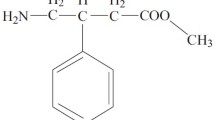
Abstract
Tests were conducted on the relative bioavailability of single lots of 16 different 250-mg tetracycline hydrochloride or tetracycline phosphate complex capsule products commercially available in the United States. All products met the specifications of the Food and Drug Administration for antibiotic certification. Complete crossover studies were carried out with each of the 16 products administered to 16 human volunteers over a period of 16 weeks. The urinary excretion of the tetracycline was followed for 72 hr after the single dose administration. No statistically significant differences were observed, in the cumulative percent of tetracycline excreted, between any of the products or any of the subjects employed in the study.
Similar content being viewed by others
References
Report of the Ad Hoc Committee on Drug Product Selection of the Academy of General Practice of Pharmacy and the Academy of Pharmaceutical Sciences.J. Am. Pharm. Assoc. NS13 (1973).
G. H. Schneller. Hazard of therapeutic nonequivalency of drug products.J. Am. Pharm. Assoc. NS9: 455 (1969).
H. Macdonald, F. Pisano, J. Berger, A. Dornbush, and E. Pelcak. Physiological availability of various tetracyclines.Drug Inform. Bull. 3: 76 (1969).
W. H. Barr, L. M. Gerbracht, K. Letcher, M. Plaut, and N. Strahl. Assessment of the biologic availability of tetracycline products in man.Clin. Pharmacol. Ther. 13: 97 (1972).
C. M. Davis, J. V. Vandersarl, and E. W. Kraus. Tetracycline inequivalence: The importance of 96-hour testing.Am. J. Med. Sci. 265: 69 (1973).
J. Tuomisto and P. Mannisto. Cross-over study of ten tetracycline preparations.Eur. J. Clin. Pharmacol. 6: 64 (1973).
Guidelines for Biopharmaceutical Studies in Man. A.Ph.A. Academy of Pharmaceutical Sciences, Washington, D.C., 1972.
M. Lever. Improved fluorometric determination of tetracycline.Biochem. Med. 6: 216 (1972).
E. J. Williams. Experimental designs balanced for the estimation of residual effects of treatments.Austral. J. Sci. Res. A 2: 149 (1949).
J. M. Jaffe, J. L. Colaizzi, R. I. Poust, and R. H. McDonald. Effect of altered urinary pH on tetracycline and doxycycline excretion in humans.J. Pharmacokin. Biopharm. 1: 267 (1973).
Author information
Authors and Affiliations
Additional information
Supported in part by a Contract from the Tennessee Department of Public Health and by USPHS Grant HL-09495.
Rights and permissions
About this article
Cite this article
Meyer, M.C., Dann, R.E., Whyatt, P.L. et al. The bioavailability of sixteen tetracycline products. Journal of Pharmacokinetics and Biopharmaceutics 2, 287–297 (1974). https://doi.org/10.1007/BF01061403
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01061403