Abstract.
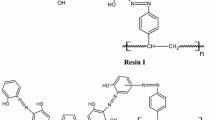
Amberlite XAD-4 has been functionalized by coupling it with 5-aminoquinoline-8-ol after acetylation. The resulting resin has been characterized by elemental analysis and IR spectra and has been used for preconcentrating uranyl ions prior to its determination by spectrophotometry. The optimum pH value for quantitative sorption is 4–6, and desorption can be achieved by using 5 mL of 1 mol L–1 HCl. The sorption capacity of the resin is 11.5 mmol g–1. The effect of various cations and anions on the preconcentration of uranium in conjunction with the determination procedure has been studied and we have found that none of the ions interfere except thorium. The enrichment factor for preconcentration of uranium was found to be 200. Ten replicate determinations of 40 µg of uranium present in 1 L of sample gave a mean absorbance of 0.185 with a relative standard deviation of 2.64%. The detection limit corresponding to three times the standard deviation of the bank was found to be 2 µg L–1. The validation of the developed preconcentration procedure was carried out by successfully analyzing standard marine sediment reference material. The uranyl content of sediment and soils is estimated by spectrophotometry after its preconcentration with the above chelating resin.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Gladis, M.J., Prasada Rao, T. Quinoline-8-ol-immobilized Amberlite XAD-4: Synthesis, characterization, and uranyl ion uptake properties suitable for analytical applications. Anal Bioanal Chem 373, 867–872 (2002). https://doi.org/10.1007/s00216-002-1387-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-002-1387-7