Abstract.
The comparability of current microcystin analysis methods has been evaluated in an international intercomparison exercise. The focus was on the analysis of microcystins by high-performance liquid chromatography coupled with ultraviolet or photodiode-array detection (HPLC–PDA/UV), currently the most widespread method for microcystin analysis, but the exercise was open for other methods such as enzyme-linked immunosorbent assay (ELISA), protein phosphatase inhibition assay (PPA) and high-performance liquid chromatography coupled with mass spectrometry (HPLC–MS).
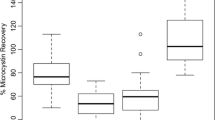
Thirty-one laboratories from 13 countries participated in the study. For a microcystin-LR (MC-LR) standard solution (S1) of undisclosed quantity, and for a field sample (S3) from a natural cyanobacterial bloom, repeatabilities between 4 and 15% and reproducibilities between 24 and 49% were obtained. No significant differences between single methods were found for S1 and S3, except for a significantly higher repeatability value of ELISA for S1. However, the analysis of microcystins in the field sample (S3) by HPLC–PDA/UV was significantly more variable than for the standard solution (S1). Both the extraction and the analysis of the microcystins appeared to contribute to this variability.
It is concluded that standard MC-LR (S1) can be measured with adequate precision by all participating laboratories independently of the method used. With respect to the different methods used the results for the field sample can also be regarded as satisfactory, but clearly showed the need for improvement by standardisation between laboratories. Furthermore, quantification with in-house standards compared to quantification using the supplied MC-LR standard indicated that routine microcystin analysis in laboratories may be also influenced by the variability of available standards, emphasising the need for the production of certified reference materials (CRM).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Fastner, J., Codd, G.A., Metcalf, J.S. et al. An international intercomparison exercise for the determination of purified microcystin-LR and microcystins in cyanobacterial field material. Anal Bioanal Chem 374, 437–444 (2002). https://doi.org/10.1007/s00216-002-1520-7
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00216-002-1520-7