2 receptors. Science 310:329–332
Viveros MP, Marco EM, File SE (2005) Endocannabinoid system and stress and anxiety responses. Pharmacol Biochem Behav 81:331–342
Walker JM, Huang SM, Strangman NM, Tsou K, Sanudo-Pena MC (1999) Pain modulation by release of the endogenous cannabinoid anandamide. Proc Natl Acad Sci USA 96:12198–12203
Wallace TL, Stellitano KE, Neve RL, Duman RS (2004) Effects of cyclic adenosine monophosphate response element binding protein overexpression in the basolateral amygdala on behavioral models of depression and anxiety. Biol Psychiatry 56:151–160
Wright CI, Beijer AVJ, Groenewegen HJ (1996) Basal amygdaloid complex afferents to the rat nucleus accumbens are compartmentally organized. J Neurosci 16:1877–1893
Zangrossi H Jr, File S E (1992) Behavioral consequences in animal tests of anxiety and exploration of exposure to cat odor. Brain Res Bull 29:381–388
Acknowledgments
We would like to thank Dr. Geneviève Bellocq-Latapie (Sanofi-Aventis) for the generous supply of rimonabant. This study was supported by grants from the Estonian Science Foundation (GARFS 6590), the Estonian Ministry of Education and Science (SF0182584Bs03) and the Centre of Molecular and Clinical Medicine, University of Tartu (VARMC-TIPP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sütt, S., Raud, S., Areda, T. et al. Cat odour-induced anxiety—a study of the involvement of the endocannabinoid system. Psychopharmacology 198, 509–520 (2008). https://doi.org/10.1007/s00213-007-0927-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0927-4