Summary
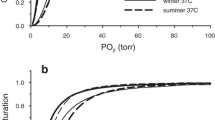
The presence of hemoglobin inAnisops assimilis has been demonstrated to be a vital factor in the physiology of this organism. The hemoglobin is composed of heterogeneous subunits which aggregate upon deoxygenation. This association-dissociation equilibrium confers a steep gradient (n H∼6) to the oxygen equilibrium curve and a low oxygen affinity (P 50∼40 mmHg). Oxygen bound by the hemoglobin is released into a gas bubble enabling the bug to regulate its density around that of water. Thus, energy is conserved during a dive, allowing the animal to remain in mid-water for long periods. This adaptive feature has facilitated the exploitation of an ecological niche available to few other organisms.
Similar content being viewed by others
References
Allison AC, Cecil R, Charlewood PA, Gratzer WB, Jacobs S, Snow WS (1960) Haemoglobin of the lamprey,Lampetra fluviatilis. Biochim Biophys Acta 42:43–48
Andrews P (1964) Estimation of the molecular weights of proteins by Sephadex gel-filtration. Biochem J 91:222–233
Antonini E, Rossi-Fanelli A, Caputo A (1962) Studies on chlorocruorin. I. The oxygen equilibrium ofSpirographis chlorocruorin. Arch Biochem Biophys 97:336–342
Antonini E, Wyman J, Bellelli L, Rumen W, Siniscalco M (1964) The oxygen equilibrium of some lamprey haemoglobins. Arch Biochem Biophys 105:404–408
Bergtrom G (1977) Partial characterization of haemoglobin of the bug,Buenoa confusa. Insect Biochem 7:313–316
Bergtrom G, Gittelman S, Laufer H, Ovitt C (1976) Haemoglobin synthesis inBuenoa confusa (Hemiptera). Insect Biochem 6:595–600
Briehl RW (1963) The relation between the oxygen equilibrium and aggregation of subunits in Lamprey haemoglobin. J Biol Chem 238:2361–2366
Chung MCM, Ellerton HD (1979) The physico-chemical and functional properties of extracellular respiratory haemoglobins and chlorocruorins. Prog Biophys Mol Biol 35:53–102
Dacie JV, Lewis SM (1975) Practical haematology. Livingstone, Edinburgh
Dinulescu G (1932) Recherches sur la biologie des Gastrophiles. Anatomie, physiologie, cycle évolutif. Ann Sci Nat (Paris) 15:1–183
Ege R (1915) On the respiratory function of the air stores carried by some aquatic insects (Corixidae, Dytiscidae, Notonecta). Z Allg Physiol 17:81–124
Falcioni M, Fortuna G, Giardina B, Brunori M, Wyman J (1977) Functional properties of partially oxidized trout hemoglobins. Biochim Biophys Acta 490:171–177
Florkin M (1960) Unity and diversity in biochemistry. Pergamon, Oxford
Florkin M, Lefebvre L, Laurent Y (1941) Oxygen dissociation curve ofChironomus haemoglobin. Acta Biol Belg 2:305–311
Hendrickson WA, Love WE (1971) Structure of lamprey haemoglobin. Nat N Biol 232:197–203
Hill AV (1910) The possible effects of the aggregation of haemoglobin on its dissociation curve. J Physiol (Lond) 40:IV-VII
Hungerford HB (1922) Oxyhaemoglobin present in backswimmerBuenoa margaritacea Bueno (Hemiptera). Can Entomol 54:262–263
Imai K, Yonetani T (1977) The haemoglobin-oxygen equilibrium associated with subunit dissociation. I. An approach with the Hill scheme. Biochim Biophys Acta 490:164–170
Keilin D, Wang YL (1946) Haemoglobin ofGastrophilus larvae —purification and properties. Biochem J 40:855–866
Krogh A (1941) The comparative physiology of respiratory mechanisms. Dover, New York
Lykkeboe G, Johansen K (1978) An O2-Hb ‘Paradox’ in frog blood? Respir Physiol 35:119–127
Maizel JV (1969) Acrylamide gel electrophoresis of proteins and nucleic acids. In: Habel K, Salzman NP (eds) Fundamental techniques in virology. Academic Press, New York, pp 125–139
Mangum CP (1976) The oxygenation of hemoglobin in lugworms. Physiol Zöol 49:85–99
Miller PL (1964a) Possible function of haemoglobin inAnisops. Nature 201:1052
Miller PL (1964b) The possible role of haemoglobin inAnisops andBuenoa. Proc R Ent Soc (Lond) Ser A 39:166–175
Miller PL (1966) Function of haemoglobin in buoyancy inAnisops pellucens. J Exp Biol 44:529–543
Mizukami H, Vinogradov SN (1972) Oxygen association equilibria ofGlycera haemoglobins. Biochim Biophys Acta 285:314–319
Perutz MF (1978) Haemoglobin structure and respiratory transport. Sci Am 239:68–86
Peters K, Richards FM (1977) Chemical cross-linking: reagents and problems in studies of membrane structure. Ann Rev Biochem 46:523–551
Poisson R (1926) L'Anisops producta Fieb (Hemiptera Notonectidae). Observations sur son anatomie et sa biologie. Arch Zool Exp Gén 65:181–208
Riggs A (1965) Functional properties of hemoglobins. Physiol Rev 45:619–673
Terwilliger RC (1975) Oxygen equilibrium and subunit aggregation of a holothurian haemoglobin. Biochim Biophys Acta 386:62–68
Walshe BM (1950) The function of haemoglobin inChironomus plumosus under natural conditions. J Exp Biol 27:73–95
Weber K, Osborn M (1969) The reliability of molecular weight determination of SDS-polyacrylamide gel electrophoresis. J Biol Chem 244:4406–4412
Weber RE (1980) Functions of invertebrate hemoglobins with special reference to environmental hypoxia. Am Zool 20:79–101
Wigglesworth VB (1943) The fate of haemoglobin inRhodnius prolixus (Hemiptera) and other blood-sucking Arthropods. Proc R Soc Lond [Biol] 131:313–339
Wigglesworth VB (1972) The principles of insect physiology, 7th ed. Chapman and Hall, London
Wolvekamp HP (1955) Die physikalische Kieme der Wasserinsekten. Experienta 11:294–301
Wyman J (1948) Heme proteins. Adv Protein Chem 4:407–531
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wells, R.M.G., Hudson, M.J. & Brittain, T. Function of the hemoglobin and the gas bubble in the backswimmerAnisops assimilis (Hemiptera: Notonectidae). J Comp Physiol B 142, 515–522 (1981). https://doi.org/10.1007/BF00688984
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00688984