Abstract
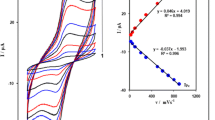
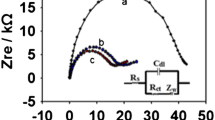
The influence of modifications of the mercuric oxide on the voltammetric properties of mercury film carbon paste electrodes was studied. The mercury film was formed electrochemically from the bulk red or yellow mercuric oxide-modified carbon paste electrodes. Differential pulse anodic stripping voltammetry and optical microscopy in polarised light were used to characterise the properties of the mercury films. The results were compared with those obtained using the conventional preparation of mercury-plated carbon paste electrodes when the mercury film is deposited on the surface of the electrode by reduction of Hg(II) ions in solution utilising a sufficient negative potential. It was shown that the mercury film formed from the yellow modification of the mercuric oxide provides better voltammetric characteristics than the red one owing to the high distribution of its small particles, i.e. the mercury droplets after electrochemical treatment. Such a mercury film has similar properties to those of a mercury film generated from solution.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 06 December 1999 / Accepted: 16 March 2000
Rights and permissions
About this article
Cite this article
Metelka, R., Vytřas, K. & Bobrowski, A. Effect of the modification of mercuric oxide on the properties of mercury films at HgO-modified carbon paste electrodes. J Solid State Electrochem 4, 348–352 (2000). https://doi.org/10.1007/s100080000133
Issue Date:
DOI: https://doi.org/10.1007/s100080000133