Summary
This study examines the anatomical substrate for the spinal micturition reflex. Light microscopy of pyridine silver-stained sections revealed that the sacral parasympathetic nucleus (SPN) exists as a broken column or chain of cell clusters located along the intermediolateral portion of the dorsal horn in sacral segments S2–S4. Quantitative analysis of neuropil components in electron micrographs provides data for each type of bouton identified in this nucleus. On the somata of these neurons, boutons containing clear spherical vesicles (S type) comprise 70% of the bouton population. Terminals containing three or more dense core vesicles (GS boutons) account for 26% and boutons containing flattened vesicles (F boutons) comprise 4% of the population. F boutons are more common on large dendrites where they comprise 10% of the total bouton population.
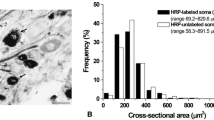
The actual population density of each bouton type is most evident when the number of boutons is expressed as boutons per 100 μm of membrane length (btn/100 μm). S type boutons are the most frequently encountered type. The population density of S boutons is the same on soma and dendrites at 6.66 btn/100 μm. F boutons are more numerous on large (> 2 μm) dendrites (1.28 btn/100 μm) than on small dendrites (0.63 btn/100 μm) or on somata (0.36 btn/ 100 μm). GS boutons occur more frequently on small dendrites (3.66 btn/100 μm) than on somata (2.29 btn/100 μm), large dendrites (2.88 btn/100 μm) or medium dendrites (2.27 btn/ 100 μm). These data suggest that the dense core vesicle-containing boutons are applied primarily to small (<1 μm) dendrites and that F boutons are associated mostly with large or proximal dendrites.
These results provide a quantitative profile of the synaptic input to the sacral autonomic (parasympathetic) neurons which innervate the urinary bladder and demonstrate specific population differences on various postsynaptic structures in this nucleus.
Similar content being viewed by others
References
Bernstein, J. J. andBernstein, M. E. (1976) Ventral horn synaptology in the rat.Journal of Neurocytology 5, 109–23.
Blackstad, T. W. andDahl, H. A. (1961) Quantitative evaluation of structures in contact with neuronal somata.Acta Morphologica Neerlando-Scandinavica 4, 329–43.
Bodian, D. (1975) Origin of specific synaptic types in the motoneuron neuropil of the monkey.Journal of Comparative Neurology 159, 225–44.
Bors, E. (1957) Neurogenic bladder.Urological Survey 7, 177–250.
Brown, H. K. andNolan, M. F. (1977) Synaptology of the sacral parasympathetic nucleus.Journal of the South Carolina Medical Association 73, 14 (abstract).
Brown, H. K. andNolan, M. F. (1978) Digitizer assisted quantitative study of boutons contacting neurons in the sacral parasympathetic nucleus.Society for Neuroscience Abstracts 4, 329 (abstract).
Conradi, S. (1969) On motoneuron synaptology in adult cats.Acta Physiologica Scandinavica Suppl.332, 5–115.
Conradi, S. andRonnevi, L. O. (1977) Ultrastructure and synaptology of the initial axon segment of cat spinal motoneurons during early postnatal development.Journal of Neurocytology 6, 195–210.
Dahlström, A. andFuxe, K. (1965) Evidence for the existence of monamine neurons in the central nervous system. II. Experimentally induced changes in the intraneuronal amine levels in bulbospinal neuron systems.Acta Physiologica Scandinavica Suppl.247, 1–38.
De Groat, W. D. andRyall, R. W. (1968) The identification and characteristics of sacral parasympathetic preganglionic neurons.Journal of Physiology 196, 563–77.
De Groat, W. C. andSaum, W. R. (1971) Sympathetic inhibition of the urinary bladder and of pelvic ganglionic transmission in the cat.Journal of Physiology 220, 297–314.
Gjone, R. andSetekleiv, J. (1963) Excitatory and inhibitory bladder responses to stimulation of the cerebral cortex in the cat.Acta Physiologica Scandinavica 59, 337–42.
Janscó, G. andKnyihar, E. (1975) Functional linkage between nociception and fluorideresistant acid phosphatase activity in the Rolando substance.Neurobiology 5, 42–3.
Knyihár, E. andCsillik, B. (1977) Protargol impregnation of plastic embedded semi-thin sections: A simple method to select degenerative areas for E/M.Journal of Neural Transmission 40, 27–32.
Kuru, M. (1965) Nervous control of micturition.Physiological Reviews 45, 425–92.
Lewin, R. J. andPorter, R. W. (1965) Inhibition of spontaneous bladder activity by stimulation of the globus pallidus.Neurology (Minneapolis) 15, 1049–52.
McLaughlin, B. (1972) The fine structure of neurons and synapses in the motor nuclei of the cat spinal cord.Journal of Comparative Neurology 144, 429–60.
Navaratnam, V. andLewis, P. R. (1970) Cholinesterase-containing neurons in the spinal cord of the rat.Brain Research 18, 411–25.
Nolan, M. F. andBrown, H. K. (1977) Synaptic distribution in the sacral parasympathetic nucleus and associated areas.Anatomical Record 187, 668 (abstract).
Odutola, A. B. (1972) The organization of cholinesterase systems of the monkey spinal cord.Brain Research 39, 353–68.
Oliver, J. E., Bradley, W. E. andFletcher, T. F. (1969) Identification of preganglionic parasympathetic neurons in the sacral spinal cord of the cat.Journal of Comparative Neurology 137, 321–8.
Petras, J. M. andCummings, J. F. (1972) Autonomie neurons in the spinal cord of the rhesus monkey: A correlation of the findings of cytoarchitectonics and sympathectomy with fiber degeneration following dorsal rhizotomy.Journal of Comparative Neurology 146, 189–218.
Petras, J. M. andCummings, J. F. (1978) Sympathetic and parasympathetic innervations of the urinary bladder and urethra.Brain Research 153, 363–9.
Réthelyi, M. (1972) Cell and neuropil architecture of the intermediolateral (sympathetic) nucleus of cat spinal cord.Brain Research 46, 203–13.
Rexed, B. (1954) A cytoarchitectonic atlas of the spinal cord in the cat.Journal of Comparative Neurology 96, 297–379.
Saito, K. (1974) The synaptology and cytology of the Clarke cell in nucleus dorsalis of the cat: an electron microscopic study.Journal of Neurocytology 3, 179–97.
Schnitzlein, H. N., Huffman, H. H., Hammlett, D. M. andHowell, E. M. (1963) A study of the sacral parasympathetic nucleus.Journal of Comparative Neurology 120, 477–94.
Sumner, B. E. H. (1975) A quantitative analysis of boutons with different types of synapses in normal and injured hypoglossal nuclei.Experimental Neurology 49, 406–17.
Tang, D. C. andRuch, T. C. (1956) Localization of brain stem and diencephalic areas controlling the micturition reflex.Journal of Comparative Neurology 106, 213–45.
Taxi, J. (1961) Etude de l'ultrastructure des zones synaptiques dans les ganglions sympathiques de la grenouille.Comptes Rendus de l'Academie des Sciences 252, 174–6.
Uchizono, K. (1965) Characteristics of excitatory and inhibitory synapses in the central nervous system of the cat.Nature 207, 642–43.
Usui, T. (1975) The fine structure of neurons and synapses in the intermediolateral nucleus (ILN) of the rat thoracic cord.Tenth International Congress of Anatomy, Tokyo p. 150 (abstract).
Westman, J. andBowsher, D. (1971) The fine structure of ‘non-specific’ grey matter (Laminae V and VII) in the cat spinal cord.Experimental Brain Research 12, 379–88.
Yamamoto, T., Satomi, H., Ise, H., Takatama, H. andTakahashi, K. (1978) Sacral spinal innervations of the rectal and vesical smooth muscles and the sphincteric striated muscles as demonstrated by the horseradish peroxidase method.Neuroscience Letters 7, 41–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brown, H.K., Nolan, M.F. Ultrastructure and quantitative synaptology of the sacral parasympathetic nucleus. J Neurocytol 8, 167–179 (1979). https://doi.org/10.1007/BF01175559
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01175559