Abstract
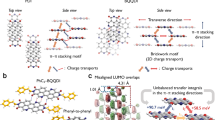
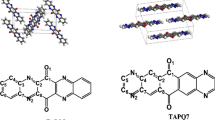
The charge transport and photophysical properties of N-heteroquinones, which can function as n-type organic semiconductors in organic field-effect transistors (OFETs) with high electron mobility, were systematically investigated using hopping model, band theory, and time-dependent density functional theory (TDDFT). The calculated absorption spectra and electron mobility are in good agreement with experimental results. To the studied compounds, subtle structural modifications can greatly reduce the reorganization energy. There are two main kinds of intermolecular interaction forces of the studied compounds in the crystal, which result from intermolecular π–π and hydrogen bonds interactions, respectively. The results of hopping model show that the electron transport properties are mainly determined by pathways containing intermolecular π–π interactions, and hole transport properties are mainly determined by pathways containing intermolecular hydrogen bonds from the standpoint of transfer integral. Moreover, electronic transfer integral value increases with the enhancement of intermolecular overlap corresponding to the overlap extent of π–π packing. Hole transfer integral value decreases with decreasing the number of hydrogen bonds. This means that charge transport properties can be efficiently tuned by controlling the relative positions of the molecules and the number of hydrogen bonds. The analysis of band structure also supports the conclusion of hopping model.




Similar content being viewed by others
References
Yoshiro Y (2009) Sci Technol Adv Mater 10:024313
Newman CR, Frisbie CD, da Silva Filho DA, Bredas JL, Ewbank PC, Mann KR (2004) Chem Mater 16:4436–4451
Zaumseil J, Sirringhaus H (2007) Chem Rev 107:1296–1323
Yamashita Y (2009) Chem Lett 38:870–875
Allard S, Forster M, Souharce B, Thiem H, Scherf U (2008) Angew Chem Int Ed 47:4070–4098
Mayer AC, Scully SR, Hardin BE, Rowell MW, McGehee MD (2007) Mater Today 10:28–33
Anthony JE (2006) Chem Rev 106:5028–5048
Murphy AR, Fréchet JMJ (2007) Chem Rev 107:1066–1096
Wen Y, Liu Y (2010) Adv Mater 22:1331–1345
de Leeuw DM, Simenon MMJ, Brown AR, Einerhand REF (1997) Synth Met 87:53–59
Tang ML, Reichardt AD, Wei P, Bao Z (2009) J Am Chem Soc 131:5264–5273
Usta H, Risko C, Wang Z, Huang H, Deliomeroglu MK, Zhukhovitskiy A, Facchetti A, Marks TJ (2009) J Am Chem Soc 131:5586–5608
Jones BA, Facchetti A, Wasielewski MR, Marks TJ (2007) J Am Chem Soc 129:15259–15278
Coropceanu V, Cornil J, da Silva Filho DA, Olivier Y, Silbey R, Brédas JL (2007) Chem Rev 107:926–952
Brédas JL, Calbert JP, da Silva Filho DA, Cornil J (2002) Proc Natl Acad Sci USA 99:5804–5809
Ando S, Murakami R, Nishida JI, Tada H, Inoue Y, Tokito S, Yamashita Y (2005) J Am Chem Soc 127:14996–14997
Bao Z, Lovinger AJ, Dodabalapur A (1996) Appl Phys Lett 69:3066–3068
Nalwa H (1997) Handbook of organic conductive molecules and polymers, vol 1 C4. Wiley, New York
Balzani V, Venturi M, Credi A (2003) Molecular devices and machines: a journey into the nano world; Vch Verlagsgesellschaft Mbh
Winkler M, Houk KN (2007) J Am Chem Soc 129:1805–1815
Miao S, Appleton AL, Berger N, Barlow S, Marder SR, Hardcastle KI, Bunz UHF (2009) Chem Eur J 15:4990–4993
Tang Q, Liang Z, Liu J, Xu J, Miao Q (2010) Chem Comm 46:2977–2979
Bredas JL, Beljonne D, Coropceanu V, Cornil J (2004) Chem Rev 104:4971–5004
Peng Q, Yi Y, Shuai Z, Shao J (2007) J Am Chem Soc 129:9333–9339
Zhang J, Frenking G (2004) J Phys Chem A 108:10296–10301
Yang GC, Su T, Shi SQ, Su ZM, Zhang HY, Wang Y (2007) J Phys Chem A 111:2739–2744
Yang GC, Liao Y, Su ZM, Zhang HY, Wang Y (2006) J Phys Chem A 110:8758–8762
Gao HZ, Qin CS, Zhang HY, Wu SX, Su ZM, Wang Y (2008) J Phys Chem A 112:9097–9103
Wu J, Wu SX, Geng Y, Yang GC, Shabbir M, Jin JL, Liao, Su ZM, (2010) Theor Chem Acc. doi:10.1007/s00214-0010-00730-x
Becke AD (1993) J Che Phys 98:1372–1377
Lee C, Yang W, Parr RG (1998) Phys Re B 37:785
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) GAUSSIAN 03, (Revision C.02). Gaussian, Inc., Pittsburgh
Nelsen SF, Trieber DA, Ismagilov RF, Teki Y (2001) J Am Chem Soc 123:5684–5694
Nelsen SF, Blomgren F (2001) J Org Chem 66:6551–6559
Sakanoue K, Motoda M, Sugimoto M, Sakaki S (1999) J Phys Chem A 103:5551–5556
Malagoli M, Brédas JL (2000) Chem Phys Lett 327:13–17
Li XY, Tong J, He FC (2000) Chem Phys 260:283–294
Lin BC, Cheng CP, Lao ZP (2003) J Phys Chem A 107:5241–5251
Marcus RA (1985) Biochim Biophys Acta 811:265
Marcus RA (1993) Rev Mod Phys 65:599
Balzani V, Juris A, Venturi M, Campagna S, Serroni S (1996) Chem Rev 96:759–834
Deng WQ, Goddard WA (2004) J Phys Chem B 108:8614–8621
Song Y, Di CA, Yang X, Li S, Xu W, Liu Y, Yang L, Shuai Z, Zhang D, Zhu D (2006) J Am Chem Soc 128:15940–15941
Schein LB, McGhie AR (1979) Phys Rev B 20:1631
Yang XD, Li Q, Shuai ZG (2007) Nanotechnology 18:424029
Yang XD, Wang L, Wang C, Long W, Shuai Z (2008) Chem Mater 20:3205–3211
Kresse G, Furthmüller J (1996) Comput Mater Sci 6:15–50
Kresse G, Hafner J (1993) Phys Rev B 47:558
Kresse G, Hafner J (1994) Phys Rev B 49:14251
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Blöchl PE (1994) Phys Rev B 50:17953
Kresse G, Joubert D (1999) Phys Rev B 59:1758
Monkhorst HJ, Pack JD (1976) Phys Rev B 13:5188
Malagoli M, Coropceanu V, Filho DA, Bredas JL (2004) J Chem Phys 120:7490–7496
da Silva Filho DA, Coropceanu V, Fichou D, Gruhn NE, Bill TG, Gierschner J, Cornil J, Brédas JL (2007) Philos Trans R Soc London A 365:1435–1452
Gruhn NE, da Silva Filho DA, Bill TG, Malagoli M, Coropceanu V, Kahn A, Brédas JL (2002) J Am Chem Soc 124:7918–7919
Coropceanu V, Malagoli M, da Silva Filho DA, Gruhn NE, Bill TG, Brédas JL (2002) Phys Rev Lett 89:275503
Lin BC, Cheng CP, You ZQ, Hsu CP (2004) J Am Chem Soc 127:66–67
Valeev EF, Coropceanu V, da Silva Filho DA, Salman S, Brédas JL (2006) J Am Chem Soc 128:9882–9886
Hutchison GR, Ratner MA, Marks TJ (2005) J Am Chem Soc 127:16866–16881
Troisi A, Orlandi G (2001) Chem Phys Lett 344:509–518
Irfan A, Zhang JP, Chang YF (2009) Chem Phys Lett 483:143–146
Li L, Tang Q, Li H, Yang X, Hu W, Song Y, Shuai Z, Xu W, Liu Y, Zhu D (2007) Adv Mater 19:2613–2617
Acknowledgments
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (Project No. 20903020; 20703008), Chang Jiang Scholars Program (2006), Program for Changjiang Scholars and Innovative Research Team in University (IRT0714), National Basic Research Program of China (973 Program—2009CB623605), the Science and Technology Development Project Foundation of Jilin Province (20090146), the Training Fund of NENU’s Scientific Innovation Project (NENU-STC08005 and -STC08012), The Project-sponsored by SRF for ROCS, SEM and Open Project Program of State Key Laboratory of Supramolecular Structure and Materials, Jilin University. And we also thank Patrik Callis (MSU) for supplying the Bozesuite program. Science Foundation for Young Teachers of Jilin Agricultural University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, G., Si, Y., Geng, Y. et al. Charge transport and electronic properties of N-heteroquinones: quadruple weak hydrogen bonds and strong π–π stacking interactions. Theor Chem Acc 128, 257–264 (2011). https://doi.org/10.1007/s00214-010-0841-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-010-0841-4