Summary
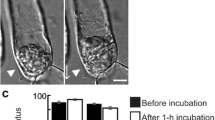
Changes in F-actin organization following mechanical isolation ofZinnia mesophyll cells were documented by rhodamine-phalloidin staining. Immediately after isolation, most cells contained irregular cortical actin fragments of varying lengths, and less than 5% of cells contained intact cortical filaments. During the first 8 h of culture, filament fragments were replaced by actin rings, stellate actin aggregates, and bundled filament fragments. Some of these aggregates had no association with organelles (“free” actin aggregates). Other aggregates were associated with chloroplasts, which changed in shape and location at the same time actin aggregates appeared. F-actin was concentrated within or around the nucleus in a small percentage of cells. After 12 h in culture, the percentage of cells with free actin rings and chloroplast-associated actin aggregates began to decline and the percentage of cells having intact cortical actin filaments increased greatly. Intermediate images were recorded that strongly indicate that free actin rings, chloroplast-associated actin rings, and other actin aggregates self-assemble by successive bundling of actin filament fragments. The fragmentation and bundling of F-actin observed in mechanically isolatedZinnia cells resembles changes in F-actin distribution reported after diverse forms of cell disturbance and appears to be an example of a generalized response of the actin cytoskeleton to cell stress.
Similar content being viewed by others
Abbreviations
- FITC:
-
fluorescein isothiocyanate
- MBS:
-
m-maleimidobenzoic acid N-hydroxysuccinimide ester
- RhPh:
-
tetramethylrhodamine isothiocyanate-phalloidin
References
Alfano F, Russell A, Gambardella R, Duckett JG (1993a) The actin cytoskeleton of the liverwortRiccia fluitans: effects of cytochalasin B and aluminium ions on rhizoid tip growth. J Plant Physiol 142: 569–574
—, Duckett JG, Gambardella R (1993b) Circular and spiral F-actin associated with chloroplasts in liverwort thalli and in gametophyte cells ofEquisetum andPteridium. G Bot Ital 127: 292–294
Allen NS, Kubinski DJ (1995) Actin dynamics and a putative gelsolin inMedicago sativa (alfalfa). Mol Biol Cell Suppl 6: 22A
Borovikov YS, Norman JC, Price LS, Weeds A, Koffer A (1995) Secretion from permeabilized mast cells is enhanced by addition of gelsolin: contrasting effects of endogenous gelsolin. J Cell Sci 108: 657–666
Cande WZ, Goldsmith MHM, Ray PM (1973) Polar auxin transport and auxin-induced elongation in the absence of cytoplasmic streaming. Planta 111: 279–296
Cho S-O, Wick SM (1990) Distribution and function of actin in the developing stomatal complex of winter rye (Secale cereale cv. Puma). Protoplasma 157: 154–164
Collings DA, Wasteneys GO, Williamson RE (1995) Cytochalasin rearranges cortical actin of the algaNitella into short, stable rods. Plant Cell Physiol 36: 765–772
— —, Miyazaki M, Williamson RE (1994) Elongation factor 1α is a component of the subcortical actin bundles of characean algae. Cell Biol Int Rep 18: 1019–1024
Cooper JA (1987) Effects of cytochalasin and phalloidin on actin. J Cell Biol 105: 1473–1478
—, Bryan J, Schwab B III, Frieden C, Loftus DJ, Elson EL (1987) Microinjection of gelsolin into living cells. J Cell Biol 104: 491–501
Dong X-J, Ryu J-H, Takagi S, Nagai R (1996) Dynamic changes in the organization of microfilaments associated with the photocontrolled motility of chloroplasts in epidermal cells ofVallisneria. Protoplasma 195 (in press)
Fukuda H, Komamine A (1980) Establishment of an experimental system for the study of tracheary element differentiation from single cells isolated from the mesophyll ofZinnia elegans. Plant Physiol 65: 57–60
Fukui Y, Katsumaru H (1980) Dynamics of nuclear actin bundle induction by dimethyl sulfoxide and factors affecting its development. J Cell Biol 84: 131–140
Garbary DJ, McDonald AR (1996) Fluorescent labelling of the cytoskeleton inCeramium strictum (Rhodophyta). J Phycol 32: 85–93
Goddard RH, La Claire JW II (1991a) Calmodulin and wound healing in the coenocytic green algaErnodesmis verticillata (Kutzing) Borgesen: immunofluorescence and effects of antagonists. Planta 183: 281–293
— — (1991b) Calmodulin and wound healing in the coenocytic green algaErnodesmis verticillata (Kutzing) Borgesen: ultrastructure of the cortical cytoskeleton and immunogold labeling. Planta 186: 17–26
Goodbody KC, Lloyd CW (1990) Actin filaments line up acrossTradescantia epidermal cells, anticipating wound-induced division planes. Protoplasma 157: 92–101
Grabski S, Xie XG, Holland JF, Schindler M (1994) Lipids trigger changes in the elasticity of the cytoskeleton in plant cells: a cell optical displacement assay for live cell measurements. J Cell Biol 126: 713–726
Green PB (1994) Connecting gene and hormone action to form, pattern and organogenesis: biophysical transductions. J Exp Bot 45: 1775–1788
Gross P, Julius C, Schmelzer E, Hahlbrock K (1993) Translocation of cytoplasm and nucleus to fungal penetration sites is associated with depolymerization of microtubules and defence gene activation in infected, cultured parsley cells. EMBO J 12: 1735–1744
Harold RL, Harold FM (1992) Configuration of actin microfilaments during sporangium development inAchlya bisexualis: comparison of two staining protocols. Protoplasma 171: 110–116
Hasek J, Streiblova E (1994) Flunarizine affects F-actin patterns inMucor rouxii germlings. Can J Microbiol 40: 730–735
Hasezawa S, Nagata T, Syono K (1988) The presence of ring formed actin filaments in plant cells. Protoplasma 146: 61–63
He Y, Wetzstein HY (1995) Fixation induces differential tip morphology and immunolocalization of the cytoskeleton in pollen tubes. Physiol Plant 93: 757–763
Higashi-Fujime S (1980) Active movement in vitro of bundles of microfilaments isolated fromNitella cell. J Cell Biol 87: 569–578
Huckriede A, Fuchtbauer A, Hinssen H, Chaponnier C, Weeds A, Jockusch BM (1990) Differential effects of gelsolins on tissue culture cells. Cell Motil Cytoskeleton 16: 229–238
Hush JM, Overall RL (1992) Re-orientation of cortical F-actin is not necessary for wound-induced microtubule re-orientation and cell polarity establishment. Protoplasma 169: 97–106
Kadota A, Wada M (1992a) Photoinduction of formation of circular structures by microfilaments on chloroplasts during intracellular orientation in protonemal cells of the fernAdiantum capillus-veneris. Protoplasma 167: 97–107
— — (1992b) Re-organization of the cortical cytoskeleton in tip-growing fern protonemal cells during phytochrome-mediated phototropism and blue light-induced apical swelling. Protoplasma 166: 35–41
— — (1989) Photoinduction of circular F-actin on chloroplast in a fern protonemal cell. Protoplasma 151: 171–174
Kaminskyj SGW, Heath IB (1994) A comparison of techniques for localizing actin and tubulin in hyphae ofSaprolegnia ferax. J Histochem Cytochem 42: 523–530
Knight BW, Roberts AW (1994) Palisade mesophyll cell expansion during leaf development inZinnia elegans (Asteraceae). Am J Bot 81: 609–615
Kobayashi H, Fukuda H, Shibaoka H (1988) Interrelationship between the spatial disposition of actin filaments and microtubules during the differentiation of tracheary elements in culturedZinnia cells. Protoplasma 143: 29–37
Kobayashi I, Kobayashi Y, Hardham AR (1994) Dynamic re-organization of microtubules and microfilaments in flax cells during the resistance response to flax rust infection. Planta 195: 237–247
— —, Yamaoka N, Kunoh H (1992) Recognition of a pathogen and a nonpathogen by barley coleoptile cells. III. Responses of microtubules and actin filaments in barley coleoptile cells to penetration attempts. Can J Bot 70: 1815–1823
Kohno T, Shimmen T (1987) Ca2+-induced fragmentation of actin filaments in pollen tubes. Protoplasma 141: 177–179
Kuroiwa T, Suzuki K, Itou R, Toda K, O'Keefe TC, Kuroiwa H (1995) Mitochondria-dividing ring: ultrastructural basis for the mechanism of mitochondrial division inCyanidioschyzon merolae. Protoplasma 186: 12–23
La Claire JW II (1989) Actin cytoskeleton in intact and wounded coenocytic green algae. Planta 177: 47–57
Lancelle SA, Hepler PK (1991) Association of actin with cortical microtubules revealed by immunogold localization inNicotiana pollen tubes. Protoplasma 165: 167–172
— — (1988) Cytochalasin-induced ultrastructural alterations inNicotiana pollen tubes. Protoplasma Suppl 2: 65–75
Lehrer SS (1981) Damage to actin filaments by glutaraldehyde: protection by tropomyosin. J Cell Biol 90: 459–466
Liebe S, Quader H (1994) Myosin in onion (Allium cepa) bulb scale epidermal cells: involvement in dynamics of organelles and endoplasmic reticulum. Physiol Plant 90: 114–124
McCurdy DW, Gunning BES (1990) Re-organization of cortical actin microfilaments and microtubules at preprophase and mitosis in wheat root-tip cells: a double label immunofluorescence study. Cell Motil Cytoskeleton 15: 76–87
Masuda Y, Takagi S, Nagai R (1991) Protease-sensitive anchoring of microfilament bundles provides tracks for cytoplasmic streaming inVallisneria. Protoplasma 162: 151–159
Menzel D (1994) Cell differentiation and the cytoskeleton inAcetabularia. New Phytol 128: 369–393
— (1987) The cytoskeleton of the giant coenocytic green algaCaulerpa visualized by immunocytochemistry. Protoplasma 139: 71–76
—, Schliwa M (1986) Motility in the siphonous green algaBryopsis. II. Chloroplast movement requires organized arrays of both microtubules and actin filaments. Eur J Cell Biol 40: 286–295
Meske V, Hartmann E (1995) Reorganization of microfilaments in protonemal tip cells of the mossCeratodon purpureus during the phototropic response. Protoplasma 188: 59–69
Miller DD, Scordilis SP, Hepler PK (1995) Identification and localization of three classes of myosins in pollen tubes ofLilium longiflorum andNicotiana alata. J Cell Sci 108: 2549–2563
Northcote DH (1995) Aspects of vascular tissue differentiation in plants: parameters that may be used to monitor the process. Int J Plant Sci 156: 245–256
Ogawa S, Ueda K, Noguchi T (1995) Division apparatus of the chloroplast inNannochloris bacillaris (Chlorophyta). J Phycol 31: 132–137
Palevitz BA (1988) Cytochalasin-induced reorganization of actin inAllium root cells. Cell Motil Cytoskeleton 9: 283–298
Parthasarathy MV (1985) F-actin architecture in coleoptile epidermal cells. Eur J Cell Biol 39: 1–12
—, Perdue TD, Witztum A, Alvernaz J (1985) Actin network as a normal component of the cytoskeleton in many vascular plant cells. Am J Bot 72: 1318–1323
Peat L, Oliveira L (1994) Organization of the cytoskeleton in the coenocytic algaVaucheria longicaulis var.macounii: an experimental study. Protoplasma 177: 95–107
Pierson ES, Derksen J, Traas JA (1986) Organization of microfilaments and microtubules in pollen tubes grown in vitro or in vivo in various angiosperms. Eur J Cell Biol 41: 14–18
Pollard TD, Cooper JA (1986) Actin and actin-binding proteins. A critical evaluation of mechanisms and functions. Annu Rev Biochem 55: 987–1035
Putnam-Evans C, Harmon AC, Palevitz BA, Fechheimer M, Cormier MJ (1989) Calcium-dependent protein kinase is localized with F-actin in plant cells. Cell Motil Cytoskeleton 12: 12–22
Quader H (1990) Formation and disintegration of cisternae of the endoplasmic reticulum visualized in live cells by conventional fluorescence and confocal laser scanning microscopy: evidence for the involvement of calcium and the cytoskeleton. Protoplasma 155: 166–175
—, Schnepf E (1989) Actin filament array during side branch initiation in protonema cells of the mossFunaria hygrometrica: an actin organizing center at the plasma membrane. Protoplasma 151: 167–170
—, Hofmann A, Schnepf E (1989) Reorganization of the endoplasmic reticulum in epidermal cells of onion bulb scales after cold stress: involvement of cytoskeletal elements. Planta 177: 273–280
Roberts AW, Haigler CH (1989) Rise in chlorotetracycline fluorescence accompanies tracheary element differentiation in suspension cultures ofZinnia. Protoplasma 152: 37–45
Ryu J-H, Takagi S, Nagai R (1995) Stationary organization of the actin cytoskeleton inVallisneria: the role of stable microfilaments at the end walls. J Cell Sci 108: 1531–1539
Sameshima M, Chijiiwa Y, Kishi Y, Hashimoto Y (1994) Novel actin rods appeared in spores ofDictyostelium discoideum. Cell Struct Funct 19: 189–194
Seagull RW (1990) The effects of microtubule and microfilament disrupting agents on cytoskeletal arrays and wall deposition in developing cotton fibers. Protoplasma 159: 44–59
— (1989) The plant cytoskeleton. Crit Rev Plant Sci 8: 131–167
Sonobe S, Shibaoka H (1989) Cortical fine actin filaments in higher plant cells visualized by rhodamine-phalloidin after pretreatment withm-maleimidobenzoyl N-hydroxysuccinimide ester. Protoplasma 148: 80–86
Tang X, Lancelle SA, Hepler PK (1989) Fluorescence microscopic localization of actin in pollen tubes: comparison of actin antibody and phalloidin staining. Cell Motil Cytoskeleton 12: 216–224
Tewinkel M, Kruse S, Quader H, Volkmann D, Sievers A (1989) Visualization of actin filament pattern in plant cells without prefixation. A comparison of differently modified phallotoxins. Protoplasma 149: 178–182
Thimann KV, Reese K, Nachmias VT (1992) Actin and the elongation of plant cells. Protoplasma 171: 153–166
Tiwari SC, Polito VS (1988) Spatial and temporal organization of actin during hydration, activation, and germination of pollen inPyrus communis L.: a population study. Protoplasma 147: 5–15
Traas JA, Doonan JH, Rawlings DJ, Shaw PJ, Watts J, Lloyd CW (1987) An actin network is present in the cytoplasm throughout the cell cycle of carrot cells and associates with the dividing nucleus. J Cell Biol 105: 387–395
Uhnak KS, Roberts AW (1995) Microtubule rearrangements accompanying dedifferentiation in mesophyll cultures ofZinnia elegans L. Protoplasma 189: 81–87
Wasteneys GO, Williamson RE (1991) Endoplasmic microtubules and nucleus-associated actin rings inNitella internodal cells. Protoplasma 162: 86–98
—, Collings DA, Gunning BES, Hepler PK, Menzel D (1996) Actin in living and fixed characean internodal cells: identification of a cortical array of fine actin strands and chloroplast actin rings. Protoplasma 190: 25–38
Williamson RE, Hurley UA (1986) Growth and regrowth of actin bundles inChara: bundle assembly by mechanisms differing in sensitivity to cytochalasin. J Cell Sci 85: 21–32
Yahara I, Harada F, Sekita S, Yoshihira K, Natori S (1982) Correlation between effects of 24 different cytochalasins on cellular structures and cellular events and those on actin in vitro. J Cell Biol 92: 69–78
Yang W, Burkhart W, Cavallius J, Merrick WC, Boss WF (1993) Purification and characterization of a phosphatidylinositol 4-kinase activator in carrot cells. J Biol Chem 268: 392–398
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Frost, A.O., Roberts, A.W. Cortical actin filaments fragment and aggregate to form chloroplast-associated and free F-actin rings in mechanically isolatedZinnia mesophyll cells. Protoplasma 194, 195–207 (1996). https://doi.org/10.1007/BF01882027
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01882027