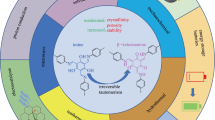
Abstract
The supramolecular chemistry of acyclic oligopyrrole derivatives mainly reported by the author’s group in the last four years has been summarized in this review. The author has demonstrated the “first step” to construct the new materials and concepts based on the new molecular systems consisting of pyrrole rings, which form the complexes, assemblies, and organized structures, by using noncovalent interactions such as metal coordination, hydrogen bonding, and π–π interaction. Acyclic π-conjugated oligopyrroles have exhibited not only host–guest binding behaviors in solutions but also the formation of, for example, (i) metal coordination polymers to give emissive colloidal spheres, (ii) solid-state assemblies of acyclic π-conjugated anion receptors and their anion complexes, (iii) anion-responsive supramolecular gels from the receptors with aliphatic chains, and (iv) solvent-assisted organized structures like vesicles derived from amphiphilic anion receptors.
















Similar content being viewed by others
References
Fischer, H., Orth, H.: Die Chemie des Pyrrols. Akademische Verlagsgesellschaft M. B. H, Leipzig (1934)
Kadish, K.M., Smith, K.M., Guilard, R. (eds.): The Porphyrin Handbook. Academic Press, San Diego (2000)
Tsoucaris, G. (ed.): Current Challenges on Large Supramolecular Assemblies. NATO Science Series. Kluwer, Dordrecht (1999)
Ciferri, A. (ed.): Supramolecular Polymers. Marcel Dekker, New York, Basel (2000)
Ozin, G.A., Arsenault, A.C.: Nanochemistry: A Chemical Approach to Nanomaterials. RSC, Cambridge (2005)
Würthner, F. (ed.): Supramolecular Dye Chemistry. Topics in Current Chemistry. Springer Verlag, Berlin (2005)
Shimizu, T., Matsuda, M., Minamikawa, H.: Supramolecular nanotube architectures based on amphiphilic molecules. Chem. Rev. 105, 1401–1443 (2005). doi:10.1021/cr030072j
Hoeben, F.J.M., Jonkheijm, P., Meijer, E.W., Schenning, A.P.H.J.: About supramolecular assemblies of π-conjugated systems. Chem. Rev. 105, 1491–1546 (2005). doi:10.1021/cr030070z
Maeda, H.: Supramolecular chemistry of acyclic oligopyrroles. Eur. J. Org. Chem. 5313–5325 (2007), and references therein. doi:10.1002/ejoc.200700382
Sauvage, J.-P. (ed.): Transition Metals in Supramolecular Chemistry. Wiley, Chichester (1999)
Fujita, M., Umemoto, K., Yoshizawa, M., Fujita, N., Kusukawa, T., Biradha, K.: Molecular paneling via coordination. Chem. Commun. (Camb.) 509–518 (2001). doi:10.1039/b008684n
Seidel, S.R., Stang, P.J.: High-symmetry coordination cages via self-assembly. Acc. Chem. Res. 35, 972–983 (2002). doi:10.1021/ar010142d
Kitagawa, S., Kitaura, R., Noro, S.: Functional porous coordination polymers. Angew. Chem. Int. Ed. 43, 2334–2375 (2004). doi:10.1002/anie.200300610
Dobrawa, R., Würthner, F.: Metallosupramolecular approach toward functional coordination polymers. J. Polym. Sci. A 43, 4981–4995 (2005). doi:10.1002/pola.20997
Schubert, U.S., Newkome, G.R., Manners, I. (eds.): Metal-Containing and Metallosupramolecular Polymers and Materials, vol. 928. ACS Symposium Series, American Chemical Society, Washington DC (2006)
Oh, M., Mirkin, C.A.: Chemically tailorable colloidal particles from infinite coordination polymers. Nature 438, 651–654 (2005). doi:10.1038/nature04191
Oh, M., Mirkin, C.A.: Ion exchange as a way of controlling the chemical compositions of nano- and microparticles made from infinite coordination polymers. Angew. Chem. Int. Ed. 45, 5492–5494 (2006). doi:10.1002/anie.200601918
Würthner, F., Stepanenko, V., Sautter, A.: Rigid-rod metallosupramolecular polymers of dendronized diazadibenzoperylene dyes. Angew. Chem. Int. Ed. 45, 1939–1942 (2006). doi:10.1002/anie.200503717
Falk, H.: The Chemistry of Linear and Oligopyrroles and Bile Pigments. Springer-Verlag, Vienna (1989)
Brückner, C., Zhang, Y., Rettig, S.J., Dolphin, D.: Synthesis, derivatization and structural characterization of octahedral tris(5-phenyl-4, 6-dipyrrinato) complexes of cobalt(III) and iron(III). Inorg. Chim. Acta 263, 279–286 (1997). doi:10.1016/S0020-1693(97)05700-9
Zhang, Y., Thompson, A., Rettig, S.J., Dolphin, D.: The use of dipyrromethene ligands in supramolecular chemistry. J. Am. Chem. Soc. 120, 13537–13538 (1998). doi:10.1021/ja9834982
Thompson, A., Rettig, S.J., Dolphin, D.: Self-assembly of novel trimers using dipyrromethene ligands. Chem. Commun. (Camb.) 631–632 (1999). doi:10.1039/a809192g
Thompson, A., Dolphin, D.: Nuclear magnetic resonance studies of helical dipyrromethene-zinc complexes. Org. Lett. 2, 1315–1318 (2000). doi:10.1021/ol000053l
Chen, Q., Zhang, Y., Dolphin, D.: Synthesis and self-assembly of novel tetra- and hexapyrroles containing dipyrrins linked by a sulfur bridge at the β-position. Tetrahedron Lett. 43, 8413–8416 (2002). doi:10.1016/S0040-4039(02)01958-5
Wood, T.E., Thompson, A.: Advances in the chemistry of dipyrrins and their complexes. Chem. Rev. 107, 1831–1861 (2007). doi:10.1021/cr050052c
Halper, S.R., Cohen, S.M.: Synthesis, structure, and spectroscopy of phenylacetylenylene rods incorporating meso-substituted dipyrrin ligands. Chem. Eur. J. 9, 4661–4669 (2003). doi:10.1002/chem.200305041
Halper, S.R., Cohen, S.M.: Heterometallic metal-organic frameworks based on tris(dipyrrinato) coordination complexes. Inorg. Chem. 44, 486–488 (2005). doi:10.1021/ic048289z
Murphy, D.L., Malachowski, M.R., Campana, C.F., Cohen, S.M.: A chiral, heterometallic metal-organic framework derived from a tris(chelate) coordination complex. Chem. Commun. (Camb.) 5506–5508 (2005). doi:10.1039/b510915a
Yu, L., Muthukumaran, K., Sazanovich, I.V., Kirmaier, C., Hindin, E., Diers, J.R., Boyle, P.D., Bocian, D.F., Holten, D., Lindsey, J.S.: Excited-state energy-transfer dynamics in self-assembled triads composed of two porphyrins and an intervening Bis(dipyrrinato)metal complex. Inorg. Chem. 42, 6629–6647 (2003). doi:10.1021/ic034559m
Maeda, H., Hashimoto, T., Fujii, R., Hasegawa, M.: Dipyrrin ZnII complexes with functional aryl groups: formation, characterization, and structures in the solid state. J. Nanosci. Nanotechnol. 9, 240–248 (2009). doi:10.1166/jnn.2009.J011
Maeda, H., Ito, M.: Dipyrrin-porphyrin hybrids: potential π-conjugated platform to fabricate coordination oligomers. Chem. Lett. 34, 1150–1151 (2005). doi:10.1246/cl.2005.1150
Maeda, H., Hasegawa, M., Hashimoto, T., Kakimoto, T., Nishio, S., Nakanishi, T.: Nanoscale spherical architectures fabricated by metal coordination of multiple dipyrrin moieties. J. Am. Chem. Soc. 128, 10024–10025 (2006). doi:10.1021/ja0637301
Maeda, H., Hashimoto, T.: Nanoscale metal coordination macrocycles fabricated by using “dimeric” dipyrrins. Chem. Eur. J. 13, 7900–7907 (2007). doi:10.1002/chem.200700444
Jeffrey, G.A., Saenger, W.: Hydrogen Bonding in Biological Structures. Springer, Berlin (1991)
Whitesides, G.M., Simanek, E.E., Mathias, J.P., Seto, C.T., Chin, D.N., Mannen, M., Gordon, D.M.: Noncovalent synthesis: using physical-organic chemistry to make aggregates. Acc. Chem. Res. 28, 37–44 (1995). doi:10.1021/ar00049a006
Prins, L.J., Reinhoudt, D.N., Timmerman, P.: Noncovalent synthesis using hydrogen bonding. Angew. Chem. Int. Ed. 40, 2382–2426 (2001). doi:10.1002/1521-3773(20010702)40:13<2382::AID-ANIE2382>3.0.CO;2-G
Ajayaghosh, A., George, S.J., Schenning, A.P.H.J.: Hydrogen-bonded assemblies of dyes and extended π-conjugated systems. In: Würthner, F. (ed.) Supramolecular Dye Chemistry. Topics in Current Chemistry, vol. 258, pp. 83–118. Springer-Verlag, Berlin (2005)
Sessler, J.L., Berthon-Gelloz, G., Gale, P.A., Camiolo, S., Anslyn, E.V., Anzenbacher Jr., P., Furuta, H., Kirkovits, G.J., Lynch, V.M., Maeda, H., Morosini, P., Scherer, M., Shriver, J., Zimmerman, R.S.: Oligopyrrole-based solid state self-assemblies. Polyhedron 22, 2963–2983 (2003). doi:10.1016/S0277-5387(03)00436-4
Sessler, J.L., Weghorn, S.J., Hiseada, Y., Lynch, V.: Hexaalkyl terpyrrole: a new building block for the preparation of expanded porphyrins. Chem. Eur. J. 1, 56–67 (1995). doi:10.1002/chem.19950010110
Scherer, M., Sessler, J.L., Gebauer, A., Lynch, V.: Synthesis and properties of pyrrole-substituted cyclopentadienes. J. Org. Chem. 62, 7877–7881 (1997). doi:10.1021/jo970818b
Scherer, M., Sessler, J.L., Moini, M., Gebauer, A., Lynch, V.M.: Self-assembly of pyrrole-ferrocene hybrids, determined inter alia by a new chemically induced electrospray mass spectrometry technique. Chem. Eur. J. 4, 152–158 (1998). doi:10.1002/(SICI)1521-3765(199801)4:1<152::AID-CHEM152>3.0.CO;2-E
Schmuck, C., Wienand, W.: Highly stable self-assembly in water: ion pair driven dimerization of a guanidiniocarbonyl pyrrole carboxylate zwitterion. J. Am. Chem. Soc. 125, 452–459 (2003). doi:10.1021/ja028485+
Wang, Y., Fu, H., Peng, A., Zhao, Y., Ma, J., Ma, Y., Yao, J.: Distinct nanostructures from isomeric molecules of bis(iminopyrrole) benzenes: effects of molecular structures on nanostructural morphologies. Chem. Commun. (Camb.) 1623–1625 (2007). doi:10.1039/b701327b
Oddo, B., Dainotti, C.: Syntheses in the pyrrole group. V. pyrrolic α-, β- and γ-diketones. Gazz. Chim. Ital. 42, 716–726 (1912)
Stark, W.M., Baker, M.G., Leeper, F.J., Raithby, P.R., Battersby, A.R.: Biosynthesis of porphyrins and related macrocycles. Part 30. Synthesis of the macrocycle of the spiro system proposed as an intermediate generated by cosynthetase. J. Chem. Soc. Perkin Trans. 1, 1187–1201 (1988). doi:10.1039/p19880001187
Maeda, H., Kusunose, Y., Terasaki, M., Ito, Y., Fujimoto, C., Fujii, R., Nakanishi, T.: Micro- and nanometer-scale porous, fibrous, and sheet architectures fabricated by supramolecular assemblies of dipyrrolyldiketones. Chem. Asian J. 2, 350–357 (2007). doi:10.1002/asia.200600379
Maeda, H., Hasegawa, M., Ueda, A.: Hydrogen bonding self-assemblies with 1-D linear, dimeric and hexagonal nanostructures of meso-pyridyl-substituted dipyrromethanes. Chem. Commun. (Camb.) 2726–2728 (2007). doi:10.1039/b703236f
Bianchi, A., Bowman-James, K., García-España, E. (eds.): Supramolecular Chemistry of Anions. Wiley-VCH, New York (1997)
Singh, R.P., Moyer, B.A. (eds.): Fundamentals and Applications of Anion Separations. Kluwer Academic/Plenum Publishers, New York (2004)
Sessler, J.L., Gale, P.A., Cho, W.-S.: Anion Receptor Chemistry. RSC, Cambridge (2006)
Vilar, R. (ed.): Recognition of Anions: Structure and Bonding. Springer-Verlag, Berlin (2008)
Schmidtchen, F.P., Berger, M.: Signaling recognition events with fluorescent sensors and switches. Chem. Rev. 97, 1515–1566 (1997). doi:10.1021/cr9603845
Beer, P.D., Gale, P.A.: Anion recognition and sensing: the state of the art and future perspectives. Angew. Chem. Int. Ed. 40, 486–516 (2001). doi:10.1002/1521-3773(20010202)40:3<486::AID-ANIE486>3.0.CO;2-P
Sessler, J.L., Camiolo, S., Gale, P.A.: Pyrrolic and polypyrrolic anion binding agents. Coord. Chem. Rev. 240, 17–55 (2003)
Martínez-Máñez, R., Sancenón, F.: Fluorogenic and chromogenic chemosensors and reagents for anions. Chem. Rev. 103, 4419–4476 (2003). doi:10.1021/cr010421e
Gale, P.A.: Amidopyrroles: from anion receptors to membrane transport agents. Chem. Commun. (Camb.) 3761–3772 (2005). doi:10.1039/b504596g
Gale, P.A.: Structural and molecular recognition studies with acyclic anion receptors. Acc. Chem. Res. 39, 465–475 (2006). doi:10.1021/ar040237q
Williams, D.H., Cox, J.P.L., Doig, A.J., Gardner, M., Gerhard, U., Kaye, P.T., Lal, A.R., Nicholls, I.A., Salter, C.J., Mitchell, R.C.: Toward the semiquantitative estimation of binding constants. Guides for peptide-peptide binding in aqueous solution. J. Am. Chem. Soc. 113, 7020–7030 (1991). doi:10.1021/ja00018a047
Searle, M.S., Williams, D.H., Gerhard, U.: Partitioning of free energy contributions in the estimation of binding constants: residual motions and consequences for amide–amide hydrogen bond strengths. J. Am. Chem. Soc. 114, 10697–10704 (1992). doi:10.1021/ja00053a003
Verdejo, B., Aguilar, J., Doménech, A., Miranda, C., Navarro, P., Jiménez, H.R., Soriano, C., García-España, E.: Binuclear Cu2+ complex mediated discrimination between l-glutamate and l-aspartate in water. Chem. Commun. (Camb.) 3086–3088 (2005). doi:10.1039/b503417e
Gunnlaugsson, T., Kruger, P.E., Jensen, P., Tierney, J., Ali, H.D.P., Hussey, G.M.: Colorimetric “naked eye” sensing of anions in aqueous solution. J. Org. Chem. 70, 10875–10878 (2005). doi:10.1021/jo0520487
Nagai, K., Maeda, K., Takeyama, Y., Sakajiri, K., Yashima, E.: Helicity induction and chiral amplification in a poly(phenylacetylene) bearing N,N-diisopropylaminomethyl groups with chiral acids in water. Macromolecules 38, 5444–5451 (2005). doi:10.1021/ma0507241
Sessler, J.L., Cyr, M.J., Lynch, V., McGhee, E., Ibers, J.A.: Synthetic and structural studies of sapphyrin, a 22-π-electron pentapyrrolic “expanded porphyrin”. J. Am. Chem. Soc. 112, 2810–2813 (1990). doi:10.1021/ja00163a059
Shionoya, M., Furuta, H., Lynch, V., Harriman, A., Sessler, J.L.: Diprotonated sapphyrin: a fluoride selective halide anion receptor. J. Am. Chem. Soc. 114, 5714–5722 (1992). doi:10.1021/ja00040a034
Sessler, J.L., Davis, J.: Sapphyrins: versatile anion binding agents. Acc. Chem. Res. 34, 989–997 (2001). doi:10.1021/ar980117g
Gale, P.A., Sessler, J.L., Král, V., Lynch, V.: Calix[4]pyrroles: old yet new anion-binding agents. J. Am. Chem. Soc. 118, 5140–5141 (1996). doi:10.1021/ja960307r
Gale, P.A., Sessler, J.L., Král, V.: Calixpyrroles. Chem. Commun. (Camb.) 1–8 (1998). doi:10.1039/a706280j
Sessler, J.L., Gross, D.E., Cho, W.-S., Lynch, V.M., Schmidtchen, F.P., Bates, G.W., Light, M.E., Gale, P.A.: Calix[4]pyrrole as a chloride anion receptor: solvent and counter cation effects. J. Am. Chem. Soc. 128, 12281–12288 (2006). doi:10.1021/ja064012h
Black, C.B., Andrioletti, B., Try, A.C., Ruiperez, C., Sessler, J.L.: Dipyrrolylquinoxalines: efficient sensors for fluoride anion in organic solution. J. Am. Chem. Soc. 121, 10438–10439 (1999). doi:10.1021/ja992579a
Sessler, J.L., Maeda, H., Mizuno, T., Lynch, V.M., Furuta, H.: Quinoxaline-oligopyrroles: improved pyrrole-based anion receptors. Chem. Commun. (Camb.) 862–863 (2002). doi:10.1039/b111708d
Sessler, J.L., An, D., Cho, W.-S., Lynch, V., Marquez, M.: Calix[n]bisdipyrrolylbenzenes: synthesis, characterization, and preliminary anion binding studies. Chem. Eur. J. 11, 2001–2011 (2005). doi:10.1002/chem.200400894
Vega, I.E.D., Camiolo, S., Gale, P.A., Hursthouse, M.B., Light, M.E.: Anion complexation properties of 2,2′-bisamidodipyrrolylmethanes. Chem. Commun. (Camb.) 1686–1687 (2003). doi:10.1039/b303532h
Vega, I.E.D., Gale, P.A., Hursthouse, M.B., Light, M.E.: Anion binding properties of 5,5′-dicarboxamido-dipyrrolylmethanes. Org. Biomol. Chem. 2, 2935–2941 (2004). doi:10.1039/b409115a
Vega, I.E.D., Gale, P.A., Light, M.E., Leob, S.J.: NH vs. CH hydrogen bond formation in metal–organic anion receptors containing pyrrolylpyridine ligands. Chem. Commun. (Camb.) 4913–4915 (2005). doi:10.1039/b510506d
Schmuck, C.: Side chain selective binding of N-acetyl-α-amino acid carboxylates by a 2-(guanidiniocarbonyl)pyrrole receptor in aqueous solvents. Chem. Commun. (Camb.) 843–844 (1999). doi:10.1039/a901126i
Schmuck, C., Geiger, L.: Carboxylate binding by guanidiniocarbonyl pyrroles: from self-assembly to peptide receptors. Curr. Org. Chem. 7, 1485–1502 (2003). doi:10.2174/1385272033486387
Schmuck, C., Geiger, L.: Efficient complexation of n-acetyl amino acid carboxylates in water by an artificial receptor: unexpected cooperativity in the binding of glutamate but not aspartate. J. Am. Chem. Soc. 127, 10486–10487 (2005). doi:10.1021/ja052699k
Maeda, H., Ito, Y., Kusunose, Y., Nakanishi, T.: Dipyrrolylpyrazoles: anion receptors in protonated form and efficient building blocks for organized structures. Chem. Commun. (Camb.) 1136–1138 (2007). doi:10.1039/b615787d
Maeda, H., Kusunose, Y.: Dipyrrolyldiketone difluoroboron complexes: novel anion sensors with C–H···X−interactions. Chem. Eur. J. 11, 5661–5666 (2005). doi:10.1002/chem.200500627
Maeda, H., Terasaki, M., Haketa, Y., Mihashi, Y., Kusunose, Y.: BF2 complexes of α-alkyl-substituted dipyrrolyldiketones as acyclic anion receptors. Org. Biomol. Chem. 6, 433–436 (2008). doi:10.1039/b718317h
Maeda, H., Kusunose, Y., Mihashi, Y., Mizoguchi, T.: BF2 complexes of β-tetraethyl-substituted dipyrrolyldiketones as anion receptors: potential building subunits for oligomeric systems. J. Org. Chem. 72, 2616–2621 (2007). doi:10.1021/jo062660d
Maeda, H., Ito, Y.: BF2 complex of fluorinated dipyrrolyldiketone: a new class of efficient receptor for acetate anions. Inorg. Chem. 45, 8205–8210 (2006). doi:10.1021/ic0608703
Fujimoto, C., Kusunose, Y.: CH···anion interaction in BF2 complexes of C3-bridged oligopyrroles. H. Maeda. J. Org. Chem. 71, 2389–2394 (2006). doi:10.1021/jo052511f
Maeda, H., Haketa, Y., Nakanishi, T.: Aryl-substituted C3-bridged oligopyrroles as anion receptors for formation of supramolecular organogels. J. Am. Chem. Soc. 129, 13661–13674 (2007). doi:10.1021/ja074435z
Maeda, H., Eifuku, N.: Receptors with mono-alkoxy-substituted aryl rings have also been synthesized: Alkoxy-substituted derivatives of π-conjugated acyclic anion receptors: effects of substituted positions. Chem. Lett. 38, 208–209 (2009). doi:10.1246/cl.2009.208
Maeda, H., Haketa, Y.: Selective iodinated dipyrrolyldiketone BF2 complexes as potential building units for oligomeric systems. Org. Biomol. Chem. 6, 3091–3095 (2008). doi:10.1039/b806161k
Maeda, H., Mihashi, Y., Haketa, Y.: Heteroaryl-substituted C3-bridged oligopyrroles: potential building subunits of anion-responsive π-conjugated oligomers. Org. Lett. 10, 3179–3182 (2008). doi:10.1021/ol801014z
Maeda, H., Bando, Y., Haketa, Y., Seki, S., Tohnai, N.: Recently, we have found the formation of multicrystalline systems of 9a. (to be submitted)
Maeda, H., Haketa, Y., Bando, Y., Sakamoto, S.: For single-crystal structures of β-ethyl-substituted receptor 9d: Synthesis, properties, and solid-state assemblies of β-alkyl-substituted dipyrrolyldiketone BF2 complexes. Synth. Met. 159 (2009). doi:10.1016/j.synthmet.2009.01.004
Maeda, H., Haketa, Y., Terashima, Y., Shimosugi, S., Ohta, K.: Effects of counter cations have also been examined. (to be submitted)
Maeda, H., Ito, Y., Haketa, Y., Eifuku, N., Lee, E., Lee, M., Hashishin, T., Kaneko, K.: Solvent-assisted organized structures based on amphiphilic anion-responsive π-conjugated systems. Chem. Eur. J. 15, (2009). doi:10.1002/chem.200802152
Maeda, H., Fujii, Y., Mihashi, Y.: Diol-substituted boron complexes of dipyrrolyl diketones as anion receptors and covalently linked ‘pivotal’ dimers. Chem. Commun. (Camb.) 4285–4287 (2008). doi:10.1039/b806361c
Sanchez-Queseda, J., Seel, C., Prados, P., de Mendoza, J.: Anion helicates: double strand helical self-assembly of chiral bicyclic guanidinium dimers and tetramers around sulfate templates. J. Am. Chem. Soc. 118, 277–278 (1996). doi:10.1021/ja953243d
Keegan, K., Kruger, P.E., Nieuwenhuyzen, M., O’Brien, J., Martin, N.: Anion directed assembly of a dinuclear double helicate. Chem. Commun. (Camb.) 2192–2193 (2001). doi:10.1039/b106981k
Coles, S.J., Frey, J.G., Gale, P.A., Hursthouse, M.B., Light, M.E., Navakhun, K., Thomas, G.L.: Anion-directed assembly: the first fluoride-directed double helix. Chem. Commun. (Camb.) 568–569 (2003). doi:10.1039/b210847j
Coles, S.J., Gale, P.A., Hursthouse, M.B., Light, M.E., Warriner, C.N.: Crystallographic and solution anion binding studies of bis-amidofurans and thiophenes. Supramol. Chem. 16, 469–486 (2004). doi:10.1080/10610270410001713303
Curiel, D., Cowley, A., Beer, P.D.: Indolocarbazoles: a new family of anion sensors. Chem. Commun. (Camb.) 236–238 (2005). doi:10.1039/b412363h
Brook, S.J., Evans, L.S., Gale, P.A., Hursthouse, M.B., Light, M.E.: ‘Twisted’ isophthalamide analogues. Chem. Commun. (Camb.) 734–735 (2005). doi:10.1039/b413654c
Gale, P.A., Light, M.E., McNally, B., Navakhun, K., Sliwinski, K.E., Smith, B.D.: Co-transport of H+/Cl− by a synthetic prodigiosin mimic. Chem. Commun. (Camb.) 3773–3775 (2005). doi:10.1039/b503906a
Hirose, J., Inoue, K., Sakuragi, H., Kikkawa, M., Minakami, M., Morikawa, T., Iwamoto, H., Hiromi, K.: Anions binding to bilirubin oxidase from Trachyderma tsunodae K-2593. Inorg. Chim. Acta 273, 204–212 (1998). doi:10.1016/S0020-1693(97)06183-5
Sato, M., Kanamori, T., Kamo, N., Demura, M., Nitta, K.: Stopped-flow analysis on anion binding to blue-form halorhodopsin from Natronobacterium pharaonis: comparison with the anion-uptake process during the photocycle. Biochemistry 41, 2452–2458 (2002). doi:10.1021/bi011788g
Swager, T.M.: Semiconducting Poly(arylene ethylene)s. In: Diederich, F., Stang, P.J., Tykwinski, R.R. (eds.) Acetylene Chemistry. Wiley-VCH, New York (2005). (Chap. 6)
McQuade, D.T., Pullen, A.E., Swager, T.M.: Conjugated polymer-based chemical sensors. Chem. Rev. 100, 2537–2574 (2000). doi:10.1021/cr9801014
Rose, A., Zhu, Z., Madigan, C.F., Swager, T.M., Bulovi’c, V.: Sensitivity gains in chemosensing by lasing action in organic polymers. Nature 434, 876–879 (2005). doi:10.1038/nature03438
Fages, F. (ed.): Low Molecular Mass Gelators, vol. 256, p. 283. Topics in Current Chemistry. Springer-Verlag, Berlin (2005)
Ishi-i, T., Shinkai, S.: Dye-Based Organogels: Stimuli-Responsive Soft Materials Based on One-Dimensional Self-Assembling Aromatic Dyes. In: Würthner, F. (ed.) Supramolecular Dye Chemistry. Topics in Current Chemistry, vol. 258, pp. 119–160. Springer-Verlag, Berlin (2005)
Terech, P., Weiss, R.G.: Low molecular mass gelators of organic liquids and the properties of their gels. Chem. Rev. 97, 3133–3159 (1997). doi:10.1021/cr9700282
Abdallah, D.J., Weiss, R.G.: Organogels and low molecular mass organic gelators. Adv. Mater. 12, 1237–1247 (2000). doi:10.1002/1521-4095(200009)12:17<1237::AID-ADMA1237>3.0.CO;2-B
Kato, T., Mizoshita, N., Kishimoto, K.: Functional liquid-crystalline assemblies: self-organized soft materials. Angew. Chem. Int. Ed. 45, 38–68 (2006). doi:10.1002/anie.200501384
van Esch, J.H., Feringa, B.L.: New functional materials based on self-assembling organogels: from serendipity towards design. Angew. Chem. Int. Ed. 39, 2263–2266 (2000). doi:10.1002/1521-3773(20000703)39:13<2263::AID-ANIE2263>3.0.CO;2-V
Kawano, S.-i., Tamaru, S.-i., Fujita, N., Shinkai, S.: Sol-gel polycondensation of tetraethyl orthosilicate (TEOS) in sugar-based porphyrin organogels: inorganic conversion of a sugar-directed porphyrinic fiber library through Sol-gel transcription processes. Chem. Eur. J. 10, 343–351 (2004). doi:10.1002/chem.200305042
Shirakawa, M., Fujita, N., Shinkai, S.: A stable single piece of unimolecularly π-stacked porphyrin aggregate in a thixotropic low molecular weight gel: a one-dimensional molecular template for polydiacetylene wiring up to several tens of micrometers in length. J. Am. Chem. Soc. 127, 4164–4165 (2005). doi:10.1021/ja042869d
Kitahara, T., Shirakawa, M., Kawano, S.-i., Beginn, U., Fujita, N., Shinkai, S.: Creation of a mixed-valence state from one-dimensionally aligned ttf utilizing the self-assembling nature of a low molecular-weight gel. J. Am. Chem. Soc. 127, 14980–14981 (2005). doi:10.1021/ja0552038
Yagai, S., Nakajima, T., Kishikawa, K., Kohmoto, S., Karatsu, T., Kitamura, A.: Hierarchical organization of photoresponsive hydrogen-bonded rosettes. J. Am. Chem. Soc. 127, 11134–11139 (2005). doi:10.1021/ja052645a
Maeda, H.: Recently, the author has summarized the recent progress of the anion-responsive supramolecular gels: Anion-responsive supramolecular gels. Chem. Eur. J. 14, 11274–11282 (2008). doi:10.1002/chem.200801333
Balaban, T.S., Tamiaki, H., Holzwarth, A.R.: Chlorins Programmed for Self-Assembly. In: Würthner, F. (ed.) Supramolecular Dye Chemistry. Topics in Current Chemistry, vol. 258, pp. 1–38. Springer-Verlag, Berlin (2005)
Webb, J.E.A., Crossley, M.J., Turner, P., Thordarson, P.: Pyromellitamide aggregates and their response to anion stimuli. J. Am. Chem. Soc. 129, 7155–7162 (2007). doi:10.1021/ja0713781
Džolić, Z., Cametti, M., Cort, A.D., Mandolini, L., Žinić, M.: Fluoride-responsive organogelator based on oxalamide-derived anthraquinone. Chem. Commun. (Camb.) 3535–3537 (2007). doi:10.1039/b707466b
Wang, C., Zhang, D., Zhu, D.: A chiral low-molecular-weight gelator based on binaphthalene with two urea moieties: modulation of the CD spectrum after gel formation. Langmuir 23, 1478–1482 (2007). doi:10.1021/la062621x
Yang, H., Yi, T., Zhou, Z., Zhou, Y., Wu, J., Xu, M., Li, F., Huang, C.: Switchable fluorescent organogels and mesomorphic superstructure based on naphthalene derivatives. Langmuir 23, 8224–8230 (2007). doi:10.1021/la7005919
Yamanaka, M., Nakamura, T., Nakagawa, T., Itagaki, H.: Reversible sol–gel transition of a tris–urea gelator that responds to chemical stimuli. Tetrahedron Lett. 48, 8990–8993 (2007). doi:10.1016/j.tetlet.2007.10.090
Stanley, C.E., Clarke, N., Anderson, K.M., Elder, J.A., Lenthall, J.T., Steed, J.W.: Anion binding inhibition of the formation of a helical organogel. Chem. Commun. (Camb.) 3199–3201 (2006). doi:10.1039/b606373j
Piepenbrock, M.-O.M., Lloyd, G.O., Clarke, N., Steed, J.W.: Gelation is crucially dependent on functional group orientation and may be tuned by anion binding. Chem. Commun. (Camb.) 2644–2646 (2008). doi:10.1039/b804259d
Applegarth, L., Clark, N., Richardson, A.C., Parker, A.D.M., Radosavljevic-Evans, I., Goeta, A.E., Howard, J.A.K., Steed, J.W.: Modular nanometer-scale structuring of gel fibres by sequential self-organization. Chem. Commun. (Camb.) 5423–5425 (2005). doi:10.1039/b511259a
Li, Q., Wang, Y., Li, W., Wu, L.: Structural characterization and chemical response of a Ag-coordinated supramolecular gel. Langmuir 23, 8217–8223 (2007). doi:10.1021/la700364t
Kishimura, A., Yamashita, T., Aida, T.: Phosphorescent organogels via “Metallophilic” interactions for reversible RGB-color switching. J. Am. Chem. Soc. 127, 179–183 (2005). doi:10.1021/ja0441007
Kim, H.-J., Zin, W.-C., Lee, M.: Anion-directed self-assembly of coordination polymer into tunable secondary structure. J. Am. Chem. Soc. 126, 7009–7014 (2004). doi:10.1021/ja049799v
Kim, H.-J., Lee, J.-H., Lee, M.: Stimuli-responsive gels from reversible coordination polymers. Angew. Chem. Int. Ed. 44, 5810–5814 (2005). doi:10.1002/anie.200501270
Voet, D., Voet, J.G.: Biochemistry. Wiley, New York (2004)
Israelachvili, J.N.: Intermolecular and Surface Forces, p. 450. Academic Press, London (1992)
Hamley, I.W.: Introduction to Soft Matter–Polymers, Colloids, Amphiphiles and Liquid Crystals, p. 342. Wiley, Chichester (2000)
Lee, M., Jeong, Y.-S., Cho, B.-K., Oh, N.-K., Zin, W.-C.: Self-assembly of molecular dumbbells into organized bundles with tunables size. Chem. Eur. J. 8, 876–883 (2002). doi:10.1002/1521-3765(20020215)8:4<876::AID-CHEM876>3.0.CO;2-M
Yoo, Y.-S., Choi, J.-H., Song, J.-H., Oh, N.-K., Zin, W.-C., Park, S., Chang, T., Lee, M.: Self-assembling molecular trees containing octa-p-phenylene: from nanocrystals to nanocapsules. J. Am. Chem. Soc. 126, 6294–6300 (2004). doi:10.1021/ja048856h
Jin, L.-Y., Ahn, J.-H., Lee, M.: Shape-persistent macromolecular disks from reactive supramolecular rod bundles. J. Am. Chem. Soc. 126, 12208–12209 (2004). doi:10.1021/ja0465552
Hong, D.-J., Lee, E., Lee, M.: Nanofibers from self-assembly of an aromatic facial amphiphile with oligo(ethylene oxide) dendrons. Chem. Commun. (Camb.) 1801–1803 (2007). doi:10.1039/b617404c
Moon, K.-S., Kim, H.-J., Lee, E., Lee, M.: Self-assembly of T-shaped aromatic amphiphiles into stimulus-responsive nanofibers. Angew. Chem. Int. Ed. 46, 6807–6810 (2007). doi:10.1002/anie.200702136
Lee, E., Jeong, Y.-H., Kim, J.-K., Lee, M.: Controlled self-assembly of asymmetric dumbbell-shaped rod amphiphiles: transition from toroids to planar nets. Macromolecules 40, 8355–8360 (2007). doi:10.1021/ma071511+
Hill, J.P., Jin, W., Kosaka, A., Fukushima, T., Ichihara, H., Shimomura, T., Ito, K., Hashizume, T., Ishii, N., Aida, T.: Self-assembled hexa-peri-hexabenzocoronene graphitic nanotube. Science 304, 1481–1483 (2004). doi:10.1126/science.1097789
Yamamoto, Y., Fukushima, T., Suna, Y., Ishii, N., Saeki, A., Seki, S., Tagawa, S., Taniguchi, M., Kawai, T., Aida, T.: Photoconductive coaxial nanotubes of molecularly connected electron donor and acceptor layers. Science 314, 1761–1764 (2006). doi:10.1126/science.1134441
Zhang, X., Chen, Z., Würthner, F.: Morphology control of fluorescent nanoaggregates by co-self-assembly of wedge- and dumbbell-shaped amphiphilic perylene bisimides. J. Am. Chem. Soc. 129, 4886–4887 (2007). doi:10.1021/ja070994u
McCormick, C.L. (ed.): Stimuli-Responsive Water Soluble and Amphiphilic Polymers. ACS, Washington, DC (2001)
Urban, M.W. (ed.): Stimuli-Responsive Polymeric Films and Coating. ACS, Washington, DC (2005)
Aathimanikandan, S.V., Savariar, E.N., Thayumanavan, S.: Temperature-sensitive dendritic micelles. J. Am. Chem. Soc. 127, 14922–14929 (2005). doi:10.1021/ja054542y
Basu, S., Vutukuri, D.R., Thayumanavan, S.: Homopolymer micelles in heterogeneous solvent mixtures. J. Am. Chem. Soc. 127, 16794–16795 (2005). doi:10.1021/ja056042a
Luisi, P.L., Walde, P. (eds.): Giant Vesicles. Wiley-VCH, Chichester (2000)
Shioi, A., Hatton, T.A.: Model for formation and growth of vesicles in mixed anionic/cationic (SOS/CTAB) surfactant systems. Langmuir 18, 7341–7348 (2002). doi:10.1021/la020268z
Li, Z., Hillmyer, M.A., Lodge, T.P.: Laterally nanostructured vesicles, polygonal bilayer sheets, and segmented wormlike micelles. Nano Lett. 6, 1245–1249 (2006). doi:10.1021/nl0608700
Acknowledgement
The author thanks the organizing committee of Host-Guest and Supramolecular Chemistry Society, Japan for giving him the HGCS Japan Award of Excellence 2008 and the opportunity of writing this review. This work was supported by Grant-in-Aid for Young Scientists (B) (No. 17750137, 19750122) and Scientific Research in a Priority Area “Super-Hierarchical Structures” (No. 18039038, 19022036) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Izumi Science and Technology Foundation, Iketani Science Technology Foundation, Mitsubishi Chemical Corporation Fund, Kumagai Foundation for Science and Technology, Nissan Science Foundation, Saneyoshi Scholarship Foundation, the Japan Securities Scholarship Foundation, the Science and Technology Foundation of Japan, Shorai Foundation for Science and Technology, and the Kao Foundation for Arts and Sciences, the “Academic Frontier” Project for Private Universities, namely the matching fund subsidy from the MEXT, 2003–2007, and the Ritsumeikan Global Innovation Research Organization (R-GIRO) project, 2008–2013. The author thanks Prof. Atsuhiro Osuka, Dr. Soji Shimizu (Tohoku University), Dr. Shigeki Mori (Tokyo University), Mr. Yasuhide Inokuma, and Mr. Shohei Saito, Kyoto University, for the X-ray analyses, Dr. Takashi Nakanishi, National Institute for Materials Science (NIMS) and Max Planck Institute of Colloids and Interfaces, for various measurements of organized structures, Prof. Myongsoo Lee and Ms. Eunji Lee, Yonsei University, for cryo-TEM measurements, Prof. Hitoshi Tamiaki, Ritsumeikan University, for helpful discussions, and all the group members.
This is a paper selected for “HGCS Japan Award of Excellence 2008”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maeda, H. Acyclic oligopyrroles as building blocks of supramolecular assemblies. J Incl Phenom Macrocycl Chem 64, 193–214 (2009). https://doi.org/10.1007/s10847-009-9568-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-009-9568-z