Abstract
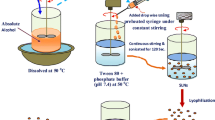
The objective of the present study was to develop a novel solid lipid nanoparticle (SLN) for the lung-targeting delivery of dexamethasone acetate (DXM) by intravenous administration. DXM loaded SLN colloidal suspensions were prepared by the high pressure homogenization method. The mean particle size, drug loading capacity and drug entrapment efficiency (EE %) of SLNs were investigated. In vitro drug release was also determined. The biodistribution and lung-targeting efficiency of DXM-SLNs and DXM-solutions (DXM-sol) in mice after intravenous administration were studied using reversed-phase high-performance liquid chromatography (HPLC). The results (expressed as mean ± SD) showed that the DXM-SLNs had an average diameter of 552 ± 6.5 nm with a drug loading capacity of 8.79 ± 0.04% and an entrapment efficiency of 92.1 ± 0.41%. The in vitro drug release profile showed that the initial burst release of DXM from DXM-SLNs was about 68% during the first 2 h, and then the remaining drug was released gradually over the following 48 hours. The biodistribution of DXM-SLNs in mice was significantly different from that of DXM-sol. The concentration of DXM in the lung reached a maximum level at 0.5 h post DXM-SLNs injection. A 17.8-fold larger area under the curve of DXM-SLNs was achieved compared to that of DXM-sol. These results indicate that SLN may be promising lung-targeting drug carrier for lipophilic drugs such as DXM.
Similar content being viewed by others
References
Chinese Pharmacopoeia, pp. 184 (2005).
Dik, W. A., McAnulty, R. J., Versnel, M. A., Naber, B. A., Zimmermann, L. J., Laurent, G. J., and Mutsaers, S. E., Short course dexamethasone treatment following injury inhibits bleomycin induced fibrosis in rats.Thorax, 58, 765–71 (2003).
Darwis, Y. and Kellaway, I. W., Nebulization of rehydrated freeze-dried beclomethasone dipropionate liposomes.Int. J. Pharm., 215, 113–121 (2001).
Eldem, T., Speiser, P., and Hincal, A., Optimization of spray- dried and -congealed lipid micropellets and characterization of their surface morphology by scanning electron microscopy.Pharm. Res., 8, 47–54 (1991).
Fundaro, A., Cavalli, R., Bargoni, A., Vighetto, D., Zara, G. P., and Gasco, M. R., Non-stealth and stealth solid lipid nanoparticles (SLN) carrying doxorubicin: pharmacokinetics and tissue distribution afteri.v. administration to rats.Pharm. Res., 42, 337–343 (2000).
Gert, S., Sheila, O. B., Toos, D., and Danilo, D. L., Surface modification of nanoparticles to oppose uptake by the mononudear phagocyte system.Adv. Drug Deliv. Rev., 17, 31–48 (1995).
Groeneveld, A. B., Vascular pharmacology of acute lung injury and acute respiratory distress syndrome.Vasc. Pharmacol., 39, 247–256 (2002).
Huo, D. J., Deng, S. H., Li, L. B., and Ji, J., Studies on the poly(lactic-co-glycolic) acid microspheres of cisplatin for lungtargeting. Inf.J. Pharm., 289, 63–67 (2005).
Jenning, V., Mader, K., and Gohla, S. H., Solid lipid nanoparticles (SLN) based on binary mixtures of liquid and solid lipids: a (1)H-NMR study.Int. J. Pharm., 205, 15–21 (2000).
Joshi, M. and Misra, A., Disposition kinetics of ketotifen from liposomal dry powder for inhalation in rat lung.Clin. Exp. Pharmacol P., 30, 153–156 (2003).
Kellaway, I. W. and Farr, S. J., Liposomes as drug delivery systems to the lung.Adv. Drug Deliv. Rev., 5, 149–161 (1990).
Li, H. P., Li, X., He, G. J., Yi, X. H., and Kaplan, A. P., The influence of dexamethasone on the proliferation and apoptosis of pulmonary inflammatory cells in bleomycininduced pulmonary fibrosis in rats.Respirology, 9, 25–32 (2004).
Lukowski, G. and Pflegel, P., Electron diffraction of solid lipid nanoparticles loaded with aciclovir.Pharmazie, 52, 642–643 (1997).
Mehnert, W. and Mader, K., Solid lipid nanoparticles: production, characterization and applications.Adv. Drug Deliv. Rev., 47, 165–196 (2001).
Moghimi, S. M., Mechanisms regulating body distribution of nanospheres conditioned with pluronic and tetronic block co-polymers.Adv. Drug Deliv. Rev., 16, 183–193 (1995).
Müller, R. H., Karsten, M., and Sven, G., Solid lipid nanoparticles (SLN) for controlled drug delivery-α review of the state of the art.Eur. J. Pharm. Biopharm., 50, 161–177 (2000).
Müller, R.H., Maaseen, S., Schwarz, C., and Mehnert, W., Solid lipid nanoparticles (SLN) as potential carrier for human use: interaction with human granulocytes.J. Control. Release, 47, 261–269 (1997).
Mühlen, zur A. and Mehnert, W., Drug release and release mechanism of prednisolone loaded solid lipid nanoparticles.Pharmazie, 53, 552 (1998).
Ozkan, Y., Dikmen, N., Isimer, A., Gunhan, O., and Aboul-Enein, H. Y., Clarithromycin targeting to lung: characterization, size distribution andin vivo evaluation of the human serum albumin microspheres.Farmaco, 55, 303–307 (2000).
Serpe, L., Catalano, M. G., Cavalli, R., Ugazio, E., Bosco, O., Canaparoa, R., Muntoni, E., Frairia, R., Gasco, M. R., Eandi, M., and Zara, G. P., Cytotoxicity of anticancer drugs incorporated in solid lipid nanoparticles on HT-29 colorectal cancer cell line.Eur. J. Pharm. Biopharm., 58, 673–680 (2004).
Serrano, A. G. and Perez-Gil, J., Protein-lipid interactions and surface activity in the pulmonary surfactant system.Chem. Phys. Lipids, 141, 105–118 (2006).
Siekmann, B. and Westesen, K., Melt-homogenized solid lipid nanoparticles stabilized by the nonionic surfactant tyloxapol. I. Preparation and particle size determination.Pharm. Pharmacol. Lett., 3, 194–197 (1994).
Speiser, P., Lipidnanopellets als Tragersystem fur Arzneimittel zur peroralen Anwendung. European Patent EP0167825 (1990).
Suzuki, S., Kawakami, S., Chansri, N., Yamashita, F., and Hashida, M., Inhibition of pulmonary metastasis in mice by all-trans retinoic acid incorporated in cationic liposomes.J. Control. Release, 116, 58–63 (2006).
Swigris, J. J, Kuschner, W. G., Kelsey, J. L., and Gould, M. K., Idiopathic pulmonary fibrosis: challenges and opportunities for the clinician and investigator.Chest, 127, 275–283 (2005).
Tao, Z. W., Xu, Q. Y., Ye, Y. Q., Peng, P., and Wang, Z. R., Preparation andin vitro characterization of microspheres containing dexamethasone sodium phosphate.Chin. Hosp. Pharm. J., 26, 515–517 (2006).
Wang, J. S., Zhu, J. B., Lu, R. Q., and Shen, W., Preparation of lung targeting azithromycin liposomes and its tissue distribution in mice.Acta Pharm. Sin., 40, 274–278 (2005).
Wang, J. X., Sun, X., and Zhang, Z. R., Enhanced brain targeting by synthesis of 3′, 5′-dioctanoyl- 5-fluoro- 2′- deoxyuridine and incorporation into solid lipid nanoparticles.Eur. J. Pharm. Biopharm., 54, 285–290 (2002).
Wolfgang, M. and Karsten, M., Solid lipid nanoparticles Production, characterization and applications.Adv. Drug Deliv. Rev., 47, 165–196 (2001).
Yang, S. C., Lu, L. F., Cai, Y., Zhu, J. B., Liang, B. W., and Yang, C. Z., Body distribution in mice of intravenously injected camptothecin solid lipid nanoparticles and targeting effect on brain.J. Control. Release, 59, 299–307 (1999).
Zara, G. P., Cavalli, R., Fundaro, A., Bargoni, A., Caputo, O., and Gasco, M. R., Pharmacokinetics of doxorubicin incorporated in solid lipid nanospheres (SLN).Pharm. Res., 40, 281–286 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiang, Qy., Wang, Mt., Chen, F. et al. Lung-targeting delivery of dexamethasone acetate loaded solid lipid nanoparticles. Arch Pharm Res 30, 519–525 (2007). https://doi.org/10.1007/BF02980228
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02980228