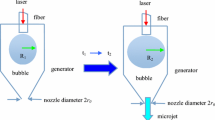
Abstract
The high-speed liquid (water) jet has distinctive characteristics in surgical applications, such as tissue dissection without thermal damage and small blood vessel preservation, that make it advantageous over more conventional instruments. The continuous pressurized jet has been used since the first medical application of water jets to liver surgery in the 1980s, but exhibited drawbacks partly related to the excess water supply required and unsuitability for application to microsurgical instruments intended for deep, narrow lesions (endoscopic instrumentation and catheters) due to limitations in miniaturization of the device. To solve these issues, we initiated work on the pulsed micro-liquid jet. The idea of the pulsed micro-liquid jet originated from the observation of tissue damage by shock/bubble interactions during extracorporeal shock wave lithotripsy and evolved into experimental application for recanalization of cerebral embolisms in the 1990s. The original method of generating the liquid jet was based on air bubble formation and microexplosives as the shock wave source, and as such could not be applied clinically. The air bubble was replaced by a holmium:yttrium-aluminum-garnet (Ho:YAG) laser-induced bubble. Finally, the system was simplified and the liquid jet was generated via irradiation from the Ho:YAG laser within a liquid-filled tubular structure. A series of investigations revealed that this pulsed laser-induced liquid jet (LILJ) system has equivalent dissection and blood vessel preservation characteristics, but the amount of liquid usage has been reduced to less than 2 \(\upmu \)l per shot and can easily be incorporated into microsurgical, endoscopic, and catheter devices. As a first step in human clinical studies, we have applied the LILJ system for the treatment of skull base tumors through the transsphenoidal approach in 9 patients (7 pituitary adenomas and 2 chordomas), supratentorial glioma (all high grade glioma) in 8 patients, including one with fine perforating vessel involvement, and cerebrovascular disease (1 arteriovenous malformation and 2 intracerebral hemorrhages) in 3 patients. Precise dissection and mass reduction of the tumor were obtained in all tumor cases except for one chordoma with significant fibrosis. Small arteries down to 100 \(\upmu \hbox {m}\) were preserved, allowing subsequent microsurgical devascularization. Veins were also preserved occasionally. The arachnoid membrane and the tumor capsule were resistant to the LILJ except for one case with prolonged exposure. No complications related to use of the LILJ system were observed. No disturbance of the surgical field by splashing, aerosol, or dissemination of pathological tissue occurred with placement of the optimal suction system. The Ho:YAG LILJ system enhances the advantages of commercialized pressure-driven continuous liquid jet instrumentation in terms of small vessel preservation and accessibility in confined spaces for minimally invasive neurosurgery, and solves some of the drawbacks involved with excessive liquid use and size.










Similar content being viewed by others
References
Aroussi, A.A., Sami, I.M., Lequerrier, A., Verhoye, J.P.: The blower: a useful tool to complete thrombectomy of the mechanical prosthetic valve. Ann. Thorac. Surg. 81, 1911–1912 (2006)
Ito, H., Muka, H., Kitamura, A.: Stereotactic aqua stream and aspirator for removal of intracerebral hematoma. Stereotact. Funct. Neurosurg. 54–55, 457–460 (1990)
Iwamoto, N., Kusaka, M., Tsurutani, T., Kameda, H., Ito, H.: Ultrasound imaging for stereotactic evacuation of hypertension-associated intracerebral hematomas with aqua-stream and aspiration. Stereotact. Funct. Neurosurg. 60, 194–204 (1993)
Izumi, R., Yabushita, K., Shimizu, K., Yagi, M., Yamaguchi, A., Konishi, K., Nakagawa, T., Miyazaki, I.: Hepatic resection using a water jet dissector. Surg. Today 23, 31–35 (1993)
Jakob, S., Kehler, U., Reusche, E., Friedrich, H.J., Arnold, H.: Endoscopic use of the water jet dissector in the cerebral ventricle system—an experimental study. Zentralbl. Neurochir. 61, 14–21 (2000) (in German)
Kaduk, W.M., Stengel, B., Pohl, A., Nizze, H., Gundlach, K.K.: Hydro-jet cutting: a method for selective surgical dissection of nerve tissue. An experimental study on the sciatic nerve of rats. J. Craniomaxillofac. Surg 27, 327–330 (1999)
Kroh, M., Hall, R., Udomsawaengsup, S., Smith, A., Yerian, L., Chand, B.: Endoscopic water jets used to ablate Barrett’s esophagus: preliminary results of a new technique. Surg. Endosc. 22, 2498–2502 (2008)
Lorenz, K.J., Kresz, A., Maier, H.: Hydrodissection for tonsillectomy. Results of a pilot study—intraoperative blood loss, postoperative pain symptoms and risk of secondary hemorrhage. HNO 53, 423–427 (2005) (in German)
Mukai, H., Yamashita, J., Kitamura, A., Ito, H.: Stereotactic aqua-stream and aspirator in the treatment of intracerebral hematoma. An experimental study. Stereotact. Funct. Neurosurg. 57, 221–227 (1991)
Oertel, J., Gaab, M.R., Knapp, A., Essig, H., Warzok, R., Piek, J.: Water jet dissection in neurosurgery: experimental results in the porcine cadaveric brain. Neurosurgery 52, 153–159 (2003)
Oertel, J., Gaab, M.R., Piek, J.: Waterjet resection of brain metastases—first clinical results with 10 patients. Eur. J. Surg. Oncol. 29, 407–414 (2003)
Oertel, J., Gaab, M.R., Warzok, R., Piek, J.: Waterjet dissection in the brain: review of the experimental and clinical data with special reference to meningioma surgery. Neurosurg. Rev. 26, 168–174 (2003)
Oertel, J., Gaab, M.R., Pillich, D.T., Schroeder, H.W., Warzok, R., Piek, J.: Comparison of waterjet dissection and ultrasonic aspiration: an in vivo study in the rabbit brain. J. Neurosurg. 100, 498–504 (2004)
Oertel, J., Gaab, M.R., Schiller, T., Schroeder, H.W., Warzok, R., Piek, J.: Towards waterjet dissection in neurosurgery: experimental in-vivo results with two different nozzle types. Acta. Neurochir. (Wien) 146, 713–720 (2004)
Oertel, J., Wagner, W., Piek, J., Schroeder, H., Gaab, M.: Waterjet dissection of gliomas-experience with 51 procedures. Minim. Invasive Neurosurg. 47, 154–159 (2004)
Oertel, J., Gaab, M.R., Runge, U., Schroeder, H.W., Piek, J.: Waterjet dissection versus ultrasonic aspiration in epilepsy surgery. Neurosurgery 56(1 Suppl), 142–146 (2005)
Oertel, J., Gen, M., Krauss, J.K., Zumkeller, M., Gaab, M.R.: The use of waterjet dissection in endoscopic neurosurgery. Technical note. J. Neurosurg. 105, 928–931 (2006)
Papachristou, D.N., Barters, R.: Resection of the liver with a water jet. Br. J. Surg. 69, 93–94 (1982)
Piek, J., Wille, C., Warzok, R., Gaab, M.R.: Waterjet dissection of the brain—experimental and first clinical results. Technical note. J. Neurosurg. 89, 861–864 (1998)
Piek, J., Oertel, J., Gaab, M.R.: Waterjet dissection in neurosurgical procedures—clinical results in 35 patients. J. Neurosurg. 96, 690–696 (2002)
Rau, H.G., Duessel, A.P., Wurzbacher, S.: The use of water-jet dissection in open and laparoscopic liver resection. HPB (Oxford) 10, 275–280 (2008)
Rennekampff, H.O., Schaller, H.E., Wisser, D., Tenenhaus, M.: Debridement of burn wounds with a water jet surgical tool. Burns 32, 64–69 (2006)
Schurr, M.O., Wehrmann, M., Kunert, W., Melzer, A., Lirici, M.M., Trapp, R., Kanehira, E., Buess, G.: Histologic effects of different technologies for dissection in endoscopic surgery: Nd: YAG laser, high frequency and water-jet. Endosc. Surg. Allied Technol. 2, 195–201 (1994)
Shekarriz, B., Upadhyay, J., Jewett, M.A.: Nerve-sparing retroperitoneal lymphadenectomy using hydro-jet dissection: initial experience. J. Endourol. 18, 273–276 (2004)
Shekarriz, B.: Hydro-Jet technology in urologic surgery. Expert Rev. Med. Devices 2, 287–291 (2005)
Shekarriz, H., Shekarriz, B., Burk, C.G., Kujath, P., Bruch, H.P.: Hydro-jet-assisted pneumonectomy: a new technique in a porcine model. J. Laparoendosc. Adv. Surg. Tech. A 12, 371–376 (2002)
Shekarriz, H., Shekarriz, B., Kujath, P., Eckmann, C., Burk, C., Comman, A., Bruch, H.P.: Hydro-Jet assisted laparoscopic cholecystectomy: a prospective randomized clinical study. Surgery 133, 635–640 (2003)
Siegert, R., Magritz, R., Jurk, V.: Water-jet dissection in parotid surgery—initial clinical results. Laryngorhinootologie 79, 780–784 (2000) (in German)
Terzis, A.J., Nowak, G., Rentzsch, O., Arnold, H., Diebold, J., Baretton, G.: A new system for cutting brain tissue preserving vessels: water jet cutting. Br. J. Neurosurg. 3, 361–366 (1989)
Tschan, C., Gaab, M.R., Krauss, J.K., Oertel, J.: Waterjet dissection of the vestibulocochlear nerve: an experimental study. J. Neurosurg. 110, 656–661 (2009)
Hirano, T., Komatsu, M., Ezura, M., Uenohara, H., Takahashi, A., Takayama, K., Yoshimoto, T.: Formation of a liquid jet by interaction between a laser-induced bubble and shock wave. Interv. Neuroradiol. 7(Suppl 1), 35–40 (2001)
Hirano, T., Komatsu, M., Saeki, T., Uenohara, H., Takahashi, A., Takayama, K., Yoshimoto, T.: Enhancement of fibrinolytics with a laser-induced liquid jet. Lasers Surg. Med. 29, 360–368 (2001)
Hirano, T., Komatsu, M., Uenohara, H., Takahashi, A., Takayama, K., Yoshimoto, T.: A novel method of drug delivery for fibrinolysis with Ho: YAG laser-induced liquid jet. Lasers Med. Sci. 17, 165–172 (2002)
Hirano, T., Nakagawa, A., Uenohara, H., Ohyama, H., Jokura, H., Takayama, K., Shirane, R.: Pulsed liquid jet dissector using holmium: YAG laser—a novel neurosurgical device for brain incision without impairing vessels. Acta. Neurochir. (Wien) 145, 401–406 (2003)
Hirano, T., Nakagawa, A., Uenohara, H., Jokura, H., Takayama, K., Shirane, R.: Shock waves concomitant with a Ho:YAG laser-induced liquid jet: Possibility of application to a medical device. Neurosonology 16, 33–36 (2003) (in Japanese)
Hirano, T., Uenohara, H., Komatsu, M., Nakagawa, A., Satoh, M., Ohyama, H., Takayama, K., Yoshimoto, T.: Holmium: YAG laser-induced liquid jet dissector: a novel prototype device for dissecting organs without impairing vessels. Minim. Invasive Neurosurg. 46, 121–125 (2003)
Hirano, T., Nakagawa, A., Ohki, T., Uenohara, H., Takayama, K., Tominaga, T.: A laser-induced liquid jet catheter system: a novel endovascular device for rapid and reliable fibrinolysis in acute cerebral embolism. Minim. Invasive Neurosurg. 51, 324–328 (2008)
Nakagawa, A., Hirano, T., Komatsu, M., Sato, M., Uenohara, H., Ohyama, H., Kusaka, Y., Shirane, R., Takayama, K., Yoshimoto, T.: Holmium: YAG laser-induced liquid jet knife: possible novel method for dissection. Lasers Surg. Med. 31, 129–135 (2002)
Nakagawa, A., Hirano, T., Jokura, H., Uenohara, H., Ohki, T., Hashimoto, T., Menezes, V., Sato, Y., Kusaka, Y., Ohyama, H., Saito, T., Takayama, K., Shirane, R., Yoshimoto, T.: Pulsed holmium: yttrium-aluminum-garnet laser-induced liquid jet as a novel dissection device in neuroendoscopic surgery. J. Neurosurg. 101, 145–150 (2004)
Nakagawa, A., Kumabe, T., Kanamori, M., Saito, R., Hirano, T., Takayama, K., Tominaga, T.: Clinical application of pulsed laser-induced liquid jet: preliminary report in glioma surgery. No Shinkei. Geka. 36, 1005–1010 (2008) (in Japanese)
Ohki, T., Nakagawa, A., Hirano, T., Hashimoto, T., Menezes, V., Jokura, H., Uenohara, H., Sato, Y., Saito, T., Shirane, R., Tominaga, T., Takayama, K.: Experimental application of pulsed Ho: YAG laser-induced liquid jet as a novel rigid neuroendoscopic dissection device. Lasers Surg. Med. 34, 227–234 (2004)
Sato, Y., Nakagawa, A., Hirano, T., Ohki, T., Uenohara, H., Takayama, K., Tominaga, T.: Pulsed laser-induced liquid jet microcatheter system for rapid and reliable fibrinolysis in acute cerebral embolisms: experiments on safety and preliminary application in porcine cranial vessels. Minim. Invasive Neurosurg. 50, 212–218 (2007)
Tominaga, T., Nakagawa, A., Hirano, T., Sato, J., Kato, K., Takayama, K.: Application of underwater shock wave and laser-induced liquid jet to neurosurgery. Shock Waves 15, 55–67 (2006)
Arora, A., Hakim, I., Baxter, J., Rathnasingham, R., Srinivasan, R., Fletcher, D.A., Mitragotri, S.: Needle-free delivery of macromolecules across the skin by nanoliter-volume pulsed microjets. Proc. Natl. Acad. Sci. USA 104, 4255–4260 (2007)
Fletcher, D.A., Planker, D.V.: Pulsed liquid microjet for microsurgery. Appl. Phys. Lett. 78, 1933–1935 (2001)
Fletcher, D.A., Palanker, D.V., Huie, P., Miller, J., Marmor, M.F., Blumenkranz, M.S.: Intravascular drug deliver with a pulsed liquid microjet. Arch. Ophthalmol. 120, 1206–1208 (2002)
Miller, J.M., Palanker, D.V., Vankov, A., Marmor, M.F., Blumenkranz, M.S.: Precision and safety of the pulsed electron avalanche knife in vitreoretinal surgery. Arch. Ophthalmol. 121, 871–877 (2003)
Kunikata, H., Tanaka, Y., Aizawa, N., Nakagawa, A., Tominaga, T., Nakazawa, T.: Experimental application of piezoelectric actuator-driven pulsed water jets in retinal vascular surgery. Transl. Vis. Sci. Technol. 3, 10 (2014)
Yamada, M., Nakano, T., Sato, C., Nakagawa, A., Fujishima, F., Kawagishi, N., Nakanishi, C., Sakurai, T., Miyata, G., Tominaga, T., Ohuchi, N.: The dissection profile and mechanism of tissue-selective dissection of the piezo actuator-driven pulsed water jet as a surgical instrument: laboratory investigation using swine liver. Eur. J. Surg. Res. 53, 61–72 (2014)
Yamashita, S., Kamiyama, Y., Nakagawa, A., Kaiho, Y., Tominaga, T., Arai, Y.: Novel pulsed water jet system permits off-clamp partial nephrectomy in swine. Int. J. Urol. 21, 1181–1182 (2014)
Endo, T., Takahashi, Y., Nakagawa, A., Niizuma, K., Fujimura, M., Tominaga, T.: Use of actuator-driven pulsed water jet in brain and spinal cord cavernous malformations resection. Neurosurgery 11(Suppl 3), 394–403 (2015)
Kawaguchi, T., Endo, T., Nakagawa, A., Fujimura, M., Sonoda, Y., Tominaga, T.: Ventricle wall dissection and vascular preservation with the pulsed water jet device: novel tissue dissector for flexible neuroendoscopic surgery. J. Neurosurg. 124, 817–822 (2016)
Nakanishi, C., Nakano, T., Nakagawa, A., Sato, C., Yamada, M., Kawagishi, N., Tominaga, T., Ohuchi, N.: Evaluation of a newly developed piezo actuator-driven pulsed water jet system for liver resection in a surviving swine animal model. Biomed. Eng. Online 15, 9 (2016)
Nakano, T., Sato, C., Sakurai, T., Kamei, T., Nakagawa, A., Ohuchi, N.: Use of water jet instruments in gastrointestinal endoscopy. World J. Gastrointest. Endosc. 8, 122–127 (2016)
Chaussy, C., Brendel, W., Schmiedt, E.: Extracorporeally induced destruction of kidney stones by shock wave. Lancet 13, 1265–1268 (1980)
Delius, M., Enders, G., Xuan, Z., Liebich, H., Brendel, W.: Biological effects of shock waves: kidney damage by shock waves in dogs—dose dependence. Ultrasound Med. Biol. 14, 117–122 (1988)
Delius, M., Jordan, M., Eizenhoefer, H., Marlinghaus, E., Heine, G., Liebich, H., Brendel, W.: Biological effects of shock waves: kidney haemorrhage by shock waves in dogs—administration rate dependence. Ultrasound Med. Biol. 14, 689–694 (1988)
Delius, M.: Medical applications and bioeffects of extracorporeal shock waves. Shock Waves 4, 55–72 (1994)
Delius, M.: Twenty years of shock wave research at the Institute for Surgical Research. Eur. Surg. Res. 34, 30–36 (2002)
Delius, M., Jordan, M., Liebich, H.G., Brendel, W.: Biological effect of shock waves on the liver and gallbladder wall of dogs—administration rate dependence. Ultrasound Med. Biol. 16, 459–466 (1990)
Delius, M., Enders, G., Heine, G., Stark, J., Remberger, K., Brendel, W.: Biological effects of shock waves: lung hemorrhage by shock waves in dogs—pressure dependence. Ultrasound Med. Biol. 13, 61–67 (1987)
Delius, M., Denk, R., Berding, C., Liebich, H., Jordan, M., Brendel, W.: Biological effects of shock waves: cavitation by shock waves in piglet liver. Ultrasound Med. Biol. 16, 467–472 (1990)
Delius, M., Gambihler, S.: Sonographic imaging of extracorporeal shock wave effects in the liver and gallbladder of dogs. Digestion 26, 55–60 (1992)
Prat, F., Ponchon, T., Berger, F., Chapelon, Y., Gagnon, P., Cathignol, D.: Hepatic lesions in the rabbit induced by acoustic cavitation. Gastroenterology 100, 1345–1350 (1991)
Schelling, G., Delius, M., Gschwender, M., Grafe, P., Gambihler, S.: Extracorporeal shock waves stimulate frog sciatic nerve indirectly via a cavitation-mediated mechanism. Biophys. J. 66, 133–140 (1994)
Williams, A.R., Delius, M., Miller, D.L., Schwarze, W.: Investigation of cavitation in flowing media by lithotripter shock waves both in vitro and vivo. Ultrasound Med. Biol. 15, 53–60 (1989)
Arndt, R.E.: Cavitation in fluid machinery and hydraulic structures. Annu. Rev. Fluid. Mech. 13, 273–328 (1981)
Kodama, T., Takayama, K.: Dynamic behavior of bubbles during extracorporeal shock-wave lithotripsy. Ultrasound Med. Biol. 24, 723–738 (1998)
Kodama, T., Uenohara, H., Takayama, K.: Innovative technology for tissue disruption by explosive-induced shock waves. Ultrasound Med. Biol. 24, 1459–1466 (1998)
Ohl, C.D., Ikink, R.: Shock-wave-induced jetting of micron-size bubbles. Phys. Rev. Lett. 90, 214502 (2003)
Ohl, C.D., Arora, M., Ikink, R., de Jong, N., Versluis, M., Delius, M., Lohse, D.: Sonoporation from jetting cavitation bubbles. Biophys. J. 91, 4285–4295 (2006)
Tachibana, K., Tachibana, S.: Albumin microbubble echo-contrast materials as an enhancer for ultrasound accelerated thrombolysis. Circulation 92, 1148–1150 (1995)
Takayama, K., Nagoya, H., Obara, T., Kuwahara, M.: Application of holographic interferometric studies of underwater shock wave focusing to medicine. Proc. SPIE. Int. Soc. Opt. Eng. 1801, 1029–1034 (1992)
Wolfrum, B., Mettin, R., Kurz, T., Lauterborn, W.: Observations of pressure-wave-excited contrast agent bubbles in the vicinity of cells. Appl. Phys. Lett. 81, 5060–5062 (2002)
Xi, X.F., Zhong, P.: Dynamic photoelastic study of the transient stress field in solids during shock wave lithotripsy. J. Acoust. Soc. Am. 109, 1226–1239 (2001)
Zhong, P., Xi, X., Zhu, S.L., Cocks, F.H., Preminger, G.M.: Recent developments in SWL physics research. J. Endourol. 13, 611–617 (1999)
Zhong, P., Zhou, Y., Zhu, S.L.: Dynamics of bubble oscillation in constrained media and mechanism of vessel rupture in SWL. Ultrasound Med. Biol. 27, 119–134 (2001)
Zhu, S.L., Dreyer, T., Liebler, M., Riedlinger, R., Preminger, G.M., Zhong, P.: Reduction of tissue injury in shock-wave lithotripsy by using an acoustic diode. Ultrasound Med. Biol. 30, 675–682 (2004)
Kodama, T., Takayama, K., Uenohara, H.: A new technology for revascularization of cerebral embolism using liquid jet impact. Phys. Med. Biol. 42, 2355–2367 (1997)
Kodama, T., Takayama, K., Nagayasu, N.: The dynamics of two air bubbles loaded by an underwater shock wave. J. Appl. Phys. 80, 5587–5592 (1996)
Kodama, T., Tatsuno, M., Sugimoto, S., Uenohara, H., Yoshimoto, T., Takayama, K.: Liquid jets, accelerated thrombolysis: a study for revascularization of cerebral embolism. Ultrasound Med. Biol. 25, 977–983 (1999)
Dear, J.P., Field, J.E.: A study of the collapse of arrays of cavities. J. Fluid Mech. 190, 409–425 (1988)
Asshauer, T., Rink, K., Delacretaz, G.: Acoustic transient generation by holmium-laser-induced cavitation bubbles. J. Appl. Phys. 76, 5007–5013 (1994)
Assahauer, T., Delacretaz, G., Jansen, E.D., Welch, A.J., Frenz, M.: Pulsed holmium laser ablation of tissue phantoms: correlation between bubble formation and acoustic transients. Appl. Phys. B 65, 647–657 (1997)
Hosseini, S.H., Menezes, V., Moosavi-Nejad, S., Ohki, T., Nakagawa, A., Tominaga, T., Takayama, K.: Development of shock wave assisted therapeutic devices and establishment of shock wave therapy. Minim. Invasive Ther. Allied Technol. 15, 230–240 (2006)
Jansen, E.D., van Leeuwen, T.G., Motamedi, M., Borst, C., Welch, A.J.: Temperature dependence of the absorption coefficient of water for midinfrared laser radiation. Lasers Surg. Med. 14, 258–268 (1994)
Nakagawa, A., Hirano, T., Kusaka, Y., Sato, M., Uenohara, H., Shirane, R., Takayama, K., Yoshimoto, T.: Biological effect of shock waves on rat brain: pathological evaluation by compact Ho:YAG-laser-induced cavitational shock wave generator. SPIE. Proc. 4948, 263–268 (2003)
Nakagawa, A., Kusaka, Y., Hirano, T., Saito, T., Shirane, R., Takayama, K., Yoshimoto, T.: Application of shock waves as a treatment modality in the vicinity of the brain and skull. J. Neurosurg. 99, 156–162 (2003)
Nakagawa, A., Fujimura, M., Kato, K., Okuyama, H., Hashimoto, T., Takayama, K., Tominaga, T.: Shock wave-induced brain injury in rat: novel traumatic brain injury animal model. Acta Neurochir. Suppl. 102, 421–424 (2008)
Kato, K., Fujimura, M., Nakagawa, A., Saito, A., Ohki, T., Takayama, K., Tominaga, T.: Pressure-dependent effect of shock waves on rat brain: induction of neuronal apoptosis mediated by a caspase-dependent pathway. J. Neurosurg. 106, 667–676 (2007)
Saito, T., Marumoto, M., Yamashita, H., Hosseini, S.H.R., Nakagawa, A., Hirano, T., Takayama, K.: Experimental and numerical studies of underwater shock wave attenuation. Shock Waves 13, 139–148 (2003)
Saito, T., Voinovich, P.A., Nakagawa, A., Hosseini, S.H.R., Takayama, K., Hirano, T.: On the efficiency of Gore-Tex layer for brain protection from shock wave damage in cranioplasty. Rev. Sci. Instrum. 75, 4789–4796 (2004)
Zivin, J.A., DeGirolami, U.: Spinal cord infarction: a highly reproducible stroke model. Stroke 11, 200–202 (1980)
Lin, R., Vora, N., Zaidi, S., Aleu, A., Jankowitz, B., Thomas, A., Gupta, R., Horowitz, M., Kim, S., Reddy, V., Hammer, M., Uchino, K., Wechsler, L.R., Jovin, T.: Mechanical approaches combined with intra-arterial pharmacological therapy are associated with higher recanalization rates than either intervention alone in revascularization of acute carotid terminus occlusion. Stroke 40, 2092–2097 (2009)
Seto, T., Takagi, K., Yoshida, K., Park, J.H., Yokota, S.: Development of high-power micropump using inertia effect of fluid for small-sized fluid actuator. J. Robot. Mechatron. 15, 128–135 (2003)
Sato, C., Nakano, T., Nakagawa, A., Yamada, M., Yamamoto, H., Kamei, T., Miyata, G., Sato, A., Fujishima, F., Nakai, M., Niinomi, M., Takayama, K., Tominaga, T., Satomi, S.: Experimental application of pulsed laser-induced water jet for endoscopic submucosal dissection: mechanical investigation and preliminary experiment in swine. Dig. Endosc. 25, 255–263 (2013)
Konishi, T., Moritake, K., Takebe, Y., Suwa, H., Handa, H.: Experimental study on the effect of ultrasonic surgical aspiration on blood flow and nerve function by use of ultrasonic Doppler flowmeter and auditory brainstem evoked response. No Shinkei. Geka. 12, 1069–1075 (1984) (in Japanese)
Matsumi, N., Matsumoto, K., Mishima, N., Moriyama, E., Furuta, T., Nishimoto, A., Taguchi, K.: Thermal damage threshold of brain tissue—histological study of heated normal monkey brains. Neurol. Med. Chir. (Tokyo) 34, 209–215 (1994)
Berger, M.S., Deliganis, A.V., Dobbins, J., Keles, G.E.: The effect of extent of resection on recurrence in patients with low grade cerebral hemisphere gliomas. Cancer 74, 1784–1791 (1994)
Keles, G.E., Chang, E.F., Lamborn, K.R., Tihan, T., Chang, C.J., Chang, S.M., Berger, M.S.: Volumetric extent of resection and residual contrast enhancement on initial surgery as predictors of outcome in adult patients with hemispheric anaplastic astrocytoma. J. Neurosurg. 105, 34–40 (2006)
Smith, J.S., Chang, E.F., Lamborn, K.R., Chang, S.M., Prados, M.D., Cha, S., Tihan, T., Vandenberg, S., McDermott, M.W., Berger, M.S.: Role of extent of resection in the long-term outcome of low-grade hemispheric gliomas. J. Clin. Oncol. 26, 1338–1345 (2008)
Hentschel, S.J., Lang, F.L.: Surgical resection of intrinsic insular tumors. Neurosurgery 57, 176–183 (2005)
Lang, F.F., Olansen, N.E., DeMonte, F., Gokaslan, Z.L., Holland, E.C., Kalhorn, C., Sawaya, R.: Surgical resection of intrinsic insular tumors: complication avoidance. J. Neurosurg. 95, 638–650 (2001)
Saito, R., Kumabe, T., Inoue, T., Takada, S., Yamashita, Y., Kanamori, M., Sonoda, Y., Tominaga, T.: Magnetic resonance imaging for preoperative identification of the lenticulostriate arteries in insular glioma surgery. J. Neurosurg. 111, 278–281 (2009)
Kumabe, T., Higano, S., Takahashi, S., Tominaga, T.: Ischemic complications associated with resection of opercular glioma. J. Neurosurg. 106, 263–269 (2007)
Couldwell, W.T., Weiss, M.H., Rabb, C., Liu, J.K., Apfelbaum, R.I., Fukushima, T.: Variations on the standard transsphenoidal approach to the sellar region, with emphasis on the extended approaches and parasellar approaches: surgical experience in 105 cases. Neurosurgery 55, 539–550 (2004)
Kassam, A., Snyderman, C.H., Mintz, A., Gardner, P., Carrau, R.L.: Expanded endonasal approach: the rostrocaudal axis. Part I. Crista galli to the sella turcica. Neurosurg. Focus 19, E3 (2005)
Kassam, A., Snyderman, C.H., Mintz, A., Gardner, P., Carrau, R.L.: Expanded endonasal approach: the rostrocaudal axis. Part II. Posterior clinoids to the foramen magnum. Neurosurg. Focus 19, E4 (2005)
Kassam, A.B., Gardner, P., Snyderman, C., Mintz, A., Carrau, R.: Expanded endonasal approach: fully endoscopic, completely transnasal approach to the middle third of the clivus, petrous bone, middle cranial fossa, and infratemporal fossa. Neurosurg. Focus 19, E6 (2005)
Kassam, A.B., Snyderman, C., Gardner, P., Carrau, R., Spiro, R.: The expanded endonasal approach: a fully endoscopic transnasal approach and resection of the odontoid process: technical case report. Neurosurgery 57(1 Suppl), E213 (2005)
Dusick, J.R., Esposito, F., Kelly, D.F., Cohan, P., DeSalles, A., Becker, D.P., Martin, N.A.: The extended direct endonasal transsphenoidal approach for nonadenomatous suprasellar tumors. J. Neurosurg. 102, 832–841 (2005)
Kitano, M., Taneda, M.: Extended transsphenoidal approach with submucosal posterior ethmoidectomy for parasellar tumors. Technical note. J. Neurosurg. 94, 999–1004 (2001)
Dusick, J.R., Esposito, F., Malkasian, D., Kelly, D.F.: Avoidance of carotid artery injuries in transsphenoidal surgery with the Doppler probe and micro-hook blades. Neurosurgery 60(4 Suppl 2), 322–328 (2007)
Laws Jr., E.R., Kern, E.B.: Complications of trans-sphenoidal surgery. Clin. Neurosurg. 23, 401–416 (1976)
Laws, E.R.Jr.: Vascular complications of transsphenoidal surgery. Pituitary 2, 163–170 (1999)
Rhoton Jr., A.L.: The sellar region. Neurosurgery 51(4 Suppl), S335–S374 (2002)
Ogawa, Y., Nakagawa, A., Takayama, K., Tominaga, T.: Pulsed laser-induced liquid jet for skull base tumor removal with vascular preservation through the transsphenoidal approach: a clinical investigation. Acta. Neurochirur. (Wien). 153, 823–830 (2011)
Ogawa, Y., Nakagawa, A., Washio, T., Arafune, T., Tominaga, T.: Tissue dissection before direct manipulation to the pathology with pulsed laser-induced liquid jet system in skull base surgery—preservation of fine vessels and maintained optic nerve function. Acta. Neurochirur. (Wien). 155, 1879–1886 (2013)
Nakagawa, A., Ogawa, Y., Amano, K., Ishii, Y., Tahara, S., Horiguchi, K., Kawamata, T., Yano, S., Arafune, T., Washio, T., Kuratsu, J., Saeki, N., Okada, Y., Teramoto, A., Tominaga, T.: Pulsed laser-induced liquid jet system for treatment of sellar and parasellar tumors: safety evaluation. J. Neurol. Surg. A. Cent. Eur. Neurosurg. 76, 473–482 (2015)
Nishihara, T., Teraoka, A., Morita, A., Ueki, K., Takai, K., Kirino, T.: A transparent sheath for endoscopic surgery and its application in surgical evaluation of spontaneous intracerebral hematomas: Technical note. J. Neurosurg. 92, 1053–1055 (2000)
Takimoto, H., Iwaisako, K., Kubo, S., Yamanaka, K., Karasawa, J., Yoshimine, T.: Transaqueductal aspiration of pontine hemorrhage with the aid of a neuroendoscope: Technical note. J. Neurosurg. 98, 917–919 (2003)
Toth, S., Vajda, J., Pasztor, E., Toth, Z.: Separation of the tumor and brain surface by “water jet” in cases of meningiomas. J. Neurooncol. 5, 117–124 (1987)
Laser-induced liquid jet used as a surgical knife. Biophotonics technology solutions. Biophotonics Int. 18–19 (2004)
Acknowledgements
Part of this work was carried out under the Collaborative Research Project of the Institute of Fluid Science, Tohoku University. This work was supported in part by a Grant-in-Aid for Scientific Research (B) (No. 18390388), (No. 19390372), a Grant-in-Aid for Young Scientists (A) (No. 19689028), (22689039) and Challenging Exploratory Research (No. 21659313), (21659334), (23659680) from the Japanese Ministry of Education, Culture, Sports, Science, and Technology, 2010 Research Grant from The General Insurance Association of Japan, The Japanese Foundation for Research and Promotion of Endoscopy, Ministry of Economy, Technology and Industry, Mitsui Welfare Foundation, Daiwa Shoken Welfare Foundation, Ogino Award from Japan Society of Biomedical Engineering, and the Tohoku University Exploratory Research Program for Young Scientists (ERYs). We also thank Shokichi Hayasaka and Toshihiro Ogawa (Institute of Fluid Science, Tohoku University) for technical support. We also thank the administrative support of Asaka Ishigamori, Emiko Kaneda, and Atsumi Takahashi.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. H. R. Hosseini and A. Higgins.
Rights and permissions
About this article
Cite this article
Nakagawa, A., Kumabe, T., Ogawa, Y. et al. Pulsed laser-induced liquid jet: evolution from shock/bubble interaction to neurosurgical application. Shock Waves 27, 1–14 (2017). https://doi.org/10.1007/s00193-016-0696-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00193-016-0696-2