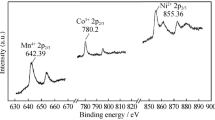
Abstract
Recycling of valuable metals from spent lithium-ion batteries (LIBs) appears inevitable for both environmental protection and resource recovery. In the present study, an efficient hydrometallurgical leaching of Co and Li from cathode materials of spent LIBs using a citric acid/sodium thiosulfate (Na2S2O3) system is explored. The effects of citric acid and Na2S2O3 concentrations, leaching time, temperature, and the solid/liquid (S/L) ratio on the leaching processes are also examined. With the exception of the S/L ratio, the increase of citric acid concentration, Na2S2O3 concentration, leaching time, and temperature all have positive effects on the leaching of Co and Li. Ultimately, approximately 96% of Co and 99% of Li are recycled from the spent LIBs in this citric acid/sodium thiosulfate system under the leaching conditions of an S/L ratio of 20 g l−1, concentration of Na2S2O3 of 0.3 M, concentration of citric acid of 1.2 M, leaching time of 30 min, and leaching temperature of 70°C. The Avrami equation is well fitted by the data of the leaching processes, and model equations are built to describe the leaching of Co and Li. Furthermore, pure sulfur can be obtained as a by-product during the leaching process, and SO2 produced during the reaction is easily collected as a raw material for industrial production of sulfuric acid. The present study represents a promising process for hydrometallurgical recovery of valuable metals from spent LIBs.





Similar content being viewed by others
References
B. Scrosati, J. Hassoun, and Y.K. Sun, Energy Environ. Sci. 4, 3287 (2011).
J. Ordoñez, E.J. Gago, and A. Girard, Renew. Sustain. Energy Rev. 60, 195 (2016).
G.J. Wang, H.P. Zhang, L.J. Fu, B. Wang, and Y.P. Wu, Electrochem. Commun. 9, 1873 (2007).
R.C. Wang, Y.C. Lin, and S.H. Wu, Hydrometallurgy 99, 194 (2009).
Y. Guo, F. Li, H. Zhu, G. Li, J. Huang, and W. He, Waste Manag. 51, 227 (2016).
L. Li, W. Qu, X. Zhang, J. Lu, R. Chen, F. Wu, and K. Amine, J. Power Sources 282, 544 (2015).
H. Ku, Y. Jung, M. Jo, S. Park, S. Kim, D. Yang, K. Rhee, E.M. An, J. Sohn, and K. Kwon, J. Hazard. Mater. 313, 138 (2016).
X. Zeng, J. Li, and N. Singh, Crit. Rev. Environ. Sci. Technol. 44, 1129 (2014).
E. Gratz, Q. Sa, D. Apelian, and Y. Wang, J. Power Sources 262, 255 (2014).
B. Kirubasankar, V. Murugadoss, J. Lin, T. Ding, M. Dong, H. Liu, J. Zhang, T. Li, N. Wang, Z. Guo, and S. Angaiah, Nanoscale 10, 20414 (2018).
Z. Qu, M. Shi, H. Wu, Y. Liu, J. Jiang, and C. Yan, J. Power Sources 410–411, 179 (2019).
K.S. Sangwan and A. Jindal, Int. J. Sustain. Eng. 6, 359 (2013).
X. Zhang, Y. Xie, X. Lin, H. Li, and H. Cao, J. Mater. Cycles Waste Manag. 15, 420 (2013).
S.A. Mazen and N.I. Abu-Elsaad, Appl. Nanosci. 5, 105 (2015).
D. Song, X. Wang, E. Zhou, P. Hou, F. Guo, and L. Zhang, J. Power Sources 232, 348 (2013).
J. Li, G. Wang, and Z. Xu, J. Hazard. Mater. 302, 97 (2016).
G.P. Nayaka, K.V. Pai, G. Santhosh, and J. Manjanna, Hydrometallurgy 161, 54 (2016).
L. Yao, H. Yao, G. Xi, and Y. Feng, RSC Adv. 6, 17947 (2016).
K. Liu and F.S. Zhang, J. Hazard. Mater. 316, 19 (2016).
Z. Niu, Y. Zou, B. Xin, S. Chen, C. Liu, and Y. Li, Chemosphere 109, 92 (2014).
N.B. Horeh, S.M. Mousavi, and S.A. Shojaosadati, J. Power Sources 320, 257 (2016).
Y. Xin, X. Guo, S. Chen, J. Wang, F. Wu, and B. Xin, J. Clean. Prod. 116, 249 (2016).
D.S. Kim, J.S. Sohn, C.K. Lee, J.H. Lee, K.S. Han, and Y.I. Lee, J. Power Sources 132, 145 (2004).
J. Xu, H.R. Thomas, R.W. Francis, K.R. Lum, J. Wang, and B. Liang, J. Power Sources 177, 512 (2008).
L. Zhao, D. Yang, and N.W. Zhu, J. Hazard. Mater. 160, 648 (2008).
L. Li, J. Ge, R. Chen, F. Wu, S. Chen, and X. Zhang, Waste Manag. 30, 2615 (2010).
L. Li, J. Lu, Y. Ren, X.X. Zhang, R.J. Chen, F. Wu, and K. Amine, J. Power Sources 218, 21 (2012).
F. Pagnanelli, E. Moscardini, P. Altimari, T. Abo Atia, and L. Toro, Waste Manag. 51, 214 (2016).
R. Golmohammadzadeh, F. Rashchi, and E. Vahidi, Waste Manag. 64, 244 (2017).
J. Kang, G. Senanayake, J. Sohn, and S.M. Shin, Hydrometallurgy 100, 168 (2010).
A. Chagnes and B. Pospiech, J. Chem. Technol. Biotechnol. 88, 1191 (2013).
F. Pagnanelli, E. Moscardini, G. Granata, S. Cerbelli, L. Agosta, A. Fieramosca, and L. Toro, J. Ind. Eng. Chem. 20, 3201 (2014).
M. Joulié, E. Billy, R. Laucournet, and D. Meyer, Hydrometallurgy 169, 426 (2017).
L. Li, J.B. Dunn, X.X. Zhang, L. Gaines, R.J. Chen, F. Wu, and K. Amine, J. Power Sources 233, 180 (2013).
L. Li, J. Ge, F. Wu, R. Chen, S. Chen, and B. Wu, J. Hazard. Mater. 176, 288 (2010).
X. Zeng, J. Li, and B. Shen, J. Hazard. Mater. 295, 112 (2015).
X. Chen, Y. Chen, T. Zhou, D. Liu, H. Hu, and S. Fan, Waste Manag. 38, 349 (2015).
G.P. Nayaka, J. Manjanna, K.V. Pai, R. Vadavi, S.J. Keny, and V.S. Tripathi, Hydrometallurgy 151, 73 (2015).
J. Wang, M. Chen, H. Chen, T. Luo, and Z. Xu, Proced. Environ. Sci. 16, 443 (2012).
J. Guan, Y. Li, Y. Guo, R. Su, G. Gao, H. Song, H. Yuan, B. Liang, and Z. Guo, ACS Sustain. Chem. Eng. 5, 1026 (2017).
E. Billy, M. Joulié, R. Laucournet, A. Boulineau, E. De Vito, and D. Meyer, ACS Appl. Mater. Interfaces 10, 16424 (2018).
P. Ashtari and P. Pourghahramani, J. Mater. Cycles Waste Manag. 20, 155 (2018).
P. Meshram, B.D. Pandey, and T.R. Mankhand, Waste Manag. 45, 306 (2015).
E.G. Pinna, M.C. Ruiz, M.W. Ojeda, and M.H. Rodriguez, Hydrometallurgy 167, 66 (2017).
R. Golmohammadzadeh, F. Faraji, and F. Rashchi, Resour. Conserv. Recycl. 136, 418 (2018).
S. Sakultung, K. Pruksathorn, and M. Hunsom, Korean J. Chem. Eng. 24, 272 (2007).
B. Fan, X. Chen, T. Zhou, J. Zhang, and B. Xu, Waste Manag. Res. 34, 474 (2016).
C.K. Lee and K.I. Rhee, Hydrometallurgy 68, 5 (2003).
M. Joulié, R. Laucournet, and E. Billy, J. Power Sources 247, 551 (2014).
R. Stoyanova, E. Zhecheva, and L. Zarkova, Solid State Ion. 73, 233 (1994).
B. Swain, J. Jeong, J.C. Lee, G.H. Lee, and J.S. Sohn, J. Power Sources 167, 536 (2007).
L. Chen, X. Tang, Y. Zhang, L. Li, Z. Zeng, and Y. Zhang, Hydrometallurgy 108, 80 (2011).
Acknowledgements
We gratefully acknowledge the financial support from Gaoyuan Discipline of Shanghai–Environmental Science and Engineering (Resource Recycling Science and Engineering), Shanghai “Chenguang” Program (15CG60), Shanghai Sailing Program (18YF1429900, 15YF1404300), Shanghai Natural Science Foundation (15ZR1416800), Natural Science Foundation of China (51678353), Cultivation discipline fund of Shanghai Polytechnic University (XXKPY1601), Shanghai Polytechnic University Leap Program (EGD19XQD02, EGD18XQD24), and Henan Key Laboratory of Coal Green Conversion (Henan Polytechnic University, CGCF201803).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gao, G., He, X., Lou, X. et al. A Citric Acid/Na2S2O3 System for the Efficient Leaching of Valuable Metals from Spent Lithium-Ion Batteries. JOM 71, 3673–3681 (2019). https://doi.org/10.1007/s11837-019-03629-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-019-03629-y