Abstract
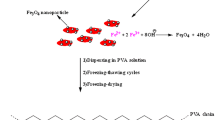
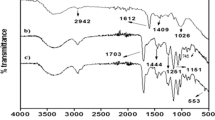
The present work involves the development of hydrogel magnetic nanocomposites for protein purification and heavy metal extraction applications. The magnetic nanoparticles (MNPs) were prepared in situ in poly(acrylamide)-gum acacia (PAM-GA) hydrogels. The formation of magnetic nanoparticles in the hydrogel networks was confirmed by Fourier transform infrared (FTIR) spectroscopy and X-ray diffraction (XRD). Scanning electron (SEM) microscopy studies revealed the formation of MNPs throughout the hydrogel networks. The average size of MNPs formed in the hydrogel networks was 3–5 nm as determined by transmission electron microscopy (TEM). The thermal properties of the hydrogel magnetic nanocomposites were evaluated by dynamic scanning calorimetry (DSC) and thermogravimetric (TG) analysis. The magnetic properties of the developed hydrogel magnetic nanocomposites were determined by a vibrating sample magnetometer (VSM). The swelling properties of the hydrogel and the hydrogel magnetic nanocomposites were studied in detail. The hydrogel magnetic nanocomposites are utilized for the removal of toxic metal ions such as Co(II), Ni(II), and Cu(II) and for protein purification. The results confirm that the hydrogel magnetic nanocomposites exhibit superior extraction properties to hydrogels.

Poly(acrylamide)-gum acacia (PAM-GA) magnetic nanocomposite hydrogels were synthesized to evaluate their applicability for protein purification and metal extraction









Similar content being viewed by others
References
Burda C, Chen XB, Narayana R, Sayed EI (2005) Chemi Revie 105:1025. doi:10.1002/chin.200527215
Kim BJ, Bang J, Hawker ET, Krammer EJ (2006) Macromolecules 39:4108. doi:10.1021/ma060308w
Kim DH, Kim SH, Lavery K, Russell TP (2004) Nano Lett 4:1841. doi:10.1021/nl049063w
Kim HC, Jia XO, Stafford CM, Kim DH, Mccarthy TJ, Tuominen M (2001) Adv Mater 13:795. doi:10.1002/1521-4095(200106)13:11<795::AIDADMA795>3.0.CO;2-1
Lazzari M, Quintela MAL (2003) Adv Mater 159:1583. doi:10.1002/adma.200300382
Yeh SW, Wu TL, Wei KH (2005) Nanotechnology 16:683. doi:10.1088/0957-4484/16/6/010
Jang SH, Jeonga GY, Mina BG, Lyoob WS, Lee SC (2008) J Hazard Mater 159:294. doi:10.1016/j.jhazmat.2008.02.018
Zhou L, Wang Y, Liu Z, Haung Q (2009) J Hazard Mater 161:995. doi:10.1016/j.jhazmat.2008.04.078
Zhou YT, Nie HL, White GB, He ZY, Zhu LM (2009) J Colloid Interface Sci 330:29. doi:10.1016/j.jcis.2008.10.026
Matlock MM, Howerton BS, Atwood DA (2002) Water Res 36:4757. doi:10.1016/S0043-1354(02)00149-5
Isshiki M (1996) Vaccum 47:885. doi:10.1016/0042-207X(96)00087-5
Grimm J, Bessarabov D, Sanderson R (1998) Desalination 115:285. doi:10.1016/S0011-9164(98)00047-2
Baccar R, Bouzid J, Feki M, Montiel F (2009) J Hazard Mater 162:1522. doi:10.1016/j.jhazmat.2008.06.041
Liu X, Hu Q, Fang Z, Zhang X, Zhang B (2009) Longmuir 25:3. doi:10.1021/la802754t
Ozay O, Kici S, Baran Y, Kubilay S, Aktas N, Sahiner N (2010) Desalination 260:57. doi:10.1016/j.desal.2010.04.067
Sahiner N, Ozay O (2011) Colloid Surf A: Physicochem Eng Asp 378:50. doi:10.1016/j.colsurfa.2011.01.053
Ilgin P, Avci G, Silan C, Ekici S, Aktas N, Ramesh SA, John VT, Sahiner N (2010) Carbohydr Polym 82:997. doi:10.1016/j.carbpol.2010.06.033
Ozay O, Aktas N, Inger E, Sahiner N (2011) Int J Hydrog Energy 36:1998. doi:10.1016/j.ijhydene.2010.11.045
Chauhan GS, Chauhan S, Sen U, Garg D (2009) Desalination 243:95. doi:10.1016/j.desal.2008.04.017
Drury JL, Mooney DY (2003) Biomaterials 24:4337. doi:10.1016/S01429612(03)00340-5
Shu XZ, Liu Y, Palumbo FS, Luo Y, Prestwich GD (2004) Biomaterials 25:1339. doi:10.1016/j.biomaterials.2003.08.014
Kim JJ, Park K (1998) Bioseparation 7:177. doi:10.1023/A:1008050124949
Gupta P, Vermani K, Garg S (2002) Drug Discov Today 7:569. doi:10.1016/S1359-6446(02)02255-9
Polizzotti BD, Fairbanks BD, Anseth KS (2008) Biomacromolecules 9:1084. doi:10.1021/bm7012636
Murthy PSK, Mohan YM, Varaprasad K, Sredhar B, Mohana Raju K (2008) J Colloid Interface Sci 318:217. doi:10.1016/j.jcis.2007.10.014
Vimala K, Sivudu KS, Mohan YM, Sreedhar B, Mohana Raju K (2009) Carbohydr Polym 75:463. doi:10.1016/j.carbpol.2008.08.009
Zrínyi M, Barsi L, Büki A (1997) Polym Gels Netw 5:415. doi:10.1016/S0966-7822(97)00010-5
Li B, Jia D, Zhou Y, Hu Q, Cai W (2006) J Magn Magn Mater 306:223. doi:10.1016/j.jmmm.2006.01.250
Bhattarai SR, Bahadur KCR, Aryal S, Khil MS, Kim HY (2007) Carbohydr Polym 69:467. doi:10.1016/j.carbpol.2007.01.006
Lao LL, Ramanujan RV (2004) J Mater Sci Mater Med 15:1061. doi:10.1023/B:JMSM.0000046386.78633.e5
Lu X, Mi YM (2005) Macromolecules 38:839. doi:10.1021/ma0486896
Jain TK, Morales MA, Sahoo SK, Leslie-Pelecky DL, Labhasetwar V (2005) Mol Pharam 2:194. doi:10.1021/mp0500014
Chia CH, Zakaria S, Ahamad S, Abdullah M, Jani SM (2006) Am J App Sci 3:1750, ISSN 1546–9239
Rozada F, Otero M, Moran A, Garcia AI (2008) Bioresour Technol 99:6332. doi:10.1016/j.biortech.2007.12.015
Ozay O, Kici SE, Baran Y, Aktas N, Sahiner N (2009) Water Res 43:4403. doi:10.1016/j.watres.2009.06.058
Sahiner N, Ozay O (2011) React Funct Polym 71:607. doi:10.1016/j.reactfunctpolym.2011.03.003
Acknowledgement
KMR and NNR thank the University Grants Commission (UGC), Government of India, New Delhi for the partial financial support (UGC Sanction Letter No.F.37-339/2009 (SR) dated 05-01-2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagireddy, N.R., Yallapu, M.M., Kokkarachedu, V. et al. Preparation and characterization of magnetic nanoparticles embedded in hydrogels for protein purification and metal extraction. J Polym Res 18, 2285–2294 (2011). https://doi.org/10.1007/s10965-011-9642-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10965-011-9642-2