Abstract
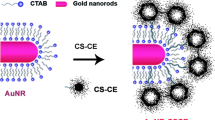
Gold nanorod (GNR) has great potential in the field of cancer therapy because of its photophysical property in converting near-infrared (NIR) laser light into heat. Fabrication of GNRs by seed-mediated growth method with the aid of cetyltrimethylammonium bromide (CTAB) is a popular approach. However, due to high cytotoxicity of CTAB, it is necessary to modify the surface of CTAB-passivated GNRs for cell-related studies. In this study, thiolated chitosan was synthesized and harnessed to replace CTAB originally used to stabilize GNRs. The average size and morphological shape of CTAB-passivated GNRs (66.0 nm) and thiolated chitosan-modified GNRs (CGNRs) (84.9 nm) were determined by dynamic light scattering and transmission electron microscopy. X-ray photoelectron spectroscopy was used to confirm the existence of Au–S binding energy at 162.4 eV. Cytotoxicity study revealed that CGNRs were much biocompatible than CTAB-stabilized GNRs. Our results showed that CGNRs functionalized with folic acid (FA) could be internalized by human colon HT-29 cancer cells via folate-mediated endocytosis. From the viability of CGNR-laden HT-29 cells irradiated with 808-nm NIR laser light, we demonstrated that CGNR is a potential photothermal nano-absorber for the ablation of malignant cells under NIR laser exposure.









Similar content being viewed by others
References
Alkilany AM, Nagaria PK, Hexel CR, Shaw TJ, Murphy CJ, Wyatt MD (2009) Cellular uptake and cytotoxicity of gold nanorods: molecular origin of cytotoxicity and surface effects. Small 5:701–708
Au L, Zheng D, Zhou F, Li ZY, Li X, Xia Y (2008) A quantitative study on the photothermal effect of immune gold nanocages targeted to breast cancer cells. ACS Nano 2:1645–1652
Bernkop-Schnurch A, Hornof M, Guggi D (2004) Thiolated chitosans. Eur J Pharm Biopharm 57:9–17
Bhattarai SR, Kc RB, Kim SY, Sharma M, Khil MS, Hwang PH, Chung GH, Kim HY (2008) N-hexanoyl chitosan stabilized magnetic nanoparticles: implication for cellular labeling and magnetic resonance imaging. J Nanobiotechnol 6:1
Cathell MD, Szewczyk JC, Bui FA, Weber CA, Wolever JD, Kang J, Schauer CL (2008) Structurally colored thiol chitosan thin films as a platform for aqueous heavy metal ion detection. Biomacromolecules 9:289–295
Chang KLB, Tai MC, Cheng FH (2001) Kinetics and products of the degradation of chitosan by hydrogen peroxide. J Agric Food Chem 49:4845–4851
Chen J, Glaus C, Laforest R, Zhang Q, Yang M, Gidding M, Welch MJ, Xia Y (2010) Gold nanocages as photothermal transducers for cancer treatment. Small 6:811–817
Cho JH, Kim SH, Park KD, Jung MC, Yang WI, Han SW, Noh JY, Lee JW (2004) Chondrogenic differentiation of human mesenchymal stem cells using a thermosensitive poly(N-isopropylacrylamide) and water-soluble chitosan copolymer. Biomaterials 25:5743–5751
Connor EE, Mwamuka J, Cole A, Murphy CJ, Wyatt MD (2005) Gold nanoparticles are taken up by human cells but do not cause acute cytotoxicity. Small 1:325–327
Dai Q, Coutts J, Zou J, Huo Q (2008) Surface modification of gold nanorods through a place exchange reaction inside an ionic exchange resin. Chem Commun 25:2858–2860
Ellman GL (1958) A colorimetric method for determining low concentrations of mercaptans. Arch Biochem Biophys 74:443–450
Felt O, Buri P, Gurny R (1998) Chitosan: a unique polysaccharide for drug delivery. Drug Dev Ind Pharm 24:979–993
Foss CA, Hornyak GL, Stockert JA, Martin CR (1994) Template-synthesized nanoscopic gold particles: optical spectra and the effects of particle size and shape. J Phys Chem 98:2963–2971
Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Price RE, Hazle JD, Halas NJ, West JL (2003) Nanoshell-mediated near infrared thermal therapy of tumors under magnetic resonance guidance. Proc Natl Acad Sci USA 100:13549–13554
Huang X, El-Sayed IH, Qian W, El-Sayed MA (2006) Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J Am Chem Soc 128:2115–2120
Huang X, Jain PK, El-Sayed IH, El-Sayed MA (2007) Gold nanoparticles: interesting optical properties and recent applications in cancer diagnostics and therapy. Nanomedicine 2:81–693
Huff TB, Hansen MN, Zhao Y, Cheng JX, Wei A (2007) Controlling the cellular uptake of gold nanorods. Langmuir 23:1596–1599
Jana NR, Gearheart L, Murphy CJ (2001) Wet chemical synthesis of high aspect ratio cylindrical gold nanorods. J Phys Chem B 105:4065–4067
Kam NW, O’Connell M, Wisdom JA, Dai H (2005) Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc Natl Acad Sci USA 102:11600–11605
Liao H, Hafner LH (2005) Gold nanorod bioconjugates. Chem Mater 17:4636–4641
Link S, El-Sayed MA (2000) Shape and size dependence of radiative, nonradiative, and photothermal properties of gold nanocrystals. Int Rev Phys Chem 19:409–433
Liu XF, Guan YL, Yang DZ, Li Z, Yao KD (2001) Antibacterial action of chitosan and carboxymethylated chitosan. J Appl Polym Sci 79:1324–1335
Loo C, Lowery A, Halas NJ, West JL, Drezek R (2005) Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett 5:709–711
Niidome T, Yamagata M, Okamoto Y, Akiyama Y, Takahashi H, Kawano T, Katayama Y, Niidome Y (2006) PEG-modified gold nanorods with a stealth character for in vivo applications. J Control Release 114:343–347
Nikoobakht B, El-Sayed MA (2001) Evidence for bilayer assembly of cationic surfactants on the surface of gold nanorods. Langmuir 17:6368–6374
Orendorff CJ, Murphy CJ (2006) Quantitation of metal content in the silver-assisted growth of gold nanorods. J Phys Chem B 110:3990–3994
Satake K, Okuyama T, Ohashi M, Shinoda T (1960) The spectrophotometric determination of amine, amino acid and peptide with 2,4,6-trinitrobenzene 1-sulfonic acid. J Biochem 47:654–660
Takahashi H, Niidome Y, Niidome T, Kaneko K, Kawasaki H, Yamada S (2006) Modification of gold nanorods using phosphatedylcholine to reduce cytotoxicity. Langmuir 22:2–5
Tong L, Zhao Y, Huff TB, Hansen MN, Wei A, Cheng JX (2007) Gold nanorods mediate tumor cell death by compromising membrane integrity. Adv Mater 19:3136–3141
Wang CH, Huang YJ, Chang CW, Hsu WM, Peng CA (2009) In vitro photothermal destruction of neuroblastoma cells using carbon nanotubes conjugated with GD2 monoclonal antibody. Nanotechnology 20:315101–315107
Wang CH, Chiou SH, Chou CP, Chen YC, Huang YJ, Peng CA (2010) Photothermolysis of glioblastoma stem-like cells targeted by carbon nanotubes conjugated with CD133 monoclonal antibody. Nanomedicine. doi:10.1016/j.nano.2010.06.010
Acknowledgments
This study was funded by Taiwan National Science Council grant (NSC96-2628-E-002-013-MY3) and Michigan Tech Fund of Michigan Technological University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Chung-Hao Wang and Chia-Wei Chang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, CH., Chang, CW. & Peng, CA. Gold nanorod stabilized by thiolated chitosan as photothermal absorber for cancer cell treatment. J Nanopart Res 13, 2749–2758 (2011). https://doi.org/10.1007/s11051-010-0162-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-010-0162-5