Abstract
Raloxifene, a selective estrogen receptor modulator, is approved for the prevention and treatment of postmenopausal osteoporosis. Prevention studies with raloxifene have demonstrated preservation of bone density and suppression of bone turnover markers in young postmenopausal women. The Multiple Outcomes of Raloxifene Evaluation study was the pivotal treatment trial for raloxifene. It demonstrated significant reduction in the risk for vertebral fractures after 1 and 3 years. Significant reduction of nonvertebral fractures with raloxifene has not yet been demonstrated. In addition to the effects of raloxifene on bone, potentially beneficial effects on the cardiovascular system, breast, and uterus have been described. Most of these nonskeletal effects have been reported as secondary endpoints from large osteoporosis trials with raloxifene. Prospective, randomized, double-blind studies of raloxifene with breast cancer prevention and cardiovascular protection as primary endpoints are now underway.
Similar content being viewed by others
References and Recommended Reading
Osborne CK, Zhao H, Fuqua SA: Selective estrogen receptor modulators. Structure, function and clinical use. J Clin Oncol 2000, 18:3172–3186.
Riggs BL, Hartmann LC: Selective estrogen-receptor modulators-mechanisms of action and application to clinical practice. N Engl J Med 2003, 348:618–629.
Kuiper JGM, Carlsson B, Grandien K, et al.: Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology 1997, 138:863–870.
Bryant HU: Mechanism of action and preclinical profile of raloxifene, a selective estrogen receptor modulator. Rev Endocr Metab Disord 2001, 2:129–138.
Wijayarantne AL, Nagel SC, Paige LA, et al.: Comparative analysis of mechanistic differences among antiestrogens. Endocrinology 1999, 140:5828–5840.
Shang Y, Brown M: Molecular determinants for the tissue specificity of SERMs. Science 2002, 295:2465–2468.
Chen Z, Yuhanna IS, Galcheva-Gargova Z, et al.: Estrogen receptor alpha mediates the nongenomic activation of endothelial nitric oxide synthase by estrogen. J Clin Invest 1999, 103:401–406.
Delmas PD, Bjarnason NH, Mitlak BH, et al.: Effects of raloxifene on bone mineral density, serum cholesterol concentration and uterine endometrium in postmenopausal women. N Engl J Med 1997, 337:1641–1647.
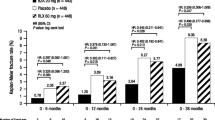
Ettinger B, Black DM, Mitlak BH, et al.: Reduction of vertebral risk in postmenopausal women with osteoporosis treated with raloxifene. Results from a 3-year randomized clinical trial. JAMA 1999, 282:637–645. This trial was pivotal in demonstrating the reduction in the risk for osteoporotic fractures with raloxifene.
Genant HK, Wu CY, Van Kuijk C, Nevitt MC: Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 1993, 8:1137–1148.
Bjarnason NH, Sarkar S, Duong T, et al.: Six and twelve month changes in bone turnover are related to reduction in vertebral fracture risk during 3 years of raloxifene treatment in post-menopausal osteoporosis. Osteoporos Int 2001, 12:922–930. Early changes in biochemical markers of bone turnover at 6 and 12 months with raloxifene predict vertebral fracture reduction at 36 months.
Sarkar S, Mitlak BH, Wong M, et al.: Relationships between bone mineral density and vertebral fracture risk with raloxifene therapy. J Bone Miner Res 2002, 17:1–10. Changes in bone mineral density correlate poorly with raloxifene’s ability to reduce vertebral fracture. Only 4% of the observed fracture reduction with raloxifene can be explained by increases in bone mineral density.
Looker AC, Wahner HW, Dunn WL, et al.: Updated data on proximal femur bone mineral levels of US adults. Osteoporos Int 1998, 8:468–489.
Johnell O, Kanis J, Black D, et al.: Raloxifene significantly reduces vertebral fracture risk in patients with osteopenia or osteoporosis defined by total hip BMD T-score. Osteoporos Int 2002, 13:S69-S70.
Maricic M, Adachi J, Meunier P, et al.: Early effects of raloxifene on clinical vertebral fractures at 12 months in postmenopausal women with osteoporosis. Arch Int Med 2002, 162:1140–1143.
Delmas PD, Ensrud KE, Adachi JD, et al.: Efficacy of raloxifene on vertebral fracture risk reduction in postmenopausal women with osteoporosis: four-year results from a randomized clinical trial. J Clin Endocrinol Metab 2002, 87:3609–3617.
Cranney A, Tugwell P, Zytaruk N, et al.: Meta-analysis of raloxifene for the prevention and treatment of postmenopausal osteoporosis. Endocr Rev 2002, 23:524–528.
Silverman SL, Minshall ME, Shen W, et al.: The relationship of health-related quality of life to prevalent and incident vertebral fractures in postmenopausal women with osteoporosis. Arthritis Rheum 2001, 44:2611–2619.
Johnell O, Scheele WH, Lu Y, et al.: Additive effects of raloxifene and alendronate on bone density and biochemical markers of bone remodeling in postmenopausal women with osteoporosis. J Clin Endocrinol Metab 2002, 87:985–992.
Neele SJM, Evertz R, De Valk-de Roo G, et al.: Effect of 1 year discontinuation of raloxifene or estrogen therapy on bone mineral density after 5 years of treatment in healthy postmenopausal women. Bone 2002, 30:599–603.
Jackson VP, San Martin JA, Secrest RJ, et al.: Comparison of the effect of raloxifene and continuous-combined hormone therapy on mammographic breast density and breast tenderness in postmenopausal women. Am J Obstet Gynecol 2003, 188:389–395.
Cummings S, Eckert S, Krueper K, et al.: The effect of raloxifene or risk of breast cancer in post-menopausal women. Results from the MORE randomized trial. JAMA 1999, 281:2189–2197.
Cauley JA, Norton L, Lippman ME, et al.: Continued breast cancer risk reduction in postmenopausal women treated with raloxifene: 4 year results from the MORE trial. Breast Cancer Res Treat 2001, 65:125–134.
Cummings SR, Duong T, Kenyon E, et al.: Estradiol level and risk of breast cancer during treatment with raloxifene. JAMA 2002, 287:216–220. Raloxifene significantly reduced the risk of breast cancer in women with high and low estradiol levels in the MORE trial.
Wickerham DL, Tan-Chiu E: Breast cancer chemoprevention: current status and future directions. Semin Oncol 2001, 28:253–259.
Walsh BW, Kuller LH, Wild RA, et al.: Effects of raloxifene on serum lipids and coagulation factors in healthy postmenopausal women. JAMA 1998, 279:1445–1451.
Walsh BW, Paul S, Wild RA, et al.: The effects of hormone replacement therapy and raloxifene on C-reactive protein and homocysteine in healthy postmenopausal women: a randomized, controlled trial. J Clin Endocrinol Metab 2000, 85:214–218.
Barrett-Conner E, Grady D, Sashegui A, et al.: Raloxifene and cardiovascular events in osteoporotic postmenopausal women. JAMA 2002, 287:847–856. In the MORE trial, raloxifene improved cardiovascular outcomes in women at highest risk.
Wenger NK, Barrett-Connor E, Collins P, et al.: Baseline characteristics of participants in the Raloxifene Use for The Heart (RUTH) trial. Am J Cardiol 2002, 90:1204–1210.
Davies GC, Huster WJ, Shen W, et al.: Endometrial response to raloxifene compared with placebo, cyclical hormone replacement therapy, and unopposed estrogen in postmenopausal women. Menopause 1999, 6:188–195.
Goldstein SR, Neven P, Zhou L, et al.: Raloxifene effect on frequency of surgery for pelvic floor relaxation. Obstet Gynecol 2001, 98:91–96.
Palomba S, Sammartino A, Dicarlo C, et al.: Effects of raloxifene treatment on uterine leiomyomas in postmenopausal women. Fertil Steril 2001, 76:38–43.
Yaffe K, Krueger K, Sarkar S, et al.: Multiple outcomes of raloxifene evaluation investigators. Cognitive function in postmenopausal women treated with raloxifene. N Engl J Med 2001, 344:1207–1213.
American College of Obsterticians and Gynecologists Committee on Practice Bulletins-Gynecology: ACOG practice bulletin. Selective estrogen receptor modulators. Int J Gynaecol Obstet 2002, 79:289–298.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gluck, O., Maricic, M. Skeletal and nonskeletal effects of raloxifene. Curr Osteoporos Rep 1, 123–128 (2003). https://doi.org/10.1007/s11914-996-0007-4
Issue Date:
DOI: https://doi.org/10.1007/s11914-996-0007-4