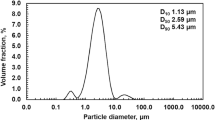
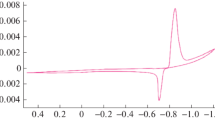
Abstract
Using the rotating disc method, the rates of dissolution of natural monoclinic pyrrhotite, FeS1.14, in oxygen-free aqueous solutionsS([H+]=0.1, [Na+]=0.9, [ClO −4 ]=1.0 mol kg−1) were determined. In the temperature range 40–90 °C the dissolution reaction occurs under kinetic control; the activation energy being 14±1 kcal mol−1 (50±5 kJ mol−1).
Zusammenfassung
Die Auflösungsgeschwindigkeit von natürlichem monoklinen Pyrrhotin, FeS1.14, wurde in sauerstofffreien LösungenS([H+]=0.1, [Na+]=0.9, [ClO −4 ]=1.0 mol kg−1) mit Hilfe der Methode der rotierenden Scheibe bestimmt. Im Temperaturbereich von 40–90° erfolgt die Auflösungsreaktion kinetisch kontrolliert, wobei eine Aktivierungsenergie von 14±1 kcal mol−1 (59±5 kJ mol−1) gefunden wurde.
Similar content being viewed by others
References
Tewari P. J., Campbell A. B., J. Phys. Chem.80, 1844 (1976).
Ingraham T. R., Parsons H. W., Cabri, L. J., Can Metall. Q.11, 407 (1972).
Subramian K. N., Stratigakos E. S., Jennings P. H., Can. Metall. Q.11, 425 (1972).
Dutrizac J. E., Macdonald R. J. C., Minerals Sci. Engng.6, 59 (1974).
Nicol M. J.,Scott P. D., Journal of the South African Institute of Mining and Metallurgy1979, 298.
Heindl R., Gamsjäger H., Monatsh. Chem.108, 86 (1977).
Arnold R. G., Reichen L. E., Amer. Min.47, 105 (1962).
Levich V. G., Physicochemical Hydrodynamics. Englewood Cliffs, N.J.: Prentice-Hall. 1972.
Pleskov Yu. V., Filinovsky V. Yu., Rotating Disc Electrode. New York: Consultants Bureau. 1976.
Zembura Z., Fuliński A., Electrochim. Acta10, 958 (1965).
Weast R. C., (Ed.), Handbook of Chemistry and Physics, 48th ed. Cleveland: The Chemical Rubber Co. 1968.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bugajski, J., Gamsjäger, H. The kinetics of the dissolution of monoclinic pyrrhotite in aqueous acidic solutions. Monatsh Chem 113, 1087–1092 (1982). https://doi.org/10.1007/BF00808622
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00808622