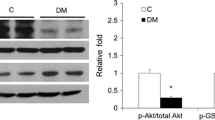
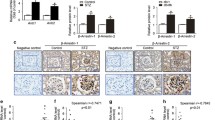
Abstract
Pyrogallol (PG) induces apoptosis in several types of cells mediated by superoxide anion (O •−2 ). Here, we investigated the effects of PG and/or MAPK (MEK, JNK, and p38) inhibitors on the changes in cell growth, cell death, reactive oxygen species (ROS), and GSH levels in As4.1 juxtaglomerular (JG) cells. PG inhibited the growth of As4.1 cells. It also induced apoptosis and the loss of mitochondrial membrane potential (MMP; ΔΨm) and increased the level of p53 protein. Intracellular O •−2 level was increased in PG-treated As4.1 cells. PG also increased the number of GSH deleted cells in As4.1 cells. All the MAPK inhibitors significantly attenuated the growth inhibition and death mediated by PG. They decreased the levels of p53 protein and MMP (ΔΨm) loss in PG-treated As4.1 cells. They also reduced O •−2 level and GSH-depleted cell number in these cells. In conclusion, MAPK inhibitors attenuated As4.1 cell growth inhibition and death mediated by PG treatment. The changes in O •−2 and GSH levels by PG and/or MAPK inhibitors appeared to affect the growth and death of As4.1 cells.







Similar content being viewed by others
Abbreviations
- PG:
-
Pyrogallol
- ROS:
-
Reactive oxygen species
- MAPK:
-
Mitogen-activated protein kinase
- MEK:
-
MAP kinase or ERK kinase
- ERK:
-
Extracellular signal-regulated kinase
- JNK:
-
c-Jun N-terminal kinase
- SOD:
-
Superoxide dismutase
- MMP (ΔΨm):
-
Mitochondrial membrane potential
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- H2DCFDA:
-
2′,7′-Dichlorodihydrofluorescein diacetate
- DHE:
-
Dihydroethidium
- GSH:
-
Glutathione
- CMFDA:
-
5-Chloromethylfluorescein diacetate
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PI:
-
Propidium iodide
References
Baran CP, Zeigler MM, Tridandapani S, Marsh CB (2004) The role of ROS and RNS in regulating life and death of blood monocytes. Curr Pharm Des 10:855–866
Blenis J (1993) Signal transduction via the MAP kinases: proceed at your own RSK. Proc Natl Acad Sci U S A 90:5889–5892
Bubici C, Papa S, Pham CG, Zazzeroni F, Franzoso G (2006) The NF-kappaB-mediated control of ROS and JNK signaling. Histol Histopathol 21:69–80
Chen TJ, Jeng JY, Lin CW, Wu CY, Chen YC (2006) Quercetin inhibition of ROS-dependent and -independent apoptosis in rat glioma C6 cells. Toxicology 223:113–126
Dasmahapatra G, Rahmani M, Dent P, Grant S (2006) The tyrphostin adaphostin interacts synergistically with proteasome inhibitors to induce apoptosis in human leukemia cells through a reactive oxygen species (ROS)-dependent mechanism. Blood 107:232–240
Estrela JM, Ortega A, Obrador E (2006) Glutathione in cancer biology and therapy. Crit Rev Clin Lab Sci 43:143–181
Genestra M (2007) Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell Signal 19:1807–1819
Gomez-Lazaro M et al (2007) Reactive oxygen species and p38 mitogen-activated protein kinase activate Bax to induce mitochondrial cytochrome c release and apoptosis in response to malonate. Mol Pharmacol 71:736–743
Guyton KZ, Liu Y, Gorospe M, Xu Q, Holbrook NJ (1996) Activation of mitogen-activated protein kinase by H2O2. Role in cell survival following oxidant injury. J Biol Chem 271:4138–4142
Han YH, Kim SZ, Kim SH, Park WH (2007) Arsenic trioxide inhibits growth of As4.1 juxtaglomerular cells via cell cycle arrest and caspase-independent apoptosis. Am J Physiol Renal Physiol 293:F511–F520
Han YH, Kim SH, Kim SZ, Park WH (2008a) Caspase inhibitor decreases apoptosis in pyrogallol-treated lung cancer Calu-6 cells via the prevention of GSH depletion. Int J Oncol 33:1099–1105
Han YH, Kim SW, Kim SH, Kim SZ, Park WH (2008b) 2, 4-dinitrophenol induces G1 phase arrest and apoptosis in human pulmonary adenocarcinoma Calu-6 cells. Toxicol In Vitro 22:659–670
Han YH, Kim SZ, Kim SH, Park WH (2008c) Apoptosis in pyrogallol-treated Calu-6 cells is correlated with the changes of intracellular GSH levels rather than ROS levels. Lung Cancer 59:301–314
Han YH, Kim SZ, Kim SH, Park WH (2009) Pyrogallol inhibits the growth of lung cancer Calu-6 cells via caspase-dependent apoptosis. Chem Biol Interact 177:107–114
Henson ES, Gibson SB (2006) Surviving cell death through epidermal growth factor (EGF) signal transduction pathways: implications for cancer therapy. Cell Signal 18:2089–2097
Higuchi Y (2004) Glutathione depletion-induced chromosomal DNA fragmentation associated with apoptosis and necrosis. J Cell Mol Med 8:455–464
Hsin YH, Chen CF, Huang S, Shih TS, Lai PS, Chueh PJ (2008) The apoptotic effect of nanosilver is mediated by a ROS- and JNK-dependent mechanism involving the mitochondrial pathway in NIH3T3 cells. Toxicol Lett 179:130–139
Kusuhara M et al (1998) p38 Kinase is a negative regulator of angiotensin II signal transduction in vascular smooth muscle cells: effects on Na +/H + exchange and ERK1/2. Circ Res 83:824–831
Ling YH, Liebes L, Zou Y, Perez-Soler R (2003) Reactive oxygen species generation and mitochondrial dysfunction in the apoptotic response to Bortezomib, a novel proteasome inhibitor, in human H460 non-small cell lung cancer cells. J Biol Chem 278:33714–33723
Mao X, Yu CR, Li WH, Li WX (2008) Induction of apoptosis by shikonin through a ROS/JNK-mediated process in Bcr/Abl-positive chronic myelogenous leukemia (CML) cells. Cell Res 18:879–888
Moreno-Manzano V, Ishikawa Y, Lucio-Cazana J, Kitamura M (2000) Selective involvement of superoxide anion, but not downstream compounds hydrogen peroxide and peroxynitrite, in tumor necrosis factor-alpha-induced apoptosis of rat mesangial cells. J Biol Chem 275:12684–12691
Nagai H, Noguchi T, Takeda K, Ichijo H (2007) Pathophysiological roles of ASK1-MAP kinase signaling pathways. J Biochem Mol Biol 40:1–6
Park WH et al (2000) Arsenic trioxide-mediated growth inhibition in MC/CAR myeloma cells via cell cycle arrest in association with induction of cyclin-dependent kinase inhibitor, p21, and apoptosis. Cancer Res 60:3065–3071
Park WH, Han YH, Kim SH, Kim SZ (2007a) Pyrogallol, ROS generator inhibits As4.1 juxtaglomerular cells via cell cycle arrest of G2 phase and apoptosis. Toxicology 235:130–139
Park WH, Han YW, Kim SH, Kim SZ (2007b) A superoxide anion generator, pyrogallol induces apoptosis in As4.1 cells through the depletion of intracellular GSH content. Mutat Res 619:81–92
Poot M, Teubert H, Rabinovitch PS, Kavanagh TJ (1995) De novo synthesis of glutathione is required for both entry into and progression through the cell cycle. J Cell Physiol 163:555–560
Poulose SM, Harris ED, Patil BS (2005) Citrus limonoids induce apoptosis in human neuroblastoma cells and have radical scavenging activity. J Nutr 135:870–877
Qiu JH, Asai A, Chi S, Saito N, Hamada H, Kirino T (2000) Proteasome inhibitors induce cytochrome c-caspase-3-like protease-mediated apoptosis in cultured cortical neurons. J Neurosci 20:259–265
Rygiel TP, Mertens AE, Strumane K, van der Kammen R, Collard JG (2008) The Rac activator Tiam1 prevents keratinocyte apoptosis by controlling ROS-mediated ERK phosphorylation. J Cell Sci 121:1183–1192
Saeki K, Hayakawa S, Isemura M, Miyase T (2000) Importance of a pyrogallol-type structure in catechin compounds for apoptosis-inducing activity. Phytochemistry 53:391–394
Sawada M et al (2001) p53 regulates ceramide formation by neutral sphingomyelinase through reactive oxygen species in human glioma cells. Oncogene 20:1368–1378
Schnelldorfer T, Gansauge S, Gansauge F, Schlosser S, Beger HG, Nussler AK (2000) Glutathione depletion causes cell growth inhibition and enhanced apoptosis in pancreatic cancer cells. Cancer 89:1440–1447
Sigmund CD et al (1990) Isolation and characterization of renin-expressing cell lines from transgenic mice containing a renin-promoter viral oncogene fusion construct. J Biol Chem 265:19916–19922
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5:415–418
Wallach-Dayan SB, Izbicki G, Cohen PY, Gerstl-Golan R, Fine A, Breuer R (2006) Bleomycin initiates apoptosis of lung epithelial cells by ROS but not by Fas/FasL pathway. Am J Physiol Lung Cell Mol Physiol 290:L790–L796
Wilcox CS (2002) Reactive oxygen species: roles in blood pressure and kidney function. Curr Hypertens Rep 4:160–166
Yamada J et al (2003) Cell permeable ROS scavengers, Tiron and Tempol, rescue PC12 cell death caused by pyrogallol or hypoxia/reoxygenation. Neurosci Res 45:1–8
Yang J et al (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275:1129–1132
Zelko IN, Mariani TJ, Folz RJ (2002) Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic Biol Med 33:337–349
Zorov DB, Juhaszova M, Sollott SJ (2006) Mitochondrial ROS-induced ROS release: An update and review. Biochim Biophys Acta 1757:509–517
Acknowledgments
This research was supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare & Family Affairs and Republic of Korea (A084194).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, Y.H., Park, W.H. Pyrogallol-induced As4.1 juxtaglomerular cell death is attenuated by MAPK inhibitors via preventing GSH depletion. Arch Toxicol 84, 631–640 (2010). https://doi.org/10.1007/s00204-010-0526-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-010-0526-8