Abstract
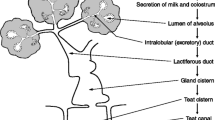
Extracellular storage allows biologically-active substances in milk to influence mammary function. Among these factors is one which regulates the rate of milk secretion acutely according to frequency or completeness of milk removal in each mammary gland. The active factor in goat's milk has been identified by screening milk constituents for their ability to inhibit milk constituent secretion in tissue and cell culture bioassays, and found to be a novel milk protein. The proteins identified by bioassayin vitro, also inhibited milk secretion in lactating goats in a reversible, concentration-dependent manner. This protein, termed FIL (feedback inhibitor of lactation), acts by reversible blockade of constitutive secretion in the mammary epithelial cell. As the inhibitor is synthesized in the same epithelial cells, feedback inhibition is, therefore, an autocrine mechanism. FIL's unusual mechanism of action also influences other aspects of mammary function. Acute disruption of mammary membrane trafficking is associated with downregulation of prolactin receptors and followed by a decrease in epithelial cell differentiation. Thus, in addition to acutely-regulating milk secretion, FIL may induce the adaptation in mammary cell differentiation which actsin vivo to sustain the secretory response to a sustained change in milk removal. In the long term, matching of milk output to demand is achieved by a change in mammary cell number. This developmental response is also local in nature. Whether it too is due to autocrine modulation by FIL of mechanisms influencing cell proliferation or survival, or elicited by another milk-borne factor, remains to be determined.
Similar content being viewed by others
Abbreviations
- FIL:
-
Feedback inhibitor of lactation
- EHS matrix:
-
Engelbreth-Holm-Swarm matrix
References
M. Peaker and M. C. Neville (1991). Commentary. Hormones in milk: chemical signals to the offspring?J. Endocrinol. 131:1–3.
C. E. Grosvenor, M. F. Picciano, and C. R. Baumrucker (1992). Hormones and growth factors in milk.Endocrine Rev. 14:710–728.
A. F. Phillips, R. K. Rao, D. M. McCracken, M. Lake, and O. Koldovský (1991). Presence of insulin-like growth factors and their binding proteins in rat milk. In M. K. Raizada and D. LeRoith (eds.),Molecular Biology and Physiology of Insulin and Insulin-like Growth Factors, Plenum Press, New York, pp. 179–186.
M. F. McGrath, R. J. Collier, D. R. Clemmons, W. H. Busby, C. A. Sweeney, and G. G. Krivi (1991). The directin vitro effect of insulin-like growth factors (IGFs) on normal bovine mammary cell proliferation and production of IGF binding proteins.Endocrinology 129:671–678.
P. J. Fielder, G. Thordarson, A. English, R. G. Rosenfield, and F. Talamantes (1992). Expression of a lactogen-dependent insulin-like growth factor-binding protein in cultured mouse mammary epithelial cells.Endocrinology 131:261–267.
P. Ervin, M. Kaminski, R. Cody, and M. Wicha (1989). Production of mammastatin, a tissue-specific growth inhibitor by normal human mammary epithelial cells.Science 244:1585–1587.
R. Grosse, F.-D. Bohmer, B. Binas, A. Kurtz, E. Spitzer, T. Muller, and W. Zschiesche (1992). Mammary derived growth inhibitor. In R. B. Dickson and M. Lippman (eds.),Cancer Treatment and Research: Genes, Oncogenes and Hormones. Springer Verlag, Heidelberg, pp. 69–94.
M. P. Thompson, H. M. Farrell, S. Mohanam, S. Liu, W. R. Kidwell, M. P. Bansal, R. G. Cook, D. Medina, C. E. Kotts, and M. Bano (1992). Identification of α-lactalbumin as a cell growth inhibitor.Protoplasma 167:134–144.
M. Peaker (1995). Autocrine control of milk secretion: development of the concept. In C. J. Wilde, M. Peaker, and C. H. Knight (eds.),Intercellular Signalling in the Mammary Gland, Plenum Press, New York, pp. 193–202.
C. J. Wilde, A. Daly, D. T. Calvert, and M. Peaker (1987). The effect of goat's milk fractions on synthesis of milk constituents by rabbit mammary explants and on milk yieldin vivo.Biochem. J. 242:285–288.
C. J. Wilde, C. V. P. Addey, L. M. Boddy, and M. Peaker (1995). Autocrine regulation of milk secretion by a protein in milk.Biochem. J. 305:51–58.
H. K. Kleinman, M. L. McGarvey, J. R. Hassall, V. L. Star, F. B. Cannon, G. W. Laurie, and G. R. Martin (1986). Basement membrane complexes with biological activity.Biochemistry 25:312–318.
M. H. Barcellos-Hoff, J. Aggeler, T. G. Ram, and M. J. Bissell (1989). Functional differentiation and alveolar morphogenesis of primary mammary cultures on reconstituted basement membrane.Development 105:223–235.
M. C. Neville, L. Stahl, L. A. Brozo, and J. Lowe-Lieber (1991). Morphogenesis and secretory activity of mouse mammary cultures on EHS matrix.Protoplasma 163:1–8.
R. R. Dils and I. A. Forsyth (1981). Preparation and culture of mammary gland explants. In S. P. Colowick and N. O. Kaplan (eds.),Methods in Enzymology (Vol. 72), Academic Press, New York, pp. 724–742.
M. D. Turner, M. E. Rennison, S. E. Handel, C. J. Wilde, and R. D. Burgoyne (1992). Proteins are secreted by both constitutive and regulated secretory pathways in lactating mouse mammary epithelial cells.J. Cell Biol. 117:269–278.
M. E. Rennison, M. Kerr, C. V. P. Addey, S. E. Handel, M. D. Turner, C. J. Wilde, and R. D. Burgoyne (1993). Inhibition of constitutive protein secretion from lactating mouse mammary epithelial cells by FIL (Feedback inhibitor of lactation), a secreted milk protein.J. Cell Sci. 106:641–648.
M. Ollivier-Bousquet (1978). Early effects of prolactin on lactating rabbit mammary gland.Cell Tissue Res. 187:25–43.
H. Razooki Hasan, D. A. White, and R. J. Mayer (1982). Extensive destruction of newly-synthesised casein in mammary explants in organ culture.Biochem. J. 202:133–138.
C. J. Wilde and C. H. Knight (1986). Degradation of newly-synthesised casein in mammary explants from pregnant and lactating goats.Comp. Biochem. Physiol. 84B:197–201.
T. H. M. Da Costa, V. Illic, and D. H. Williamson (1995).In vitro effects of oxytocin and ionomycin on lipid secretion by rat mammary gland. Role of the myoepithelial cells. In C. J. Wilde, M. Peaker, and C. H. Knight (eds.),Intercellular Signalling in the Mammary Gland, Plenum Press, New York, pp. 265–266.
D. R. Blatchford, K. A. K. Hendry, M. D. Turner, R. D. Burgoyne, and C. J. Wilde (1995). Vectorial secretion by constitutive and regulated secretory pathways in mammary epithelial cells.Epith. Cell Biol. 4:8–16.
K.-I. Enomoto, K. Furuya, S. Yamagishi, and T. Maeno (1992). Mechanically-induced electrical and intracellular calcium responses in normal and cancerous mammary cells.Cell Calcium 13:501–511.
K. Furuya, K.-I. Enomoto, and S. Yamagishi (1993). Spontaneous calcium oscillations and mechanically and chemically-induced calcium responses in mammary epithelial cells.Pflügers Arch. 422:295–304.
K.-I. Enomoto, K. Furuya, S. Yamagishi, T. Oka, and T. Maeno (1994). The increase in intracellular Ca2+ concentration by mechanical stimulation is propagated via release of pyrophorylated nucleotides in mammary epithelial cells.Pflügers Arch. 427:533–542.
J. L. Linzell and M. Peaker (1971). The effects of oxytocin and milk removal on milk secretion in the goat.J. Physiol. London 216:717–734.
M. E. Rennison, S. E. Handel, C. J. Wilde, and R. D. Burgoyne (1992). Investigation of the role of microtubules in protein secretion from lactating mouse mammary epithelial cells.J. Cell Sci. 102:239–247.
R. D. Burgoyne and A. Morgan (1993). Regulated exocytosis.Biochem. J. 293:305–316.
S. E. Handel, M. E. Rennison, C. J. Wilde, and R. D. Burgoyne (1991). Annexin II (Calpactin I) in the mouse mammary gland: immunolocalisation by light and electron microscopy.Cell Tissue Res. 264:549–554.
T. W. Keenan, D. P. Dylewski, D. Ghosal, and B. H. Keon (1992). Milk lipid globule precursor release from endoplasmic reticulum reconstituted in a cell-free system.Eur. J. Biochem. 57:21–29.
B. H. Keon, D. Ghosal, and T. W. Keenan (1993). Association of cytosolic lipids with fatty acid synthase from lactating mammary gland.Int. J. Biochem. 25:533–543.
R. A. Hawkins and D. H. Williamson (1972). Measurements of substrate uptake by mammary gland of the rat.Biochem. J. 129:1171–1173.
K. J. Heesom, P. F. A Souza, V. Ilic, and D. H. Williamson (1992). Chain-length dependency of interactions of medium-chain fatty acids with glucose metabolism in acini isolated from lactating rat mammary glands. A putative feedback to control milk lipid synthesis.Biochem. J. 281:273–278.
D. H. Williamson, V. Ilic, and P. Lund (1995). A role for medium-chain fatty acids in the regulation of lipid synthesis in milk stasis? In C. J. Wilde, M. Peaker, and C. H. Knight (eds.),Intercellular Signalling in the Mammary Gland, Plenum Press, New York, pp. 239–251.
W. L. Hurley, D. R. Blatchford, K. A. K. Hendry, and C. J. Wilde (1994). Extracellular matrix and mouse mammary cell function: comparison of substrata in culture.In Vitro Cell Dev. Biol. 30A:529–538.
C. J. Wilde, C. V. P. Addey, and M. Peaker (1996). Active immunisation of lactating goats against an autocrine inhibitor of milk secretion.J. Physiol. 491:465–469.
J. B. Helms, A. Karrenbauer, K. W. A. Wirtz, J. E. Rothman, and F. T. Wieland (1990). Reconstitution of steps in the constitutive secretory pathway in permeabilised cells.J. Biol. Chem. 265:20027–20032.
S. G. Miller and H.-P. H. Moore (1991). Reconstitution of constitutive secretion using semi-intact cells: regulation by GTP but not calcium.J. Cell Biol. 261:11398–11403.
F. A. Barr, A. Leyte, S. Mollner, T. Pfeuffer, S. A. Tooze, and W. B. Huttner (1991). Trimeric G-proteins of the trans-Golgi network are involved in the formation of constitutive secretory vesicles and immature secretory granules.FEBS Lett. 294:239–243.
J. G. Donaldson, R. A. Kahn, J. Lippincott-Schwartz, and R. D. Klausner (1991). Binding of ARF and β-COP to Golgi membranes: possible regulation by trimeric G protein.Science 254:1197–1199.
J. Lucocq, G. Warren, and J. Pryde (1991). Okadaic acid induces Golgi apparatus fragmentation and arrest of intracellular transport.J. Cell Sci. 100:753–759.
H. W. Davidson, C. H. McGowan, and W. E. Balch (1992). Evidence for the regulation of exocytotic transport by protein phosphorylation.J. Cell Biol. 116:1343–1355.
M. A. De Matteis, G. Santini, R. A. Kahn, G. Tullio, and A. Liuni (1993). Receptor and protein kinase-C-mediated regulation of ARF binding to the Golgi complex.Nature 364:818–821.
M. D. Turner, C. J. Wilde, and R. D. Burgoyne (1992). Exocytosis from permeabilised lactating mouse mammary epithelial cells.Biochem J. 286:13–15.
J. M. Bryson, C. J. Wilde, and C. V. P. Addey (1993). Effect of unilateral changes in milking frequency on mammary mRNA concentrations in the lactating goat.Biochem. Soc. Trans. 21:294S.
C. J. Wilde, C. H. Knight, C. V. P. Addey, D. R. Blatchford, M. Travers, C. N. Bennett, and M. Peaker (1990). Autocrine regulation of mammary cell differentiation.Protoplasma 159:112–117.
C. J. Wilde, A. J. Henderson, C. H. Knight, D. R. Blatchford, A. Faulkner, and M. Peaker (1987). Effect of thrice daily milking on mammary enzyme activity, cell population and milk yield in the goat.J Anim. Sci. 64:533–539.
J. Lippincott-Schwartz, L. Juan, J. S. Bonifacino, and R. D. Klausner (1989). Rapid redistribution of Golgi proteins into the ER in cells treated with brefeldin A: evidence for membrane cycling from Golgi to ER.Cell 56:801–813.
L. Orci, M. Tagaya, M. Amherdt, A. Perrelet, J. G. Donaldson, J. Lippincott-Schwartz, R. D. Klausner, and J. E. Rothman (1991). Brefeldin A, a drug that blocks secretion, prevents the assembly of non-clathrin-coated buds on Golgi cisterae.Cell 64:1183–1195.
C. N. Bennett, C. H. Knight, and C. J. Wilde (1991). Regulation of mammary prolactin binding by secreted milk proteins.J. Endocrinol. 13:117–125.
C. J. Wilde, D. T. Calvert, and M. Peaker (1988). Effect of a fraction of goat milk serum proteins on milk accumulation and enzyme activities in rabbit mammary gland.Biochem. Soc. Trans. 15:916–917.
C. J. Wilde, D. R. Blatchford, and M. Peaker (1991). Regulation of mouse mammary cell differentiation by extracellular milk proteins.Exp. Physiol. 76:379–387.
M. T. Travers and M. C. Barber (1993). Isolation of a goat acetyl-CoA carboxylase complementary DNA and effect of milking frequency on the expression of the acetyl-CoA carboxylase and fatty acid synthase genes in goat mammary gland.Comp. Biochem. Physiol. 105B:123–128.
F. Rösl (1992). A simple and rapid method for detection of apoptosis in human cells.Nucl. Acids Res. 20:5243–5244.
A. H. Wyllie, J. F. R. Kerr, and A. R. Currie (1980). Cell death: the significance of apoptosis.Int. Rev. Cytol. 68:251–306.
L. H. Quarrie, C. V. P. Addey, and C. J. Wilde (1995). Apoptosis in lactating and involuting mouse mammary tissue demonstrated by nick-end DNA labelling.Cell Tissue Res. 281:413–419.
N. I. Walker, R. E. Bennett, and J. F. R. Kerr (1989). Cell death by apoptosis during involution of the lactating breast in mice and rats.Am. J. Anat. 185:19–32.
R. Strange, F. Li, S. Saurer, A. Burkhardt, and R. R. Friis (1992). Apoptotic cell death and tissue remodelling during mouse mammary gland involution.Development 115:49–58.
L. H. Quarrie, C. V. P. Addey, and C. J. Wilde (1996). Programmed cell death during mammary tissue involution induced by weaning, litter removal and milk stasis.J. Cell. Physiol. (in press).
L. H. Quarrie, C. V. P. Addey, and C. J. Wilde (1995). Local control of mammary apoptosis by milk stasis. In C. J. Wilde, M. Peaker, and C. H. Knight (eds.),Intercellular Signalling in the Mammary Gland, Plenum Press, New York, pp. 95–96.
L. H. Quarrie, C. V. P. Addey, and C. J. Wilde (1994). Local regulation of mammary apoptosis in the lactating goat.Biochem. Soc. Trans. 22:178S.
L. G. Sheffield and L. C. Kotolski (1992). Prolactin inhibits programmed cell death during mammary gland involution.FASEB J. 6:A1184.
M. T. Travers, M. C. Barber, E. Tonner, L. H. Quarrie, C. J. Wilde, and D. J. Flint (1996). The role of prolactin and growth hormone in the regulation of casein gene expression and mammary cell survival: relationships to milk synthesis and secretion.Endocrinology (in press).
N. Boudreau, C. J. Simpson, Z. Werb, and M. J. Bissell (1995). Suppression of ICE and apoptosis in mammary epithelial cells by extracellular matrix.Science 267:891–893.
R. S. Guenette and M. Tenniswood (1995). The role of insulin like growth factors binding proteins (IGFBPs) in regulating active cell death in regressing rat prostate and mammary gland.J. Cell. Biochem. (Suppl.)19B:280.
E. Tonner, J. Beattie, and D. J. Flint (1995). Production of an insulin-like growth factor binding protein by the involuting rat mammary gland. In C. J. Wilde, M. Peaker, and C. H. Knight (eds.),Intercellular Signalling in the Mammary Gland, Plenum Press, New York, pp. 103–104.
C. Sell, R. Baserga, and R. Rubin (1995). Insulin-like growth factor I (IGF-1) and the IGF-1 receptor prevent etoposide-induced apoptosis.Cancer Res. 55:303–306.
C. H. Knight and M. Peaker (1979). Adaptive hyperplasia and compensatory growth in the salt glands of ducks and geese.J. Physiol. (Lond.) 294:145–151.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peaker, M., Wilde, C.J. Feedback control of milk secretion from milk. J Mammary Gland Biol Neoplasia 1, 307–315 (1996). https://doi.org/10.1007/BF02018083
Issue Date:
DOI: https://doi.org/10.1007/BF02018083