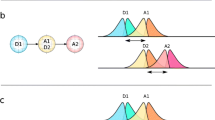
Abstract
The need to describe and understand signaling pathways in live cell is seen as a primary route to identifying and developing targeted medicines. Signaling cascade is also seen as a complex communication and involves interactions between multiple interconnecting proteins. Where subcellularly and how different proteins interact need to be preserved during investigation. Furthermore, these complex events occurring simultaneously may lead to a single or multiple end point or cell function such as protein synthesis, cell cytoskeleton formation, DNA damage repair, or autophagy. There is therefore a need of real-time noninvasive methods for protein assays to enable direct visualization of the interactions in their natural environment and hence overcome the limitations of methods that rely on invasive cell disruption techniques. Förster resonance energy transfer (FRET) coupled with fluorescence lifetime imaging microscopy (FLIM) is an advanced imaging method to observe protein–protein interactions at nanometer scale inside single living cells in real-time. Here we describe the development and use of two-channel pulsed interleave excitation (PIE) for multiple protein interactions in the mTORC1 pathway. The proteins were first tagged with multiple color fluorescent protein derivatives. The FRET-FLIM combination means that the information gained from using standard steady-state FRET between interacting proteins is considerably improved by monitoring changes in the excited-state lifetime of the donor fluorophore where its quenching in the presence of the acceptor is evidence for a direct physical interaction.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Beretta L, Gingras AC, Svitkin YV et al (1996) Rapamycin blocks the phosphorylation of 4E-BP1 and inhibits cap-dependent initiation of translation. EMBO J 15(3):658–664
Brunn GJ, Hudson CC, Sekulić A et al (1997) Phosphorylation of the translational repressor PHAS-I by the mammalian target of rapamycin. Science 277(5322):99–101
Gingras AC, Kennedy SG, O'Leary MA et al (1998) 4E-BP1, a repressor of mRNA translation, is phosphorylated and inactivated by the Akt(PKB) signaling pathway. Genes Dev 12(4):502–513
Holz MK, Ballif BA, Gygi SP et al (2005) mTOR and S6K1 mediate assembly of the translation preinitiation complex through dynamic protein interchange and ordered phosphorylation events. Cell 123(4):569–580
Arif A, Terenzi F, Potdar AA et al (2017) EPRS is a critical mTORC1–S6K1 effector that influences adiposity in mice. Nature 542:357–361
Caccamo A, Branca C, Talboom JS et al (2015) Reducing ribosomal protein S6 kinase 1 expression improves spatial memory and synaptic plasticity in a mouse model of Alzheimer ‘s disease. J Neurosci 35:14042–14056
Kim SY, Baik KH, Baek KH et al (2014) S6K1 negatively regulates TAK1 activity in the toll-like receptor. Mol Cell Biol 34:510–521
Marabita M, Baraldo M, Solagna F et al (2016) S6K1 is required for increasing skeletal muscle force during hypertrophy article S6K1 is required for increasing skeletal muscle force during hypertrophy. Cell Rep 17:501–513
Choo AY, Yoon SO, Kim SG et al (2008) Rapamycin differentially inhibits S6Ks and 4E-BP1 to mediate cell-type-specific repression of mRNA translation. Proc Natl Acad Sci U S A 105(45):17414–17419. https://doi.org/10.1073/pnas.0809136105
Choo AY, Blenis J (2009) Not all substrates are treated equally implications for mTOR, rapamycin-resistance and cancer therapy. Cell Cycle 8:567–572. https://doi.org/10.4161/cc.8.4.7659
Förster T (1948) Zwischenmolekulare Energiewanderung und Fluoreszenz [Intermolecular energy migration and fluorescence]. Annalen der Physik (in German). 437: 55–75. B För s t e r T (1946) Energiewanderung und Fluoreszenz. Naturwissenschaften 33:166–175? (Translation: by Klaus Suhling). J Biomed Opt 17:011002
Pawley JB (2006) Handbook of biological confocal microscopy, 3rd edn. Springer, New York, NY, pp 1–985. https://doi.org/10.1007/978-0-387-45524-2_8. ISBN 978-0-387-25921-5
Fábián ÁI, Rente T, Szöllősi J et al (2010) Strength in numbers: effects of acceptor abundance on FRET efficiency. ChemPhysChem 11:3713–3721. https://doi.org/10.1002/cphc.201000568
Maliwal BP, Raut S, Fudala R et al (2012) Extending Förster resonance energy transfer measurements beyond 100 Å using common organic fluorophores: enhanced transfer in the presence of multiple acceptors. J Biomed Opt 17:11006. https://doi.org/10.1117/1.JBO.17.1.011006
Walczewska-Szewc K, Bojarski P, d’Auria S (2013) Extending the range of FRET—the Monte Carlo study of the antenna effect. J Mol Model 19:4195–4201. https://doi.org/10.1007/s00894-013-1810-3
Koushik SV, Blank PS, Vogel SS (2009) Anomalous surplus energy transfer observed with multiple FRET acceptors. PLoS One 4(11):e8031. https://doi.org/10.1371/journal.pone.0008031
Bunt G, Wouters FS (2017) FRET from single to multiplexed signaling events. Biophys Rev 9:119–129. https://doi.org/10.1007/s12551-017-0252-z
Wolfgang B (2015) Advanced single photon counting applications. Springer, New York, pp 1–624. https://doi.org/10.1007/978-3-319-14929-5
Botchway SW, Scherer KM, Hook S et al (2015) A series of flexible design adaptations to the Nikon E-C1 and E-C2 confocal microscope systems for UV, multiphoton and FLIM imaging. J Microsc 258(1):68–78. https://doi.org/10.1111/jmi.12218
Scully AD, Macrobert AJ, Botchway S et al (1996) Development of a laser-based fluorescence microscope with subnanosecond time resolution. J Fluoresc 6(2):119–125. https://doi.org/10.1007/BF00732051
Suhling K, Siegel J, Phillips D et al (2002) Imaging the environment of green fluorescent protein. Biophys J 83(3589–3595):2002
Marcu L, French PMW, Elson DS (2014) Fluorescence lifetime spectroscopy and imaging—principles and applications in biomedical diagnostics. CRC Press, Boca Raton, Florida, pp 1–570. SBN 9781439861677—CAT# K12851
Estandarte AK, Botchway S, Lynch C et al (2016) The use of DAPI fluorescence lifetime imaging for investigating chromatin condensation in human chromosomes. Sci Rep 6:31417. https://doi.org/10.1038/srep31417
Yadav RB, Burgos P, Parker AW et al (2013) mTOR direct interactions with Rheb-GTPase and raptor: sub-cellular localization using fluorescence lifetime imaging. BMC Cell Biol 14:3. https://doi.org/10.1186/1471-2121-14-3
Stubbs CD, Botchway SW, Slater SJ et al (2005) The use of time-resolved fluorescence imaging in the study of protein kinase C localisation in cells. BMC Cell Biol 6(1):22
Jeshtadi A, Burgos P, Stubbs CD et al (2010) Interaction of poxvirus intracellular mature virion proteins with the TPR domain of kinesin light chain in live infected cells revealed by two-photon-induced fluorescence resonance energy transfer fluorescence lifetime imaging microscopy. J Virol 84(24):12886–12894. https://doi.org/10.1128/JVI.01395-10
Osterrieder A, Carvalho CM, Latijnhouwers M et al (2009) Fluorescence lifetime imaging of interactions between Golgi tethering factors and small GTPases in plants. Traffic 10(8):1034–1046. https://doi.org/10.1111/j.1600-0854.2009.00930.x
Nettleship JE, Assenberg R, Diprose JM et al (2010) Recent advances in the production of proteins in insect and mammalian cells for structural biology. J Struct Biol 172(1):55–65. https://doi.org/10.1016/j.jsb.2010.02.006
Acknowledgments
This work was supported by a BBSRC iCASE PhD studentship (BB/L016052/1) to AA. We thank STFC for funding access to the Central Laser Facility. This work also acknowledges Professor Raymond Owens (PP-UK) for providing support and plasmid vectors for the cloning work.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ahmed, A., Schoberer, J., Cooke, E., Botchway, S.W. (2021). Multicolor FRET-FLIM Microscopy to Analyze Multiprotein Interactions in Live Cells. In: Poterszman, A. (eds) Multiprotein Complexes. Methods in Molecular Biology, vol 2247. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1126-5_16
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1126-5_16
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1125-8
Online ISBN: 978-1-0716-1126-5
eBook Packages: Springer Protocols