Abstract.
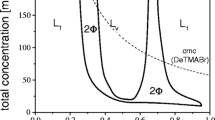
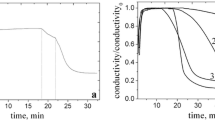
The gelation of mixed cationic/anionic surfactant vesicles of sodium dodecyl sulfate/didodecyldimethylammonium bromide and sodium dodecylbenzenesulfonate/cetyltrimethylammonium tosylate by hydrophobically modified sodium polyacrylate is studied rheologically. When the vesicles are cationically charged, mixtures with this anionic polyelectrolyte form precipitates. When the vesicles are anionically charged, however, these mixtures display a progression from a Maxwell fluid to a critical gel to a solidlike gel with increasing vesicle and/or polyelectrolyte concentration. Consideration of the viscous behavior with increasing vesicle and polymer volume fraction indicates that the gel network is formed by the bridging of the hydrophobically modified polymer between vesicles. The similarity between the gelation results for the two anionic systems suggests the results can be generalized to other similarly charged mixtures.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Ashbaugh, H.S., Boon, K. & Prud'homme, R.K. Gelation of "catanionic" vesicles by hydrophobically modified polyelectrolytes. Colloid Polym Sci 280, 783–788 (2002). https://doi.org/10.1007/s00396-002-0702-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00396-002-0702-3