Abstract
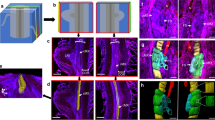
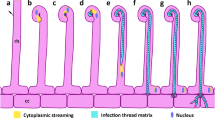
The location and topography of infection sites in soybean (Glycine max (L.) Merr.) root hairs spot-inoculated with Rhizobium japonicum have been studied at the ultrastructural level. Infections commonly developed at sites created when the induced deformation of an emerging root hair caused a portion of the root-hair cell wall to press against an adjacent epidermal cell, entrapping rhizobia within the pocket between the two host cells. Infections were initiated by bacteria which became embedded in the mucigel in the enclosed groove. Infection-thread formation in soybean appears to involve degradation of mucigel material and localized disruption of the outer layer of the folded hair cell wall by one or more entrapped rhizobia. Rhizobia at the site of penetration are separated from the host cytoplasm by the host plasmalemma and by a layer of wall material that appears similar or identical to the normal inner layer of the hair cell wall. Proliferation of the bacteria results in an irregular, wall-bound sac near the site of penetration. Tubular infection threads, bounded by wall material of the same appearance as that surrounding the sac, emerge from the sac to carry rhizobia roughly single-file into the hair cell. Growing regions of the infection sac or thread are surrounded by host cytoplasm with high concentrations of organelles associated with synthesis and deposition of membrane and cell-wall material. The threads follow a highly irregular path toward the base of the hair cell. Threads commonly run along the base of the hair cell for some distance, and may branch and penetrate into subjacent cortical cells at several points in a manner analagous to the initial penetration of the root hair.
Similar content being viewed by others
References
Bauer, W.D. (1981) Infection of legumes by rhizobia. Annu. Rev. Plant Physiol. 32, 407–449
Berry, A.M., Torrey, J.G. (1983) Root hair deformation in the infection process of Alnus rubra. Can. J. Bot. 61, 2863–2876
Bhuvaneswari, T.V., Bhagwat, A.A., Bauer, W.D. (1981) Transient susceptibility of root cells in four common legumes to nodulation by rhizobia. Plant Physiol. 68 1144–1149
Bhuvaneswari, T.V., Turgeon, B.G., Bauer, W.D. (1980) Early events in the infection of soybean Glycine max (L.) Merr. by Rhizobium japonicum. I. Localization of infectible root cells. Plant Physiol. 66, 1027–1031
Bieberdorf, F.W. (1938) The cytology and histology of root nodules of some leguminosae. J. Am. Soc. Agron. 30, 375–389
Callaham, D.A., Torrey, J.G. (1981) The structural basis for infection of root hairs of Trifolium repens by Rhizobium. Can. J. Bot. 59, 1647–1664
Calvert, H.E., Pence, M., Pierce, M., Malik, N.S.A., Bauer, W.D. (1984) Anatomical analysis of the development and distribution of Rhizobium infections in soybean roots. Can. J. Bot. (in press)
Chandler, M.R. (1978) Some observations of infection of Arachis hypogaea L. by Rhizobium. J. Exp. Bot. 29, 749–755
Chandler, M.R., Date, R.A., Roughley, R.J. (1982) Infection and root-nodule development in Stylosanthes species by Rhizobium. J. Exp. Bot. 33, 47–57
Dart, P.J. (1974) Development of root nodule symbiosis. The infection process. In: The biology of nitrogen fixation, pp. 381–429, Quispel, A., ed. North Holland, Amsterdam
Dart, P.J. (1977) Infection and development of leguminous nodules. In: A treatise on dinitrogen fixation, pp. 367–372, Hardy, R.W.F., Silver, W.S., eds. Wiley, New York
Fahraeus, G. (1957) The infection of clover root hairs by nodule bacteria studied by a simple glass slide technique. J. Gen. Microbiol. 16, 374–381
Fahraeus, G., Ljunggren, H., (1967) The possible significance of pectic enzymes in root hair infection by nodule bacteria. Physiol. Plant. 12, 145–154
Foster, R.C. (1981) The ultrastructure and histochemistry of the rhizosphere. New Phytol. 89, 263–273
Haak, A. (1964) Über den Einfluss der Knöllchenbakterien auf die Wurzelhaare von Leguminosen und Nichtleguminosen. Zentralbl. Bakteriol. Parasitenkd. Infektionskr. Abt. 2 117, 343–361
Higashi, S. (1966) Electron microscopic studies on the infection thread developing in the root hair of Trifolium repens L. infected with Rhizobium trifolii. J. Gen. Appl. Microbiol. 12, 147–156
Higashi, S., Abe, M. (1980) Scanning electron microscopy of Rhizobium trifolii infection sites on root hairs of white clover. Appl. Environ. Microbiol. 40, 1094–1099
Hubbell, D.H. (1981) Legume infection by Rhizobium: a conceptual approach. BioScience 31, 832–837
Hubbel, D.H., Morales, V.M., Umali-Garcia, M. (1978) Pectolytic enzymes in Rhizobium. Appl Environ. Microbiol. 35, 210–213
Ljunggren, H. (1969) Mechanism and pattern of Rhizobium invasion into leguminous root hairs. Physiol. Plant., Suppl. V
Martinez-Molina, E., Morales, V.M., Hubbell, D.H. (1979) Hydrolytic enzyme production by Rhizobium. Appl. Environ. Microbiol. 38, 1186–1188
Munns, D.N. (1968) Enzymatic breakdown of pectin and acid-inhibition of the infection of Medicago roots by Rhizobium. Plant and Soil 60, 117–120
Napoli, C.A., Hubbell, D.H. (1975) Ultrastructure of Rhizobium-induced infection threads in clover root hairs. Appl. Microbiol. 30, 1003–1009
Newcomb, W. (1981) Nodule morphogenesis and differentiation. In: Biology of the Rhizobiaceae, pp. 247–297, Giles, K.L., Atherly, A.G., eds. Int. Rev. Cytol, Suppl. 13. Academic Press, New York London
Newcomb, E.H., Bonnett, H.T. (1965) Cytoplasmic microtubule and wall microfibril orientation in root hairs of radish. J. Cell Biol. 27, 575–589
Newcomb, W., McIntyre, L. (1981) Development of root nodules in mung bean (Vigna radiata): a reinvestigation of endocytosis. Can. J. Bot. 59, 2478–2499
Nutman, P.S. (1956) The influence of the legume in root-nodule symbiosis. A comparative study of host determinants and functions. Biol. Rev. (Cambridge). 31, 109–151
Pueppke, S.G. (1983) Rhizobium infection threads in root hairs of Glycine max (L.) Merr., Glycine soja Sieb. & Zucc., and Vigna unguiculata (L.) Walp. Can. J. Microbiol. 29, 69–76
Robertson, J.G., Lyttleton, P., Bullivant, S., Grayston, G.F. (1978) Membranes in lupin root nodules. I. The role of golgi bodies in the biogenesis of infection threads and peribacteroid membranes. J. Cell Sci. 30, 129–149
Sahlman, K., Fahraeus, G. (1963) An electron microscope study of root hair infection by Rhizobium. J. Gen. Microbiol. 33, 425–427
Spurr, A.R. (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43
Stacey, G., Paau, A.S., Brill, W.J. (1980) Host recognition in the Rhizobium-soybean symbiosis. Plant Physiol. 66, 609–614
Tsien, H.C., Dreyfus, B.L., Schmidt, E.L. (1983) Initial stages in the morphogenesis of nitrogen fixing stem nodules of Sesbania rostrata. J. Bacteriol. 156, 888–897
Turgeon, B.G., Bauer, W.D. (1982) Early events in the infection of soybean Rhizobium japonicum. Time course and cytology of the initial infection process. Can. J. Bot. 60, 152–161
Turgeon, B.G., Bauer, W.D. (1983) Spot inoculation of soybean roots with Rhizobium japonicum. Protoplasma 115, 122–128
Verma, D.P.S., Long, S. (1983) The molecular biology of Rhizobium-legume symbiosis. Int. Rev. Cytol. 14, 211–245
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Turgeon, B.G., Bauer, W.D. Ultrastructure of infection-thread development during the infection of soybean by Rhizobium japonicum . Planta 163, 328–349 (1985). https://doi.org/10.1007/BF00395142
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00395142