Abstract
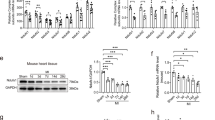
Ischemia impairs the adenine nucleotide translocase (ANT), which transports ADP and ATP across the inner mitochondrial membrane. We investigated whether ANT1 overexpression has protective effects on ischemic hearts. Myocardial infarction was induced in wild-type (WT) and heart-specific ANT1-transgenic (ANT1-TG) rats, and hypoxia was set in isolated cardiomyocytes. ANT1 overexpression reduced the myocardial infarct area and increased the survival rate of infarcted rats. Reduced ANT1 expression and increased 4-hydroxynonenal modification of ANT paralleled to impaired ANT function in infarcted WT hearts. ANT1 overexpression improved ANT expression and function. This was accompanied by reduced mitochondrial cytochrome C release and caspase-3 activation. ANT1-TG hearts suffered less from oxidative stress, as shown by lower protein carbonylation and 4-hydroxynonenal modification of ANT. ANT1 overexpression also increased cell survival of hypoxic cardiomyocytes and attenuated reactive oxygen species (ROS) production. This was linked to higher stability of mitochondrial membrane potential and lower activity of ROS detoxifying catalase. ANT1-TG cardiomyocytes also showed higher resistance against H2O2 treatment, which was independent of catalase activity. In conclusion, ANT1 overexpression compensates impaired ANT activity under oxygen-restricted conditions. It reduces ROS production and oxidative stress, stabilizes mitochondrial integrity, and increases survival, making ANT1 a component in ROS management and heart protection during ischemia.
Key messages
-
ANT1 overexpression reduces infarct size and increases survival after infarction.
-
ANT1 overexpression compensates restricted ANT expression and function in infarcted hearts.
-
Increased ANT1 expression enhances mitochondrial integrity.
-
ANT1-overexpressing hearts reduce oxidative stress by decreasing ROS generation.
-
ANT1 is a component in ROS management and heart protection.






Similar content being viewed by others
References
Klingenberg M (2008) The ADP and ATP transport in mitochondria and its carrier. Biochim Biophys Acta 1778:1978–2021
Halestrap AP, Richardson AP (2015) The mitochondrial permeability transition: a current perspective on its identity and role in ischaemia/reperfusion injury. J Mol Cell Cardiol 78:129–141
Dorner A, Olesch M, Giessen S, Pauschinger M, Schultheiss HP (1999) Transcription of the adenine nucleotide translocase isoforms in various types of tissues in the rat. Biochim Biophys Acta 1417:16–24
Duan J, Karmazyn M (1989) Relationship between oxidative phosphorylation and adenine nucleotide translocase activity of two populations of cardiac mitochondria and mechanical recovery of ischemic hearts following reperfusion. Can J Physiol Pharmacol 67:704–709
Nickel A, Kohlhaas M, Maack C (2014) Mitochondrial reactive oxygen species production and elimination. J Mol Cell Cardiol 73:26–33
Borutaite V, Mildaziene V, Katiliute Z, Kholodenko B, Toleikis A (1993) The function of ATP/ADP translocator in the regulation of mitochondrial respiration during development of heart ischemic injury. Biochim Biophys Acta 1142:175–180
Chen JJ, Bertrand H, Yu BP (1995) Inhibition of adenine nucleotide translocator by lipid peroxidation products. Free Radic Biol Med 19:583–590
Zwizinski CW, Schmid HH (1992) Peroxidative damage to cardiac mitochondria: identification and purification of modified adenine nucleotide translocase. Arch Biochem Biophys 294:178–183
Bittar N, Shug AL, Folts JD, Koke JR (1976) Ischemic-like change produced in canine heart by atractyloside: a chemical model. Recent Adv Stud Cardiac Struct Metab 12:167–174
Esposito LA, Melov S, Panov A, Cottrell BA, Wallace DC (1999) Mitochondrial disease in mouse results in increased oxidative stress. Proc Natl Acad Sci U S A 96:4820–4825
Graham BH, Waymire KG, Cottrell B, Trounce IA, MacGregor GR, Wallace DC (1997) A mouse model for mitochondrial myopathy and cardiomyopathy resulting from a deficiency in the heart/muscle isoform of the adenine nucleotide translocator. Nat Genet 16:226–234
Walther T, Tschope C, Sterner-Kock A, Westermann D, Heringer-Walther S, Riad A, Nikolic A, Wang Y, Ebermann L, Siems WE et al (2007) Accelerated mitochondrial adenosine diphosphate/adenosine triphosphate transport improves hypertension-induced heart disease. Circulation 115:333–344
Koch M, Spillmann F, Dendorfer A, Westermann D, Altmann C, Sahabi M et al (2006) Cardiac function and remodeling is attenuated in transgenic rats expressing the human kallikrein-1 gene after myocardial infarction. Eur J Pharmacol 550:143–148
Koch M, Savvatis K, Scheeler M, Dhayat S, Bonaventura K, Pohl T et al (2010) Immunosuppression with an interleukin-2 fusion protein leads to improved LV function in experimental ischemic cardiomyopathy. Int Immunopharmacol 10:207–212
Smith AL (1967) Preparation, properties and conditions for assay of mitochondria: slaughterhouse material, small scale. Methods Enzymol 10:81–86
Vyssokikh MY, Katz A, Rueck A, Wuensch C, Dorner A, Zorov DB et al (2001) Adenine nucleotide translocator isoforms 1 and 2 are differently distributed in the mitochondrial inner membrane and have distinct affinities to cyclophilin D. Biochem J 358:349–358
Winter J, Klumpe I, Heger J, Rauch U, Schultheiss HP, Landmesser U et al (2016) Adenine nucleotide translocase 1 overexpression protects cardiomyocytes against hypoxia via increased ERK1/2 and AKT activation. Cell Signal 28:152–159
Kluck RM, Martin SJ, Hoffman BM, Zhou JS, Green DR, Newmeyer DD (1997) Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J 16:4639–4649
Choksi KB, Boylston WH, Rabek JP, Widger WR, Papaconstantinou J (2004) Oxidatively damaged proteins of heart mitochondrial electron transport complexes. Biochim Biophys Acta 1688:95–101
Halestrap AP, McStay GP, Clarke SJ (2002) The permeability transition pore complex: another view. Biochimie 84:153–166
Pell S, Fayerweather WE (1985) Trends in the incidence of myocardial infarction and in associated mortality and morbidity in a large employed population, 1957–1983. N Engl J Med 312:1005–1011
Brown DA, O’Rourke B (2010) Cardiac mitochondria and arrhythmias. Cardiovasc Res 88:241–249
Chevrollier A, Loiseau D, Gautier F, Malthiery Y, Stepien G (2005) ANT2 expression under hypoxic conditions produces opposite cell-cycle behavior in 143B and HepG2 cancer cells. Mol Carcinog 42:1–8
Eaton P, Li JM, Hearse DJ, Shattock MJ (1999) Formation of 4-hydroxy-2-nonenal-modified proteins in ischemic rat heart. Am J Physiol 276:H935–H943
Tsutsui H, Ide T, Kinugawa S (2006) Mitochondrial oxidative stress, DNA damage, and heart failure. Antioxid Redox Signal 8:1737–1744
Vieira HL, Belzacq AS, Haouzi D, Bernassola F, Cohen I, Jacotot E et al (2001) The adenine nucleotide translocator: a target of nitric oxide, peroxynitrite, and 4-hydroxynonenal. Oncogene 20:4305–4316
Belzacq AS, Vieira HL, Kroemer G, Brenner C (2002) The adenine nucleotide translocator in apoptosis. Biochimie 84:167–176
Narula J, Pandey P, Arbustini E, Haider N, Narula N, Kolodgie FD et al (1999) Apoptosis in heart failure: release of cytochrome c from mitochondria and activation of caspase-3 in human cardiomyopathy. Proc Natl Acad Sci U S A 96:8144–8149
Heger J, Abdallah Y, Shahzad T, Klumpe I, Piper HM, Schultheiss HP et al (2012) Transgenic overexpression of the adenine nucleotide translocase 1 protects cardiomyocytes against TGFbeta(1)-induced apoptosis by stabilization of the mitochondrial permeability transition pore. J Mol Cell Cardiol 53:73–81
Chinnaiyan AM (1999) The apoptosome: heart and soul of the cell death machine. Neoplasia 1:5–15
Pasdois P, Parker JE, Griffiths EJ, Halestrap AP (2011) The role of oxidized cytochrome c in regulating mitochondrial reactive oxygen species production and its perturbation in ischaemia. Biochem J 436:493–505
Forman HJ, Torres M (2001) Redox signaling in macrophages. Mol Aspects Med 22:189–216
Jaganjac M, Cipak A, Schaur RJ, Zarkovic N (2016) Pathophysiology of neutrophil-mediated extracellular redox reactions. Front Biosci (Landmark Ed) 21:839–55
Acknowledgements
This work was supported by the “Deutsche Forschungsgemeinschaft (DFG)” through the Sonderforschungsbereich Transregio 19, projects C7, C3, Z3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests related to this study.
Additional information
Inga Klumpe and Konstantinos Savvatis contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 233 kb)
Rights and permissions
About this article
Cite this article
Klumpe, I., Savvatis, K., Westermann, D. et al. Transgenic overexpression of adenine nucleotide translocase 1 protects ischemic hearts against oxidative stress. J Mol Med 94, 645–653 (2016). https://doi.org/10.1007/s00109-016-1413-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-016-1413-4