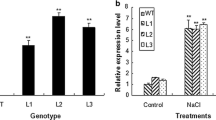
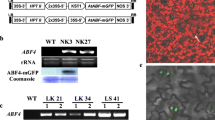
Abstract
The expression of members of two closely related abscisic acid (ABA)-responsive pea protein families, ABR17 and ABR18 (ABA-responsive 17200-Mr and 18100-Mr, respectively), is developmentally, tissueand stress-specifically regulated. Two-dimensional polyacrylamide gel electrophoresis revealed a number of ABR polypeptides on fluorographs of immunoprecipitated translation products of mRNAs, depending on the tissue, stage of development or type of stress. High endogenous ABA, or added ABA, enhanced the accumulation of translatable mRNA for specific ABR members under certain conditions, but high endogenous ABA was not a pre-requisite for accumulation of translatable ABR mRNA. The accumulation of ABR polypeptides was examined by Western blot analysis of acetate-buffer-extracted proteins. In fully expanded, young unstressed leaves, the ABR17 polypeptides (ABR18 polypeptides not detectable) accumulated to markedly higher levels in the epidermis than in the mesophyll. Dehydration stress caused an increased (ABR17) and detectable (ABR18) polypeptide accumulation which occurred predominantly in the epidermis. Detached leaves were used further to characterise factors affecting ABR polypeptide accumulation. An enhanced (ABR17) and detectable (ABR18) polypeptide accumulation occurred in the presence of ABA (10−4 M) but ABR18-polypeptide accumulation required light. The accumulation of both ABR polypeptides was stimulated in the presence of metabolisable and non-metabolisable carbohydrate sources but not in water or glutamine, indicating an osmotic rather than metabolic response. This carbohydrate-stimulated accumulation was markedly enhanced by light but unaffected by 3-(3,4-dichlorophenyl)-1,1-dimethylurea, an inhibitor of photosynthesis, indicating other photoreceptive processes besides photosynthesis were involved. The function of the ABR proteins remains unknown but their accumulation in aging tissues indicates a role in senescence. The results clearly demonstrate highly complex interactions between different environmental and developmental signals leading to the expression of these stressrelated proteins. In light of these results, the induction of protein expression of the newly-termed intracellular pathogenesis-related proteins, to which the ABR proteins are closely related, is discussed.
Similar content being viewed by others
Abbreviations
- ABA:
-
(±)cis, trans-abscisic acid
- ABR17:
-
Mr17200 ABA-responsive protein
- ABR18:
-
Mr-18100 ABA-responsive protein
- 2-D:
-
two-dimensional
- DCMU:
-
3-(3,4-dichlorophenyl)-1,1-dimethylurea
- FW:
-
fresh weight
- IgG:
-
immunoglobulin G
- Mr :
-
apparent molecular mass
- SDS-PAGE:
-
sodium dodecyl sulphate-polyacrylamide gel electrophoresis
References
Ames, G.F-L., Nikaido, K. (1976) Two-dimensional gel electrophoresis of membrane proteins. Biochemistry 15, 616–623
Asselin, A., Grenier, J., Côté, F. (1985) Light-influenced extracellular accumulation of b (pathogenesis-related) proteins in Nicotiana green tissue induced by various chemicals or prolonged floating on water. Can. J. Bot. 63, 1276–1283
Barratt, D.H.P. (1986a) In vitro pod culture of Pisum sativum. Plant Sci. 43,n 223–228
Barratt, D.H.P. (1986b) Modulation by abscisic acid of storage protein accumulation in Vicia faba L. cotyledons cultured in vitro. Plant Sci. 46, 159–167
Barratt, D.H.P., Clark, J.A. (1991) Proteins arising during the late stages of embryogenesis in Pisum sativum L. Planta 184, 14–23
Barratt, D.H.P., Domoney, C., Wang, T.L. (1989a) Purification and partial characterisation of two abscisic-acid-responsive proteins induced in cultured embryos of Pisum sativum L. Planta 180, 16–23
Barratt, D.H.P., Whitford, P.N., Cook, S.K., Butcher, G., Wang, T.L. (1989b) An analysis of seed development in Pisum sativum. VIII. Does abscisic acid prevent precocious germination and control storage protein synthesis? J. Exp. Bot. 40, 1009–1014
Bartels, D., Singh, M., Salamini, F. (1988) Onset of desiccation tolerance during development of the barley embryo. Planta 175, 485–492
Bartels, D., Schneider, K., Terstappen, G., Piatkowski, D., Salamini, F. (1990) Molecular cloning of abscisic acid-modulated genes which are induced during desiccation of the resurrection plant Craterostigma plantagineum. Planta 181, 27–34
Bartels, D., Hanke, C., Schneider, K., Michel, D., Salamini, F. (1992) A desiccation-related Elip-like gene from the resurrection plant Craterostigma plantagineum is regulated by light and ABA. EMBO J. 11, 2761–2778
Bartholomew, D.M., Bartley, G.E., Scolnik, P.A. (1991) Abscisic acid control of rbcS and cab transcription in tomato leaves. Plant Physiol. 96, 291–296
Berge, S.K., Bartholomew, D.M., Quatrano, R.S. (1989) Control of the expression of wheat embryo genes by abscisic acid. In: Molecular basis of plant development, pp. 193–201, Goldberg, R.B., ed. Liss, New York
Blackman, S.A., Wettlaufer, S.H., Obendorf, R.L., Leopold, A.C. (1991) Maturation proteins associated with desiccation tolerance in soybean. Plant Physiol. 96, 868–874
Bostock, R.M., Quatrano, R.S. (1992) Regulation of Em gene expression in rice. Plant Physiol. 98, 1356–1363
Bowles, D.J. (1990) Defense-related proteins in higher plants. Annu. Rev. Biochem. 59, 873–907
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254
Breiteneder, H., Pettenburger, K., Bito, A., Valenta, R., Kraft, D., Rumpold, H., Scheiner, O., Breitenbach, M. (1989) The gene coding for the major birch pollen allergen Betv1 is highly homologous to a pea disease resistance response gene. EMBO J. 8, 1935–1938
Casey, R., Domoney, C., Nielsen, N.C. (1985) Isolation of a cDNA clone for pea (Pisum sativum) seed lipoxygenase. Biochem. J. 232, 79–85
Chiang, C.C., Hadwiger, L.A. (1990) Cloning and characterization of a disease resistance response gene in pea inducible by Fusarium solani. Mol. Plant Microbe Int. 3, 78–85
Claussen, W., Loveys, B.R., Hawker, J.S. (1986) Influence of sucrose and hormones on the activity of sucrose synthase and invertase in detached leaves and leaf sections of eggplants (Solanum melongena). J. Plant Physiol. 124, 345–357
Cromwell, D.N., Maliyakal, E.J., Russell, D., Amasino, R.M. (1992) Characterization of a stress-induced, developmentally regulated gene family from soybean. Plant Mol. Biol. 18, 459–466
Domoney, C., Casey, R. (1983) Cloning and characterization of complementary DNA for convicilin, a major seed storage protein in Pisum sativum L. Planta 159, 446–453
Dure III, L., Crouch, M.L., Harada, J., Ho, T-H.D., Mundy, J., Quatrano, R., Thomas, T., Sung, Z.R. (1989) Common amino acid sequence domains among the LEA proteins of higher plants. Plant Mol. Biol. 12, 475–486
Eyal, Y., Sagee, O., Fluhr, R. (1992) Dark-induced accumulation of a basic pathogenesis-related (PR-1) transcript and a light requirement for its induction by ethylene. Plant Mol. Biol. 19, 589–599
Foyer, C.H. (1990) The effect of sucrose and mannose on cytoplasmic protein phosphorylation, sucrose phosphate synthetase activity and photosynthesis in leaf protoplasts from spinach. Plant Physiol. Biochem. 28, 151–160
Fraser, R.S.S., Whenham, R.J. (1982) Plant growth regulators and virus infection: A critical review. Plant Growth Regul. 1, 37–59
Fristensky, B., Horovitz, D., Hadwiger, L.A. (1988) cDNA sequences for pea disease resistance response genes. Plant Mol. Biol. 11, 713–715
Guerrero, F.D., Mullet, J.E. (1988) Reduction of turgor induces rapid changes in leaf translatable RNA. Plant Physiol. 80, 588–591
Harada, J.J., Delisle, A.J., Baden, C.S., Crouch, M.L. (1989) Unusual sequence of an abscisic acid-inducible mRNA which accumulates late in Brassica napus seed development. Plant Mol. Biol. 12, 395–401
Hedley, C.L., Smith, C.M., Ambrose, M.J., Cook, S.K., Wang, T.L. (1986) An analysis of seed development in Pisum sativum. II. The effect of the r locus on growth and development of the seed. Ann. Bot. 58, 371–379
Heikkila, J.J., Papp, J.E.T., Schultz, G.A., Bewley, J.D. (1984) Induction of heat shock protein messenger RNA in maize mesocotyls by water stress, abscisic acid and wounding. Plant Physiol. 76, 270–274
Hetherington, A.M., Quatrano, R.S. (1991) Mechanisms of action of abscisic acid at the cellular level. New Phytol. 119, 9–32
Hong, B., Uknes, S.J., Ho, T-H.D. (1988) Cloning and characterisation of a cDNA encoding a mRNA rapidly induced by ABA in barley aleurone layers. Plant Mol. Biol. 11, 495–506
Iturriaga, E.A., Leech, M.J., Barratt, D.H.P., Wang, T.L. (1993) Two ABA-responsive proteins from pea (Pisum sativum L.) are members of a developmentally regulated, stress-induced gene family. Plant Mol. Biol., in press
Litts, J.C., Colwell, G.W., Chakerian, R.L., Quatrano, R.S. (1987) The nucleotide sequence of a cDNA clone encoding the wheat Em protein. Nucleic Acids Res. 15, 3607–3618
Lotan, T., Fluhr, R. (1990) Xylanase, a novel elicitor of pathogenesis-related proteins in tobacco, uses a non-ethylene pathway for induction. Plant Physiol. 93, 811–817
Marx, G.A. (1978) Argenteum: A mutant under nuclear and extra nuclear control. Pisum Newslett. 10, 34–37
Matton, D.P., Brisson, N. (1989) Cloning, expression and sequence conservation of pathogenesis-related gene transcripts of potato. Mol. Plant Microbe Int. 2, 325–331
Matton, D.P., Bell, B., Brisson, N. (1990) Nucleotide sequence of a pathogenesis-related gene of potato. Plant Mol. Biol. 14, 863–865
Mohapatra, S.S., Poole, R.J., Dhindsa, R.S. (1988) Abscisic acidregulated gene expression in relation to freeze tolerance in alfalfa. Plant Physiol. 87, 468–473
Morris, P.C., Kumar, A., Bowles, D.J., Cuming, A.C. (1990) Osmotic stress and abscisic acid induce expression of the wheat Em genes. Eur. J. Biochem. 190, 625–630
Mundy, J., Chua, N.-H. (1988) Abscisic acid and water-stress induce the expression of a novel rice gene. EMBO J. 7, 2279–2286
O'Farrell, P.H. (1975) High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 250, 4007–4021
Parthier, B. (1990) Jasmonates: Hormonal regulators or stress factors in leaf senescence? J. Plant Growth Regul. 9, 57–63
Peña-Cortés, H., Willmitzer, L., Sánchez-Serrano, J.J. (1991) Abscisic acid mediates wound induction but not developmental-specific expression of the proteinase inhibitor II gene family. Plant Cell 3, 963–972
Pfitzner, U.M., Goodman, H.M. (1987) Isolation and characterisation of cDNA clones encoding pathogenesis-related proteins from tobacco mosaic virus infected tobacco plants. Nucleic Acids Res. 15, 4449–4465
Raynal, M., Depigny, D., Cooke, R., Delseny, M. (1989) Characterisation of a radish nuclear gene expressed during late seed maturation. Plant Physiol. 91, 829–836
Schmelzer, E., Krüger-Lebus, S., Hahlbrock, K. (1989) Temporal and spatial patterns of gene expression around sites of attempted fungal infection in parsley leaves. Plant Cell 1, 993–1001
Singh, N.K., Nelson, D.E., Kuhn, D., Hasegawa, P.M., Bressan, R.A. (1989) Molecular cloning of osmotin and regulation of its expression by ABA and adaption to low water potential. Plant Physiol. 90, 1096–1101
Skriver, K., Mundy, J. (1990) Gene expression in response to abscisic acid and osmotic stress. Plant Cell 2, 503–512
Somssich, I.E., Schmelzer, E., Kawalleck, P., Hahlbrock, K. (1988) Gene structure and in situ transcript localization of pathogenesis-related protein 1 in parsley. Mol. Gen. Genet. 213, 93–98
van de Locht, U., Meier, I., Hahlbrock, K., Somssich, I.E. (1990) A 125 bp promoter fragment is sufficient for strong elicitormediated gene activation in parsley. EMBO J. 9, 2945–2950
Walter, M.H., Liu, J-W., Grand, C., Lamb, C.J., Hess, D. (1990) Bean pathogenesis-related (PR) proteins deduced from elicitorinduced transcripts are members of a ubiquitous new class of conserved PR proteins including pollen allergens. Mol. Gen. Genet. 222, 353–360
Warner, S.A.J., Scott, R., Draper, J. (1992) Characterisation of a wound-induced transcript from the monocot asparagus that shows similarity with a class of intracellular pathogenesisrelated (PR) proteins. Plant Mol. Biol. 19, 555–561
Whitford, P.N., Croker, S.J. (1991) An homogeneous radioimmunoassay for abscisic acid using a scintillation proximity assay technique. Phytochem. Anal. 2, 134–136
Xu, N., Coulter, K.M., Bewley, J.D. (1990) Abscisic acid and osmoticum prevent germination of developing alfalfa embryos, but only osmoticum maintains the synthesis of developmental proteins. Planta 182, 382–390
Zeevaart, J.A.D., Creelman, R.A. (1988) Metabolism and physiology of abscisic acid. Annu. Rev. Plant Physiol. Plant Mol. Biol. 39, 439–473
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barratt, D.H.P., Clark, J.A. A stress-induced, developmentally regulated, highly polymorphic protein family in Pisum sativum L.. Planta 191, 7–17 (1993). https://doi.org/10.1007/BF00240890
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00240890