Abstract
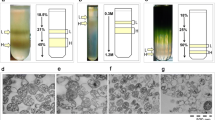
Glyoxysomes isolated from castor-bean (Ricinus communis L.) endosperm were treated with water, 0.2 M KCl, 1 M KCl, or 0.1 M Na2CO3. Glyoxysomal sacs, i.e. membranes which retained some visible matrix, resulted from the treatments with water and KCl. Glyoxysomal ghosts, i.e. intact membranes free of matrix, were only obtained following treatment with carbonate. The ghosts were free of activities of matrix enzymes, particularly palmitoyl-CoA oxidation, isocitrate dehydrogenase (EC 1.1.1.42) and isocitrate lyase (EC 4.1.3.1), and contained only negligible amounts of malate synthase (EC 4.1.3.2), malate dehydrogenase (EC 1.1.1.37), β-hydroxyacyl-CoA dehydrogenase (EC 1.1.1.98) and catalase (EC 1.11.1.6). Distribution and appearance of membrane-associated particles in the protoplasmic and ectoplasmic faces of freeze-fracture replicas of the glyoxysomal membrane were the same in intact tissue, isolated glyoxysomes, and ghosts. Membranes purified by treatment with 0.2 M KCl or 0.1 M carbonate catalyzed the reduction of cytochrome-c when NADH or NADPH was provided as the electron donor. β-Oxidation, localized in the matrix, could be linked to reduction of cytochrome-c or ferricyanide when purified membranes were combined with the matrix supernatant. Cytochrome-c could also be reduced by coupling enzyme activities in the matrix, NADP-isocitrate dehydrogenase or malate dehydrogenase, with those of the membrane. These results indicate that electrons from β-oxidation, malate oxidation or isocitrate oxidation can be transferred directly to the redox components of the glyoxysomal membrane. We, therefore, conclude that any NADH and NADPH formed by enzymes in the matrix can be recycled continuously within the organelle.
Similar content being viewed by others
Abbreviations
- EF:
-
ectoplasmic face
- ER:
-
endoplasmic reticulum
- PF:
-
protoplasmic face
References
Alexson, S.E.H., Fujiki, Y., Shio, H., Lazarow, P.B. (1985) Partial disassembly of peroxisomes. J. Cell Biol. 101, 294–305
Angermuller, S., Fahimi, H.D. (1981) Selective cytochemical location of peroxidase, cytochrome oxidase and catalase in rat liver with 3,3′-diaminobenzidine. Histochemistry 71, 34–44
Bieglmayer, C., Graf, J., Ruis, H. (1973) Membranes of glyoxysomes from castor bean endosperm. Enzymes bound to purified-membrane preparation. Eur. J. Biochem. 37, 553–562
Bradford, M. (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–252
Branton, D., Bullivant, S., Gilula, N.B., Karnovsky, M.J., Moor, H., Mühlethaler, K., Northcote, D.H., Packer, L., Satir, B., Satir, P., Speth, V., Staehelin, L.A., Steere, R.L., Weinstein, R.S. (1975) Freeze-etching nomenclature. Science 190, 54–56
Bronfman, M., Beaufay, H. (1973) Alteration of subcellular organelles induced by compression. FEBS Lett. 36, 163–168
Cooper, T.G., Beevers, H. (1969a) Mitochondria and glyoxysomes from castor bean endosperm. Enzyme constituents and catalytic capacity. J. Biol. Chem. 244, 3507–3513
Cooper, T.G., Beevers, H. (1969b) β-Oxidation in glyoxysomes from castor bean endosperm. J. Biol. Chem. 244, 3514–3520
Donaldson, R.P. (1982) Nicotinamide cofactors (NAD and NADP) in glyoxysomes, mitochondria, and plastids isolated from castor bean endosperm. Arch. Biochem. Biophys. 215, 274–279
Donaldson, R.P. (1986) The coupling of beta-oxidation and the glyoxylate cycle to electron transport in glyoxysomes. (Abstr.) Eur. J Cell Biol. 41 (Suppl.) 14), 12
Donaldson, R.P., Beevers, H. (1978) Organelle membranes from germinating castor bean endosperm. I. A comparison of purified endoplasmic reticulum, glyoxysomes, and mitochondria. Protoplasma 97, 317–327
Donaldson, R.P., Tully, R.E., Young, O.A., Beevers, H. (1981) Organelle membranes from germinating castor bean endosperm. II. Enzymes, cytochromes, and permeability of the glyoxysomal membrane. Plant Physiol. 67, 21–25
Fujiki, Y., Fowler, S., Shio, H., Hubbard, A.L., Lazarow, P.B. (1982a) Polypeptide and phospholipid composition of the membrane of rat liver peroxisomes, comparison with endoplasmic reticulum and mitochondrial membranes. J. Cell Biol. 93, 103–110
Fujiki, Y., Hubbard, A.L., Fowler, S., Lazarow, P.B. (1982b) Isolation of intracellular membranes by means of sodium carbonate treatment, application to endoplasmic reticulum. J. Cell Biol. 93, 97–102
Gerhardt, B.P., Beevers, H. (1970) Developmental studies on glyoxysomes in Ricinus endosperm. J. Cell Biol. 44, 94–102
Hicks, D.B., Donaldson, R.P. (1982) Electron-transport in glyoxysomal membranes. Arch. Biochem. Biophys. 215, 280–288
Huang, A.H.C., Beevers, H. (1973) Localization of enzymes within microbodies. J. Cell Biol. 58, 379–389
Huttinger, M., Pavelka, M., Goldenberg, H., Kramar, R. (1981) Membranes of rat liver peroxisomes. Histochemistry 71, 259–267
Koller, W., Kindl, H. (1977) Glyoxylate cycle enzymes of the glyoxysomal membrane from cucumber cotyledons. Arch. Biochem. Biophys. 181, 236–248
Lord, J.M., T. Kagawa, T., Beevers, H. (1972) Intracellular distribution of enzymes of the cytidine diphosphate choline pathway in castor bean endosperm. Proc. Natl. Acad. Sci. USA 69, 2429–2432
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J. (1951) Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275
Luster, D.G., Donaldson, R.P. (1987) Glyoxysomal membrane electron transport proteins. In: Peroxisomes in biology and medicine, pp. 189–193, Fahimi, H.D., Sies, H., eds., Springer-Verlag, Heidelberg
Miernyk, J.A., Trelease, R.N., Choinski, J.S. (1979) Malate synthase activity in cotton and other undergerminated oil-seeds. A survey. Plant Physiol. 63, 1068–1071
Miflin, B., Beevers, H. (1974) Isolation of intact plastids from a range of plant tissues. Plant Physiol. 53, 870–874
Overath, P., Raufuss, E.M., Stoffel, W., Ecker, W. (1967) The induction of the enzymes of fatty acid degradation in E. coli. Biochem. Biophys. Res. Commun. 29, 28–33
Robinson, J.B., Srere, P.A. (1985) Organization of Krebs tricarboxylic acid cycle enzymes in mitochondria. J. Biol. Chem. 260, 10800–10805
Spurr, A.R. (1969) A low viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43
Steere, R.L. (1981) Preparation of freeze-fracture, freeze-etch, freeze-dry and frozen surface replica specimens for electron microscopy in the Denton DFE-2 and DFE-3 freeze-etch units. In: Current Trends in Morph. Tech., vol. II, pp. 131–181, Johnson, J.E., Jr., ed. CRC Press, Boca Raton, Fla. USA
Trelease, R.N., Becker, W.M., Burke, J.J. (1974) Cytochemical localization of malate synthase in glyoxysomes. J. Cell Biol. 60, 483–495
Vigil, E.L. (1970) Cytochemical and developmental changes in microbodies (glyoxysomes) and related organelles of castor bean endosperm. J. Cell Biol. 46, 435–454
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fang, T.K., Donaldson, R.P. & Vigil, E.L. Electron transport in purified glyoxysomal membranes from castor-bean endosperm. Planta 172, 1–13 (1987). https://doi.org/10.1007/BF00403023
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00403023