Abstract
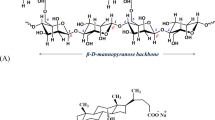
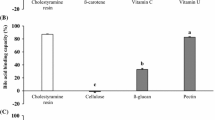
The interactions between pectin and bile acids (BA) were investigated after in vitro incubation at 37°C in the pH range of 5.0 – 8.0, separation of the non-absorbed BA using membrane filtration and their estimation by HPLC and HPTLC. In general, the interactions increased with the degree of esterification and the molecular weight (viscosity) of the pectin. They were reduced if the carboxyl groups in the polysaccharide molecules are arranged in a blockwise manner and in the presence of Ca2+. The highest interactions were found for unconjugated BA, followed by the corresponding glycine-conjugates. Taurine-conjugates showed the lowest interaction with pectin. Chenodeoxycholic acid and deoxycholic acid reacted more effectively than the trihydroxy bile acid cholic acid in the in vitro experiments. The results show that structural parameters of both BA and pectin play an important role in the extent of their interaction.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 28 November 1996 / Revised version: 20 January 1997
Rights and permissions
About this article
Cite this article
Dongowski, G. Effect of pH on the in vitro interactions between bile acids and pectin. Z Lebensm Unters Forsch 205, 185–192 (1997). https://doi.org/10.1007/s002170050149
Issue Date:
DOI: https://doi.org/10.1007/s002170050149