Abstract
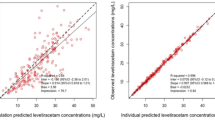
Serial plasma samples were collected after administration of 13 intravenous doses of phenytoin to 11 patients with head injury; 5 to patients who had been receiving enteral feeds for less than 5 days (group 1), and 8 to patients who had been receiving enteral feeds for longer than 5 days (group 2). Average plasma phenytoin concentrations were higher in group 1 than in group 2 (p=0.003). The median intravenous study dose was the same (300 mg) in both groups (p=0.1 7). Group 2 received slightly higher doses expressed as mg/kg (median of 5.45 mg/kg compared to 4.29 mg/kg in group 1, p=0.21). Phenytoin was more rapidly eliminated following intravenous dosing in patients receiving long-term enteral feeding.V max was higher in group 2 than in group 1 (medians, 709versus 394 mg/day) andK m smaller (medians, 2.5versus 3.9 mg/l), but volume of distribution was similar in both groups (p=0.88). The kinetic parameters of phenytoin in group 1 were similar to previously published population pharmacokinetic parameters. In order to maintain phenytoin concentrations adequate for seizure prophylaxis in patients receiving longterm enteral feeding it would be advisable to decrease the dosing interval as well as increasing the phenytoin dose when the drug is administered intravenously.
Similar content being viewed by others
References
North JB, Hanieh A, Challen RG, Penhall RK, Hahn CS, Frewin DB. Postoperative epilepsy: a double-blind trial of phenytoin after craniotomy. Lancet 1980;1:384–6.
Temkin NR, Dikman SS, Wilensky AJ, Keihm J, Chabal S, Winn HR. A randomised, double-blind study of phenytoin for the prevention of post-traumatic seizures. N Engl J Med 1990;323:497–502.
Welty T, Horner TG. Pathophysiology and treatment of subarachnoid hemorrhage. Clin Pharm 1990;9:35–9.
Bauer LA. Interference of oral phenytoin absorption by continuous nasogastric feedings. Neurology 1982;32:570–2.
Cosh DG, Rowett DS, Lee PS, McCarthy PJ. Case report — phenytoin therapy complicated by concurrent chloramphenicol and enteral nutrition. Aust J Hosp Pharm 1987;17:51–3.
Guidry JR, Eastwood TF, Curry SC. Phenytoin absorption in volunteers receiving selected enteral feeding. West J Med 1989;150:659–61.
Hatton RC. Dietary interaction with phenytoin. Clin Pharm 1984;3:110–1.
Jann MW, Bean J, Fidone GS. Interaction of dietary pudding with phenytoin [letter]. Pediatrics 1986;78:952–3.
Longe R, Smith O. Phenytoin interaction with an oral feeding results in loss of seizure control. J Am Geriatr Soc 1988;36:542–4.
Maynard GA, Jones KM, Guidry JR. Phenytoin absorption from tube feedings. Arch Intern Med 1987;147:1821.
McGilvery J. Interaction of phenytoin and nasogastric feeds. Aust J Hosp Pharm 1985;15:128.
Ozuna J, Friel P. Effect of enteral tube feeding on serum phenytoin levels. J Neurosurg Nurs 1984;16:289–91.
Pearce GA. Apparent inhibition of phenytoin absorption by an enteral nutrition formula. Aust J Hosp Pharm 1988;18:289–92.
Saklad JJ, Graves RH, Sharp WP. Interaction of oral phenytoin with enteral feedings. J Parenter Enter Nutr 1986;10:322–3.
Weinryb J, Cogen R. Interaction of nasogastric phenytoin and enteral feeding solution. J Am Geriatr Soc 1989;37:195–6.
Worden JP, Wood CA, Workman CH. Phenytoin and nasogastric feeding [letter]. Neurology 1984;34:132.
Krueger KA, Garnett WR, Comstock TJ, Fitzsimmons WE, Karnes TH, Pellock JM. Effect of two administration schedules of an enteral nutrient formula on phenytoin bioavailability. Epilepsia 1987;28:706–12.
Marvel ME, Bertino JS. Comparative effects of elemental and complex feeding formulation on the absorption of phenytoin suspension. J Parent Enter Nutr 1991;15:316–8.
Nishimura LY, Armstrong EP, Plezia PM, Iaccono RP. Influence of enteral feeding on phenytoin sodium absorption from capsules. DICP 1988;22:130–3.
Cockcroft D, Gault M. Prediction of creatinine clearance from serum creatinine. Nephron 1976;16:31–41.
Remmel RP, Miller SA, Graves NM. Simultaneous assays for felbamate plus carbamazepine, phenytoin and their metabolites by liquid chromatography with mobile phase optimisation. Ther Drug Monit 1990;12:90–6.
Godley PJ, Ludden TM, Clementi WA, Godley SE, Ramsey RR. Evaluation of a Bayesian regression-analysis computer program using non-steady-state phenytoin concentrations. Clin Pharm 1987;6:634–9.
Pulver LK, Tett SE, Montgomery WS. Clinical evaluation of a predictive model for phenytoin plasma concentrations. Aust J Hosp Pharm 1991;21:303–7.
Bauer LA, Blouin RA. Age and phenytoin kinetics in adult epileptics. Clin Pharmacol Ther 1982;31:301–4.
Grasela TH, Sheiner LB, Rambeck B, Boenigk HE, Dunlop A, Mullen PW, et al. Steady state pharmacokinetics of phenytoin from routinely collected patient data. Clin Pharmacokinet 1983;8:355–64.
Lund L. Anticonvulsant levels of diphenylhydantoin relative to plasma levels. Arch Neural 1974;31:289–94.
Vozeh S, Muir KT, Sheiner LB, Follath F. Predicting individual phenytoin dosage. J Pharmacokinet Biopharm 1981;9:131–46.
Messahel FM, Solis GL, Aguwa CN. Does parenteral nutrition lower serum phenytoin levels? Curr Ther Res 1990;47:1017–20.
Boucher BA, Rodman JH, Jaresko GS, Rasmussen SN, Watridge CB, Fabian TC. Phenytoin pharmacokinetics in critically ill trauma patients. Clin Pharmacol Ther 1988;44:675–83.
Hooper WD, Bochner F, Eadie MJ, Tyrer JH. Plasma protein binding of diphenylhydantoin, effect of sex hormones, renal and hepatic disease. Clin Pharmacol Ther 1974;15:276–82.
Winter ME, Tozer TN. Phenytoin. In: Evans WE, Schentag JJ, Jusko WJ, editors. Applied pharmacokinetics — principles of therapeutic drug monitoring. 2nd ed. Spokane: Applied Therapeutics, 1986:493–539.
Blum MR, Riegelman S, Becker CE. Altered protein binding of diphenylhydantoin in uraemic plasma. N Engl J Med 1972;286:109–11.
Peterson GM, Khoo BHC, Von Witt RJ. Clinical response in epilepsy in relation to total and free serum levels of phenytoin. Ther Drug Monitor 1991;13:415–9.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Randall, C.T.C., Tett, S.E. Phenytoin pharmacokinetics after intravenous administration to patients receiving enteral tube feeding. Pharm World Sci 16, 217–224 (1994). https://doi.org/10.1007/BF01871260
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01871260