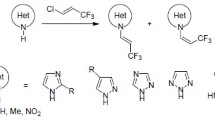
Abstract
Reactions of l-(benzotriazol-l-yl)-l-(4-R-phenylamino)- and l-(3,5-dimethylpyrazol-l-yl)-l-(4-R-phenylamino)-2-nitro-3,4,4-trichloro-l, 3-butadienes and also l-(benzotriazol-l-yl)-l-(4-R-phenylamino)- and l-(3,5-dimethylpyrazol-l-yl)-l-(4-R-phenylamino)-2-nitro-4-bromo-3,4-dichloro-l,3-butadienes (R = EtO, MeO, Me) with amines proceed with replacement of the terminal chlorine in the trichlorovinyl group or of bromine in the bromodichlorovinyl group by an amine rest to yield the corresponding l-azolyl-4-amino-l-arylamino-2-nitro-3,4-dichloro-l,3-butadienes. When binucleophilic reagents (ethylenediamine and 2-aminoethanol) are used only one N-nucleophilic site of the nucleophile takes part in the reaction.
Similar content being viewed by others
References
Zapol’skii, V.A., Potkin, V.I., and Kaberdin, R.V.,Zh. Org. Khim., 1999, vol. 35, no. 3, pp. 469–473.
Zapol’skii, V.A., Potkin, V.I., Nechai, N.I., Kaberdin, R.V., and Pevzner, M.S.,Zh. Org. Khim., 1997, vol. 33, no. 10, pp. 1541–1547.
Zapol’skii, V.A., Potkin, V.I., Nechai, N.I., Kaberdin, R.V., and Pevzner, M.S.,Zh. Org. Khim., 1997, vol. 33, no. 11, pp. 1715–1720.
Katritzky, A.R. and Lan, X.,Chem. Soc. Rev., 1994, vol. 23, pp. 363–373.
Shainyan, B.A.,Usp. khim., 1986, vol. 55, no. 6, pp. 942–973.
Pearson, R.G.,Hard and Soft Acids and Bases, Stroudenburg: Dowden, Hatchinson, Ross, 1973.
Perekalin, V.V., Sopova, A.S., and Lipina, E.S.,Nepredel’nye nitrosoedineniya (Unsaturated Nitrocompounds), Leningrad: Khimiya, 1982.
Takhistov, V.V.,Prakticheskaya mass-spektrometriya organicheskikh soedinenii Practical Mass Spectrometry of Organic Compounds), Leningrad: Leningrad Gos. Univ., 1977.
Takhistov, V.V., Rodin, A.A., and Maksimova, B.N.,Usp. Khim., 1991, vol. 60, no. 10, pp. 2143–2167.
Author information
Authors and Affiliations
Additional information
For communication III, see [1].
Rights and permissions
About this article
Cite this article
Potkin, V.I., Zapol’skii, V.A., Knizhnikov, V.A. et al. Azolyl derivatives of nitrohalobutadienes: IV. Synthesis of 4-amino derivatives of 1-azolyl-1-arylamino-2-nitrotrihalo-1,3-butadienes. Russ J Electrochem 36, 877–883 (2000). https://doi.org/10.1007/BF02757449
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02757449