Abstract
In this work, the complex of borneol–methyl-β-cyclodextrin was prepared both by supercritical carbon dioxide processing and by the sealed heating treatment at mild pressure and temperature. An amorphous complex was obtained by the sealed heating treatment. A crystalline inclusion complex was obtained by the supercritical carbon dioxide processing. The crystalline complex is more stable than the amorphous complex. The apparent aqueous solubility of borneol could be enhanced about 70 times by complexation with methyl-β-cyclodextrin.
Graphical Abstract
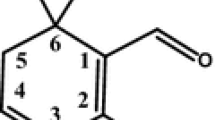
In this work, a 1:1 crystalline inclusion complex between borneol and methyl-β-cyclodextrin was obtained by supercritical carbon dioxide processing at temperature about 100 °C and pressure about 100 bar. This inclusion complex is more stable than the amorphous complex prepared by the sealed heating method. The apparent aqueous solubility of borneol could be enhanced about 70 times by complexation with methyl-β-cyclodextrin. The possible complex structure is shown below:









Similar content being viewed by others
References
Granger, R.E., Campbell, E.L., Johnston, G.A.R.: (+)- and (−)-borneol: efficacious positive modulators of GABA action at human recombinant α1β2γ2L GABAA receptors. Biochem. Pharmacol. 69, 1101–1111 (2005). doi:10.1016/j.bcp.2005.01.002
Bhatia, S.P., McGinty, D., Letizia, C.S., Api, A.M.: Fragrance material review on l-borneol. Food Chem. Toxicol. 46(Suppl 1), s81–s84 (2008). doi:10.1016/j.fct.2008.06.054
Bhatia, S.P., Letizia, C.S., Api. A.M.: Fragrance material review on borneol. Food Chem. Toxicol. 46(Suppl 1), s77–s80, (2008). doi:10.1016/j.fct.2008.06.031
Challa, R., Ahuja, A., Ali, J., Khar, R.K.: Cyclodextrins in drug delivery: an updated review. AAPS PharmSciTech 6(2), 43 (2005). doi:10.1208/pt060243
Song, H.T., Guo, T., Zhao, M.H., Zhang, R.H., Li, X., Bi, K.S.: Studies on physicochemical properties of borneol beta-cyclodextrin inclusion complex. J. Shenyang Pharm. Univ. 19, 249–252 (2002)
Yuan, M., Zhao, R.Y., Wang, P., Zhang, Z.L.: Study on the borneol–β-cyclodextrin inclusion complex. Qilu Pharm. Aff. 26, 242–245 (2007)
Wang, J., Zhang, T.J., Liao, M.L.: Study on the inclusion complex of borneol with hydroxypropyl-β-cyclodextrin. Tianjin J. Tradit. Chin. Med. 24, 150–152 (2007)
Van Hees, T., Piel, G., Evrared, B., Otte, X., Thunnus, L., Delattre, L.: Application of supercritical carbon dioxide for the preparation of a piroxicam-β-cyclodextrin inclusion compound. Pharm. Res. 16, 1864–1870 (1999). doi:10.1023/A:1018955410414
Charoenchaitrakool, M., Dehghani, F., Foster, N.R.: Utilization of supercritical carbon dioxide for complex formation of ibuprofen and methyl-cyclodextrin. Int. J. Pharm. 239, 103–112 (2002). doi:10.1016/S0378-5173(02)00078-9
Junco, S., Casimiro, T., Ribeiro, N., den Ponte, M.N., Marques, H.C.: A comparative study of naproxen-beta cyclodextrin complexes prepared by conventional methods and using supercritical carbon dioxide. J. Incl. Phenom. Macrocycl. Chem. 44, 117–121 (2002). doi:10.1023/A:1023022008337
Lai, S., Locci, E., Piras, A., Porcedda, S., Lai, A., Marongiu, B.: Imazalil–cyclomaltoheptaose (β-cyclodextrin) inclusion complex: preparation by supercritical carbon dioxide and 13C CPMAS and 1H NMR characterization. Carbohydr. Res. 338, 2227–2232 (2003). doi:10.1016/S0008-6215(03)00358-6
Locci, E., Lai, S., Piras, A., Marongiu, B., Lai, A.: 13C-CPMAS and 1H-NMR study of the inclusion complexes of β-cyclodextrin with carvacrol, thymol, and eugenol prepared in supercritical carbon dioxide. Chem. Biodivers. 1, 1354 (2004). doi:10.1002/cbdv.200490098
Bandia, N., Weib, W., Robertsc, C.B., Kotrac, L.P., Kompella, U.B.: Preparation of budesonide and indomethacin-hydroxypropyl-cyclodextrin (HPBCD) complexes using a single-step, organic-solvent-free supercritical fluid process. Eur. J. Pharm. Sci. 23, 159–168 (2004). doi:10.1016/j.ejps.2004.06.007
Rodier, E., Lochard, H., Sauceau, M., Letourneau, J.J., Freiss, B., Fages, J.: A three step supercritical process to improve the dissolution rate of eflucimibe. Eur. J. Pharm. Sci. 26, 184–193 (2005). doi:10.1016/j.ejps.2005.05.011
Shehatta, I., Al-Marzouqi, A.H., Jobe, B., Dowaidar, A.: Enhancement of aqueous solubility of itraconazole by complexation with cyclodextrins using supercritical carbon dioxide. Can. J. Chem. 83, 1833–1838 (2005). doi:10.1139/v05-181
Al-Marzouqi, A.H., Shehatta, I., Jobe, B., Dowaidar, A.: Phase solubility and inclusion complex of itraconazole with β-cyclodextrin using supercritical carbon dioxide. J. Pharm. Sci. 95, 292–304 (2006). doi:10.1002/jps.20535
Wang, B., He, J., Sun, D.H., Zhang, R., Han, B.X.: Utilization of supercritical carbon dioxide for preparation of 3-hydroxyflavone and β-cyclodextrin complex. J. Incl. Phenom. Macrocycl. Chem. 55, 37–40 (2006). doi:10.1007/s10847-005-9015-8
Al-Marzouqi, A.H., Jobe, B., Dowaidar, A., Maestrelli, F., Murab, P.: Evaluation of supercritical fluid technology as preparative technique of benzocaine–cyclodextrin complex comparison with conventional methods. J. Pharm. Biomed. 43, 566–574 (2007). doi:10.1016/j.jpba.2006.08.019
Turk, M., Upper, G., Steurenthaler, M., Hussein, K., Wahl, M.A.: Complex formation of ibuprofen and β–cyclodextrin by controlled particle deposition (CPD) using SC-CO2. J. Supercrit. Fluids 39, 435–443 (2007). doi:10.1016/j.supflu.2006.02.009
Bounaceur, A., Rodier, E., Fages, J.: Maturation of ketoprofen/β-cyclodextrin mixture with supercritical carbon dioxide. J. Supercrit. Fluids 41, 429–439 (2007). doi:10.1016/j.supflu.2006.11.004
Hussein, K., Turk, M., Wahl, M.A.: Comparative evaluation of ibuprofen/β-cyclodextrin complexes obtained by supercritical carbon dioxide and other conventional methods. Pharm. Res. 24, 585–592 (2007). doi:10.1007/s11095-006-9177-0
Al-Marzouqi, A.H., Jobe, B., Corti, G., Cirri, M., Mura, P.: Physicochemical characterization of drug-cyclodextrin complexes prepared by supercritical carbon dioxide and by conventional techniques. J. Incl. Phenom. Macrocycl. Chem. 57, 223–231 (2007). doi:10.1007/s10847-006-9192-0
Moribe, K., Fujito, T., Tozuka, Y., Yamamoto, K.: Solubility-dependent complexation of active pharmaceutical ingredients with trimethyl-β-cyclodextrin under supercritical fluid condition. J. Incl. Phenom. Macrocycl. Chem. 57, 289–295 (2007). doi:10.1007/s10847-006-9175-1
Lee, S.Y., Jung, In-II, Kim, J.K., Lim, G.B., Ryu, J.H.: Preparation of itraconazole/HP-B-CD inclusion complexes using supercritical aerosol solvent extraction system and their dissolution characteristics. J. Supercrit. Fluids 44, 400–408 (2008). doi:10.1016/j.supflu.2007.09.006
Al-Marzouqi, A.H., Solieman, A., Shehadi, I., Adem, A.: Influence of the preparation method on the physicochemical properties of econazole-β-cyclodextrin complexes. J. Incl. Phenom. Macrocycl. Chem. 60, 85–93 (2008). doi:10.1007/s10847-007-9356-6
Acknowledgments
This study is supported by Molecular Science Center of Institute of Chemistry, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
He, J., Li, W. Preparation of borneol–methyl-β-cyclodextrin inclusion complex by supercritical carbon dioxide processing. J Incl Phenom Macrocycl Chem 65, 249–256 (2009). https://doi.org/10.1007/s10847-009-9575-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-009-9575-0