Summary
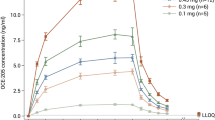
The pharmacokinetics and haematological effects of 1-deamino-8-D-arginine vasopressin (desmopressin, DDAVP) after intravenous, subcutaneous and intranasal administration has been studied in man. Using a sensitive, specific radioimmunoassay for DDAVP, the AUC was determined for each route of administration. It was not significantly different for the i.v. and s.c. routes. There was no effect of the route on the plasma half-life of DDAVP which ranged from 2.7 to 4.6 h. Absorption of DDAVP after intranasal (i.n.) administration was poor. Based on AUC data, bioavailability via the two s.c. methods and the i.n. route was 112%, 94% and 2%, respectively.
DDAVP has a pronounced effect on coagulation and fibrinolytic parameters, causing a 4.0-(i.v.), 2.9- (s.c.), 3.1- (s.c.; 40 µg/ml) and 1.2- (i.n.) fold increase in factor VIII: Ag. The corresponding effect on tissue-type plasminogen activator (t-PA) was 1.9- (i.v.), 1.3- (s.c.), 2.2- (s.c.; 40 µg/ml) and 1.0- (i.n.) increase over the basal value. There was also a 1.4- to 1.6-fold increase in leukocyte count 4 h after s.c. and i.v. DDAVP. At plasma DDAVP levels greater than 300 pg/ml no correlation was found between the AUC and the maximum plasma DDAVP and biological response, which indicates a ceiling limit for exogenous stimulation of the coagulation and fibrinolytic systems.
Similar content being viewed by others
References
Andersson KE, Arner B, Hedner P, Mulder JL (1972) Effects of 8-lysine vasopressin and synthetic analogues on release of ACTH. Acta Endocrinol 69: 640–648
Cash JD, Gader AMA, DaCosta J (1974) The release of plasminogen activator and factor VIII to lysine vasopressin, arginine vasopressin, 1-desamino-8-D-vasopressin, angiotensin and oxytocin in man. Br J Haematol 15: 363–364 (abstract)
Cream JJ (1968) Prednisolone-induced granulocytosis. Br J Haematol 15: 259–267
DeSio L, Mariani G, Muzzuconi MG, Chistolini M, Tirindelli MC, Mandelli F (1985) Comparison between subcutaneous and intravenous DDAVP in mild and moderate hemophilia. Thromb Haemost 54: 387–389
Edwards CRW, Kitau MJ, Chard T, Besser GM (1973) Vasopressin analogue DDAVP in diabetes insipidus: Clinical and laboratory studies. Br Med J 3: 375–378
Gader AM, DaCosta J, Cash JD (1973) A new vasopressin analogue and fibrinolysis. Lancet 2: 1417–1418
Grossman A, Fabri A, Goldberg PL, Besser GM (1980) Two new modes of desmopressin (DDAVP) administration. Br Med J 2: 1215
Hammer M, Vilhardt H (1985) Peroral treatment of diabetes insipidus with a polypeptide hormone analog, desmopressin. J Pharmacol Exp Ther 224: 754–760
Harris AH, Nilsson IM, Wagner ZG, Alkner U: Intranasal administration of peptides: Nasal deposition, biological response, and absorption of desmopressin. J Pharm Sci 75: 1085–1088, 1986
Köhler M, Hellstern P, Reiter B, von Blohn G, Wenzel E (1984) The subcutaneous administration of the vasopressin analogue 1-deamino-8-D-arginine vasopressin in patients with von Willebrand's disease and haemophilia. Klin Wochenschr 62: 543–548
Köhler M, Hellstern P, Miyashita C, von Blohn G, Wenzel E (1986) Comparative study of intranasal, subcutaneous, and intravenous administration of desamino-D-arginine vasopressin (DDAVP). Thromb Haemost 55: 108–111
Mannucci PM, Pareti FI, Holmberg L, Nilsson IM, Ruggeri ZM (1976) Studies on the prolonged bleeding time in von Willebrand's disease. J Lab Clin Med 88: 662–671
Mannucci PM, Ruggeri ZM, Pareti FI, Capitanio A (1977a) DDAVP: A new pharmaceutical approach to the management of haemophilia and von Willebrand's disease. Lancet 1: 869–872
Mannucci PM, Ruggeri ZM, Pareti FI, Capitanio A (1977b) DDAVP in haemophilia. Lancet 2: 1171–1172
Mannucci PM, Canciani MT, Rota L, Donovan BS (1981) Response of factor VIII/ von Willebrand factor to DDAVP in healthy subjects, and patients with haemophilia A and von Willebrand's disease. Br J Haematol 47: 283–293
Mannucci PM (1986) Desmopressin (DDAVP) for treatment of disorders of haemostasis. In: Progress in hemostasis and thrombosis. Grune & Stratton, pp 19–45
Mörsdorf S, Köhler M, Leipnitz G, Wenzel E (1988) The clinical significance of different routes of desmopressin (DDAVP) administration in various bleeding disorders. Folia Haematol (in press)
Pullan PT, Burger HT, Johnston CJ (1978) Pharmacokinetics of 1-desamino-8-D-arginine vasopressin (DDAVP) in patients with central diabetes insipidus. Clin Endocrinol 9: 273–278
Sörensen PS, Vilhardt H, Gjerris F, Warberg J (1984) Impermeability of the blood-cerebrospinal fluid barrier to 1-deamino-8-D-arginine vasopressin (DDAVP) in patients with acquired, communicating hydrocephalus. Eur J Clin Invest 14: 435–439
Vavra K, Machova, Holocek V, Cort JH, Zaoral M, Sorm F (1968) Effect of a synthetic analogue of vasopressin in animals, and in patients with diabetes insipidus. Lancet 1: 948–952
Vicente V, Alberca I, Mannucci PM (1985) High levels of circulating von Willebrand factor inhibit the release of this protein but not of plasminogen activator after DDAVP. Thromb Res 38: 101–105
Wahlberg TB, Blombäck M, Magnusson D (1984) Influence of sex, blood group, secretor character, smoking habits, acetylsalicyliyc acid, oral contraceptives, fasting and general health state on blood coagulation variables in randomly selected young adults. Haemostasis 14: 312–319
Zaoral M, Kolc J, Sorm F (1967) Synthesis of 1-deamino-8-D-aminobutyrine vasopressin, 1-deamino-8-D-lysine vasopressin and 1-deamino-8-D-arginine vasopressin. Collect Czech Chem Commun 32: 1250–1257
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. P.G.Scheurlen on the occasion of his 65th birthday
Rights and permissions
About this article
Cite this article
Köhler, M., Harris, A. Pharmacokinetics and haematological effects of desmopressin. Eur J Clin Pharmacol 35, 281–285 (1988). https://doi.org/10.1007/BF00558266
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558266