Abstract
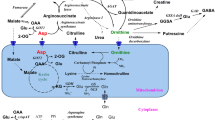
Glucagon increased the activities of alanine amino transferase (AAT), fructose-1∶6-bisphosphatase (fru-P2ase) and glucose-6-phosphatase (G-6-Pase) in goat brain tissue by about 100%, 150% and 50% respectively. These increase in activities were reversed by β-antagonists propranolol. Well known α-agonist and antagonist like phenylephrine and phenoxybenzamine also increased AAT and G-6-Pase activities and these increased activities were reversed by propranolol. Phenylephrine and phenoxybenzamine however did not increase brain Fru-P2ase activity.
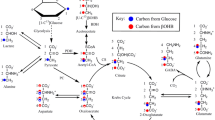
However the most interesting finding is that cerebral cortical slices could produce glucose from alanine and this glucose production was enhanced by glucagon, phenylephrine and phenoxybenzamine. Propranolol reversed the effects of these agonists and antagonist to a great extent. From all these experiments we suggest brain to be a gluconeogenic organ although much less efficient than liver.
Similar content being viewed by others
References
Scrutton MC, Utter MF: The regulation of glycolysis and gluconeogenesis in animal tissues. Ann Rev Biochem 37:249–302, 1968
Phillips ME, Coxon VR: Incorporation of Isotopic Carbon into cerebral glycogen from non-glucose substrates. Biochem J 46: 185–189, 1975
Felig P, Pozefsky T, Marliss E, Cahill GF Jr: Alanine: Key role in gluconeogenesis. Science 167:1003–1004, 1970
Begum NA, Datta AG: Effect of glucagon and some other alpha and beta adrenergic agonists and antagonists on alanine aminotransferase of perfused rat liver. Mol Cell Biochem 105:7–13, 1991
Begum NA, Datta AG: Effect of adrenergic agonists and antagonists on alanine amino transferase, fructose-1,6-bisphosphatase and glucose production in hepatocytes. Mol Cell Biochem 113:2: 93–103, 1992
Orlicky J, Ruscak M, Ruscakova D, Hager H: Two forms of alanine aminotransferase in rat brain during ontogeny. J Neurochem 32:1551–1558, 1979
Majumder AL, Eisenberg F Jr: Unequivocal demonstration of fructose-1,6-bisphosphatase in mammalian brain. Proc Natl Acad Sci USA 74:8:3222–3225, 1977
Anchors JM, Haggerty DF, Karnovsky ML: Cerebral glucose-6-phosphatase and the movement of 2 deoxy-D-glucose across cell membranes. J Biol Chem 252:7035–7041, 1977
Segal JL, Matsuzawa T: L-alanine aminotransferase (rat liver). In: H Tabor, GW Tabor (eds.) Methods of Enzymology. Academic Press, New York, London, 1970, pp 153–159, Vol 17A
Ames BN: Assay of inorganic phosphate, total phosphate and phosphatases. In: EF Neufeld, V Ginsburg (eds.) Methods in Enzymology. Academic Press, New York, London, 1966, pp 115–118, Vol VIII
Zakim D, Vessey DA: Techniques for the characterization of UDP-glucuronyltransferase, glucose-6-phosphatase and other tightly-bound microsomal enzymes. In: G David (ed.) Methods of Biochemical Analysis An Interscience(R) Publication. John Wiley & Sons, New York, London, Sydney, Toronto, 1973, pp 1–37, Vol 21
Bernt E, Bergmeyer HU: Determination of glucose with glucose oxidase and peroxidase In: HU Bergmeyer (ed.) Methods of Enzymatic Analysis. Academic Press, New York, 1965, pp 123–129
Liu F, Fromm HJ: Purification and characterization of fructose-1,6-bisphosphatase from bovine brain. Arch Biochem Biophys 260:2:609–615, 1988
Hevor T, Gayet J: Stimulation of fructose-1,6-bisphosphatase activity and synthesis in the cerebral cortex of rats submitted to the convulsant methionine sulfoximine. J Neurochem 36:949–958, 1981
Prasannan KG, Subramanyam K: Effects of insulin on the synthesis of glycogen in cerebral cortical slices of alloxan diabetic rats. Endocrinology 82:1–6, 1968
Schaftingen EV: Fructose-2,6-bisphosphate. In: A Meister (ed.) Advances in Enzymology. An Interscience(R) Publication. John Wiley & Sons, New York, Chichester, Brisbane, Toronto, Singapore, 1987, pp 315–395, Vol 59
Exton JH, Robison JA, Sutherland EW, Park CR: Studies on the role of adenosine 3′5′ monophosphate in the hepatic actions of glucagon and catecholamines. J Biol Chem 246:6166–6177, 1971
Exton JH: Mechanisms involved in effects of catecholamines on liver carbohydrate metabolism. Biochem Pharmacol 28:2237–2240, 1979
Hutson NJ, Brumley FT, Assimacopoulos FD, Harper SC, Exton JH: Studies on the α-adrenergic activation of hepatic glucose output. J Biol Chem 251:5200–5208, 1976
Manchon GG, Ayuso MS, Parrilla R: Control of gluconeogenesis: Role of fatty acids in the α-adrenergic response. Biochimica et Biophysica Acta 972:192–199, 1988
Petitti N, Etgen AM: Protein kinase C and phospholipase C mediate α1- and β-adrenoceptor intercommunication in rat hypothalamic slices. J Neurochem 56:628–635, 1991
Hoosein MN, Gurd RS: Identification of glucagon receptors in rat brain. Proc Natl Acad Sci USA 81:4368–4372, 1984
Das A, Pansky B, Budd GC: Glucagon-like immunoreactivity in mouse and rat retina. Neurosci Lett 60:215–218, 1985
Durango RF, Sanchez D, Cruz AF: Identification of glucagon receptors in rat retina. J Neurochem 54:1233–1237, 1990
Tager H, Hohenboken M, Markese J, Dinerstein RJ: Identification and localization of glucagon-related peptides in rat brain. Proc Natl Acad Sci 77:6229–6233, 1980
Triepel J, Elger KH, Forssmann GW: Glicentin-immunoreactive perikarya and varicosities in the guineapig central nervous system. Neurosci Lett 30:585–589, 1982
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bhattacharya, S.B., Datta, A.G. Is brain a gluconeogenic organ?. Mol Cell Biochem 125, 51–57 (1993). https://doi.org/10.1007/BF00926834
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00926834