Abstract
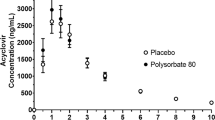
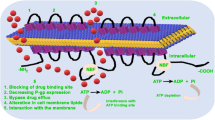
Tween 80 (Polysorbate 80) is a hydrophilic nonionic surfactant commonly used as an ingredient in dosing vehicles for pre-clinicalin vivo studies (e.g., pharmacokinetic studies, etc.). Tween 80 increased apical to basolateral permeability of digoxin in Caco-2 cells suggesting that Tween 80 is anin vitro inhibitor of P-gp. The overall objective of the present study was to investigate whether an inhibition of P-gp by Tween 80 can potentially influencein vivo absorption of P-gp substrates by evaluating the effect of Tween 80 on the disposition of digoxin (a model P-gp substrate with minimum metabolism) after oral administration in rats. Rats were dosed orally with digoxin (0.2 mg/kg) formulated in ethanol (40%, v/v) and saline mixture with and without Tween 80 (1 or 10%, v/v). Digoxin oral AUC increased 30 and 61% when dosed in 1% and 10% Tween 80, respectively, compared to control (P<0.05). To further examine whether the increase in digoxin AUC after oral administration of Tween 80 is due, in part, to a systemic inhibition of digoxin excretion in addition to an inhibition of P-gp in the Gl tract, a separate group of rats received digoxin intravenously (0.2 mg/kg) and Tween 80 (10% v/v) orally. No significant changes in digoxin IV AUC was noted when Tween 80 was administered orally. In conclusion, Tween 80 significantly increased digoxin AUC and Cmax after oral administration, and the increased AUC is likely to be due to an inhibition of P-gp in the gut (i.e., improved absorption). Therefore, Tween 80 is likely to improve systemic exposure of P-gp substrates after oral administration. Comparing AUC after oral administration with and without Tween 80 may be a viable strategy in evaluating whether oral absorption of P-gp substrates is potentially limited by P-gp in the gut.
Similar content being viewed by others
References
Batrakova, E. V., Han, H. Y., Alakhov, V. Y., Miller, D. W., and Kabanov, A. V., Effects of pluronic block copolymers on drug absorption in Caco-2 cell monolayers.Pharm. Res., 15, 850–855 (1998).
Borst, P., Evers, R., Kool, M., and Wijnholds, J., The multidrug resistance protein family.Biophys. Acta, 1461, 347–357 (1999).
Cornaire, G., Woodley, J. F., Saivin, S., Legendre, J. Y., Decourt, S., Cloarec, A., and Houin, G., Effect of polyoxyl 35 castor oil and Polysorbate 80 on the intestinal absorption of digoxinin vitro.Arzneimittel-forschung., 50, 576–579 (2000).
Fromm, M. R., Kim, R. B., Stein, C. M., Wilkinson, G. R., and Roden, D. M., Inhibition of P-glycoprotein-mediated drug transport: A unifying mechanism to explain the interaction between digoxin and quinidine.Circulation, 99, 552–557 (1999).
Gottesman, M. M. and Pastan, I., Biochemistry of multidrug resistance mediated by the multidrug transporter.Annu. Rev. Biochem., 62, 385–427 (1993).
Hedman, A., Angelin, B., Arvidsson, A., Dahlqvist, R., and Nilsson, B., Interactions in the renal and biliary elimination of digoxin: stereoselective difference between quinine and quinidine.Clin. Pharmacol. Ther., 47, 20–26 (1990).
Hinderling, P. H. and Hartmann, D., Pharmacokinetics of digoxin and main metabolites/ derivatives in healthy humans.Ther. Drug Monit., 13, 381–401 (1991).
Hugger, E. D., Novak, B. L., Burton, P. S., Audus, K. L., and Borchardt, R. T., A comparison of commonly used polyeht-oxylated pharmaceutical excipients on their ability to inhibit P-glycoprotein activityin vitro.J. Pharm. Sci., 91, 1991–2002 (2002).
lisalo, E., Clinical pharmacokinetics of digoxin.Clin. Pharmacokinet., 2, 1–16 (1977).
Knutsen, T., Mickley, L. A., Ried, T., Green, E. D., du Manoir, S., Schrock, E., Macville, M., Ning, Y., Robey, R., Polymeropoulos, M., Torres, R., and Fojo, T., Cytogenetic and molecular characterization of random chromosomal rearrangements activating the drug resistance gene, MDR1/P-glycoprotein, in drug-selected cell lines and patients with drug refractory ALL.Genes Chromosomes Cancer, 23, 44–54 (1998).
Litman, T., Druley, T. E., Stein, W. D., and Bates, S. E., From MDR to MXR: new understanding of multidrug resistance systems, their properties and clinical significance.Cell. Mol. Life Sci. 58, 931–959 (2001).
Malingre, M. M., Schellens, J. H., Van Tellingen, O., Ouwehand, M., Bardelmeijer, H. A., Rosing, H., Koopman, F. J., Schot, M. E., Ten Bokkel Huinink, W. W., and Beijnen, J. H., The cosolvent Cremophor EL limits absorption of orally administered paclitaxel in cancer patients.Br. J. Cancer, 85, 1472–1477 (2001).
Nerurkar, M. M., Ho, N. F., Burton, P. S., Vidmar, T. J., and Borchardt, R. T., Mechanistic roles of neutral surfactants on concurrent polarized and passive membrane transport a model peptide in Caco-2 cells.J. Pharm. Sci., 86, 813–821 (1997).
Salphati, L. and Benet, L. Z., Effects of ketoconazole on digoxin absorption and disposition in rat.Pharmacology, 56, 308–313 (1998).
Salphati, L. and Benet, L. Z., Metabolism of digoxin and digoxigenin digitoxosides in rat liver microsomes: involvement of cytochrome P4503A.Xenobiotica, 29, 171–85 (1999).
Silverman, J. A., Multidrug-resistance transporters.Pharm. Biotechnol., 12, 353–86 (1999).
Song, S., Suzuki, H., Kawai, R., and Sugiyama, Y., Effect of PSC 833, a P-glycoprotein modulator, on the disposition of vincristine and digoxin in rats.Drug Metab. Disp., 27, 689–694 (1999).
Stephens, R. H., O’Neill, C. A., Warhurst, A., Carlson, G. L., Rowland, M., and Warhurst, G., Kinetic profiling of P-glycoprotein-mediated drug efflux in rat and human intestinal epithelia.J. Pharmacol. Exp. Ther., 296, 584–591 (2001).
Tanigawara, Y., Role of P-glycoprotein in drug disposition.Ther. Drug Monit., 22, 137–140 (2000).
Troutman, M. D., Luo, G., Gan, L. S., and Thakker, D. R., The role of P-glycoprotein in drug disposition: significance to drug development. In Rodrigues, A. D. (ed.). Drug-drug interactions. Marcel Dekker, New York. pp. 295–357 (2001).
van Zuylen, L., Verweij, J., and Sparreboom, A., Role of formulation vehicles in taxane pharmacology.Invest. New Drugs, 19, 125–141 (2001).
Verschraagen, M., Koks, C. H., Schellens, J. H., and Beijnen, J. H., P-glycoprotein system as a determinant of drug interactions: the case of digoxin-verapamil.Pharmacol. Res., 40, 301–306 (1999).
Yamazaki, M., Neway, W. E., Ohe, T., Chen, I., Rowe, J. F., Hochman, J. H., Chiba, M., and Lin, J. H.,In vitro substrate identification studies for p-glycoprotein-mediated transport: species difference and predictability ofin vivo results.J. Pharmacol. Exp. Ther., 296, 723–735 (2001).
Yao, M., Zhang, H., Chong, S., Zhu, M., and Morrison, R. A., A rapid and sensitive LC/MS/MS assay for quantitative determination of digoxin in rat plasma.J. Pharmaceu. Biomed. Anal., 32, 1189–1197 (2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Yao, M., Morrison, R.A. et al. Commonly used surfactant, Tween 80, improves absorption of P-glycoprotein substrate, digoxin, in rats. Arch Pharm Res 26, 768–772 (2003). https://doi.org/10.1007/BF02976689
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02976689