Abstract
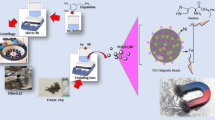
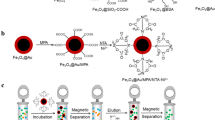
The aim of this study was to develop a simple, cheap, and rapid method for purification of His-tag recombinant proteins with high yields. The new immobilized metal ion affinity adsorbent containing superparamagnetic nanoparticles and hydrophilic resins are proposed here to improve the purification of His-tagged recombinant proteins. In this report, we have described the preparation of nanosized superparamagnetic nanoparticles (Fe3O4) which were prepared by chemical precipitation method followed by surface modification using phosphonomethyl iminodiacetic acid. The stable surface functionalized nanoparticles were further linked with Ni2+ for purification of 6× His-tagged proteins. The phosphonate group of the N-phosphonomethyl iminodiacetic acid ligand acts as a surface anchoring agent on magnetite nanoparticles and the remaining free –COOH groups outside for binding with Ni2+ ions. The nanoparticles were approximately 6–8 nm in size and were stable and had negligible non-specific binding for protein. The proteins were purified within 1 h and observed on sodium dodecyl sulfate-polyacrylamide electrophoresis gel.








Similar content being viewed by others
References
Bele M, Hribar G, Campelj S, Makovec D, Porekar VG, Zorko M, Gaberscek M, Jamnik J, Venturini P (2008) Zinc-decorated silica-coated magnetic nanoparticles for protein binding and controlled release. J Chromatogr B 867:160–164
Cheng J, Teply BA, Jeong SY, Yim CH, Ho D, Sherifi I, Jon S, Farokhzad OC, Khademhosseini A, Langer RS (2006) Magnetically responsive polymeric microparticles for oral delivery of protein drugs. Pharm Res 23:557–564. doi:10.1007/s11095-005-9444-5
Das M, Mishra D, Maiti TK, Basak A, Pramanik P (2008) Bio-functionalization of magnetite nanoparticles using an aminophosphonic acid coupling agent: new, ultradispersed, iron-oxide folate nanoconjugates for cancer-specific targeting. Nanotechnology 19:415101–415115
Das M, Mishra D, Dhak P, Gupta S, Maiti TK, Basak A, Pramanik P (2009) Biofunctionalized, phosphonate-grafted, ultrasmall iron oxide nanoparticles for combined targeted cancer therapy and multimodal imaging. Small 24:2883–2893
Hochuli E, Döbeli H, Schacher A (1987) New metal chelate adsorbent selective for proteins and peptides containing neighbouring histidine residues. J Chromatogr A 411:177–184
Hung CW, Holoman TRP, Kofinas P, Bentley WE (2008) Towards oriented assembly of proteins onto magnetic nanoparticles. Biochem Eng J 38:164–170
Kracalıkova K, Tishchenko G, Bleha M (2006) Effect of the matrix structure and concentration of polymer-immobilized Ni2+–iminodiacetic acid complexes on retention of IgG1. J Biochem Biophys Methods 67:7–25
Kreller DI, Gibson G, Novak W, Van Loon GW, Hugh HJ (2003) Competitive adsorption of phosphate and carboxylate with natural organic matter on hydrous iron oxides as investigated by chemical force microscopy. Colloids Surf A 212:249
Liao Y, Cheng Y, Li Q (2007) Preparation of nitrilotriacetic acid/Co2+-linked, silica/boron-coated magnetite nanoparticles for purification of 6× histidine-tagged proteins. J Chromatogr A 1143:65–71
Maceira VS, Duarte MAC, Farle M, Quintela AL, Sieradzki K, Diaz R (2006) Bifunctional gold-coated magnetic silica spheres. Chem Mater 18:2701–2706
Meng J, Walter JG, Kökpinar Ö, Stahl F, Scheper T (2008) Automated microscale His-tagged protein purification using Ni-NTA magnetic agarose beads. Chem Eng Technol 31:463–468
Mohapatra S, Pramanik P (2009) Synthesis and stability of functionalized iron oxide nanoparticles using organophosphorus coupling agents. Colloids Surf A 339:35–42
Mohapatra S, Pramanik N, Ghosh SK, Pramanik P (2006) Synthesis and characterization of ultrafine Poly(vinylalcohol phosphate) coated magnetite nanoparticles. J Nanosci Nanotechnol 6:823
Mohapatra S, Pal D, Ghosh SK, Pramanik P (2007a) Design of superparamagnetic iron oxide nanoparticle for purification of recombinant proteins. J Nanosci Nanotechnol 7:1–7
Mohapatra S, Mallick SK, Maiti TK, Ghosh SK, Pramanik P (2007b) Synthesis of highly stable folic acid conjugated magnetite nanoparticles for targeting cancer cells. Nanotechnology 18:385102–385111
Nagao D, Yokoyama M, Yamauchi N, Matsumoto H, Kobayashi Y, Konno M (2008) Synthesis of highly monodisperse particles composed of a magnetic core and fluorescent shell. Langmuir 24:9804–9808
Portet D, Denizot B, Rump E, Hindre F, LeJeune JJ, Jallet P (2001) Comparative biodistribution of thin-coated iron oxide nanoparticles TCION: effect of different bisphosphonate coatings. Drug Dev Res 54:173
Riviere C, Lecoeur C, Wilhelm C, Pechoux C, Combrisson H, Yiou R, Gazeau F (2009) The MRI assessment of intraurethrally delivered muscle precursor cells using anionic magnetic nanoparticles. Biomaterials 30:6920–6928
Roberts D, Zhu WL, Fromenn CM, Rosenzweig Z (2000) Synthesis of gadolinium oxide magnetoliposomes for magnetic resonance imaging. J Appl Phys 87:6208
Robinson I, Alexander C, Tung LD, Fernig DG, Thanh NTK (2009) Fabrication of water-soluble magnetic nanoparticles by ligand-exchange with thermo-responsive polymers. J Magn Magn Mater 321:1421–1423
Schreiber HA, Prechl J, Jiang H, Zozulya A, Fabry Z, Denes F, Sandor M (2010) Using carbon magnetic nanoparticles to target, track, and manipulate dendritic cells. J Immunol Methods 356:47–59
Shen W, Zhong H, Neff D, Norton ML (2009) NTA directed protein nanopatterning on DNA origami nanoconstructs. J Am Chem Soc 131:6660–6661
Tseng P, Carlo DD, Judy JW (2009) Rapid and dynamic intracellular patterning of cell-internalized magnetic fluorescent nanoparticles. Nano Lett 9:3053–3059
Xu C, Xu K, Gu H, Zhong X, Guo Z, Zheng R, Zhang X, Xu B (2004) Nitrilotriacetic acid-modified magnetic nanoparticles as a general agent to bind histidine-tagged proteins. J Am Chem Soc 126:3392–3393
Yee C, Kataby G, Ulman G, Prozorov T, White H, King A, Rafailovich M, Sokolov J, Gedanken A (1999) Self-assembled monolayers of alkanesulfonic and -phosphonic acids on amorphous iron oxide nanoparticles. Langmuir 15:7111
Acknowledgments
Acknowledgement is due to the Department of Biotechnology, Govt. of India and CSIR, Govt. of India for funding and Indian Institute of Technology, Kharagpur for execution of these studies. AC to CSIR, Govt. of India for fellowship.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sahu, S.K., Chakrabarty, A., Bhattacharya, D. et al. Single step surface modification of highly stable magnetic nanoparticles for purification of His-tag proteins. J Nanopart Res 13, 2475–2484 (2011). https://doi.org/10.1007/s11051-010-0140-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-010-0140-y